Abstract
Introduction
Plants were widely used in folk medicine all over the world, which was predicated on centuries-old customs and beliefs. They are part of the tradition of every country and involve practices that have been passed down from generation to generation as folk medicines and have finally developed as modern medicinal treatments after scientific analysis of their effects. 1
Historically, the plant kingdom has been a rich source of novel lead compounds that contribute to a variety of pharmacological and therapeutic activities. Around 25% of pharmaceutical drugs in the modern era were derived from plants. 2
However, due to the numerous intrinsic difficulties associated with isolating and identifying pharmaceuticals derived from natural products, most pharmaceutical companies have shifted their focus toward synthetic molecules as a source of new drugs because of their effectiveness and ease of production. Unfortunately, the low rate of new drugs entering the pharmaceutical market, together with unanticipated negative side effects, contraindications, and interactions between synthetic drugs, fell short of aspirations for global health3-7. Additionally, as consumers’ knowledge of nutrition and health issues, as well as the potential and benefits of medicinal plants and their metabolites, increased, so did their interest in and demand for medicinal plants for use in food, medicine, and other applications. 8 The World Health Organization estimated that around 80% of people worldwide use medicinal plant products to treat an array of diseases. 9 It has confirmed the necessity of using modern technologies and proper standards to ensure the quality of medicinal plant products. 10 Consequently, global attention once again shifted to the development of drugs based on natural products. So there have been an increasing number of publications on medicinal plants used in folk medicine as a significant approach to developing novel drugs. 11
Phlomis longifolia Boiss. & C.I. Blanche. belongs to the genus Phlomis and is a large genus in the family Lamiaceae, with more than 100 species distributed throughout the continents of Europe, Asia, and North Africa. 12 It is an herbaceous plant with entire, opposite, crossed, and wrinkled dark green leaves. Whorls of bright yellow flowers are arranged around the stems, which are typically square in section with rounded corners, though tomentum on stems can give the appearance that they are circular. The calyx is tubular and has five visible veins. It has five teeth, all of which are equal. The flowering period is from May to July. It grows in forests and grasslands. It is native to Syria, Lebanon, Turkey, and Cyprus, and is spread in Europe and North Africa. 13 Its flowers are sweet and are consumed directly through sucking, which is why it is locally called the Al-Masas plant.
Plants from the genus Phlomis are frequently used in traditional medicine across the globe. They are used as a boiling or soaking herbal tea to treat fever, cough, and colds, as well as stomach, intestinal, and abdominal pain and digestive issues, and as a sedative.14–18 They can also be applied as pastes or powders to treat burns, wounds, and skin diseases19-21. Locally, P. longifolia flowers are used to treat stomach pain and indigestion. The plant's leaves and calyxes are also used to treat diabetes and kidney stones. Additionally, one of the most popular applications of the plant's leaves is to clean cooking utensils of dirt that has accumulated on them.
Despite P. longifolia's numerous medical applications, there aren't many investigations of the plant's chemical composition. Therefore, the aim of this work was to identify the chemical components with potential pharmacological properties of essential oils extracted from the aerial parts of the Syrian P. longifolia plant using gas chromatography coupled to mass spectrometry (GC-MS) technology for the first time.
Results and Discussion
GC-MS Analysis of Essential Oils
The hydrodistillation of aerial parts of P. longifolia produced yellowish oil with a yield of 0.14%, 0.075%, and 0.19% for flowers, calyxes, and leaves, respectively. A total of 63, 61, and 48 compounds (Tables 1, 2, and 3) were identified by GC-MS for both flowers, calyxes, and leaves, which represent 98.0%, 97.1%, and 97.9% of total oil, respectively. Moreover, the total ion chromatogram of the samples is shown in Fig. S1.
Chemical Composition of the Essential Oil Extracted from the Flowers of P. longifolia.

RT: Retention time (minutes), M.F.: Molecular formula, M.W.: Molecule weight, Area%: Percentage Peak Area, Class: Classification compound, H: Hydrocarbons, M: Monoterpenes, S: Sesquiterpenes, D: Diterpenes, F: Fatty acids & Esters, O: Other oxygenated compounds.
Chemical Composition of the Essential Oil Extracted from the Calyxes of P. longifolia.
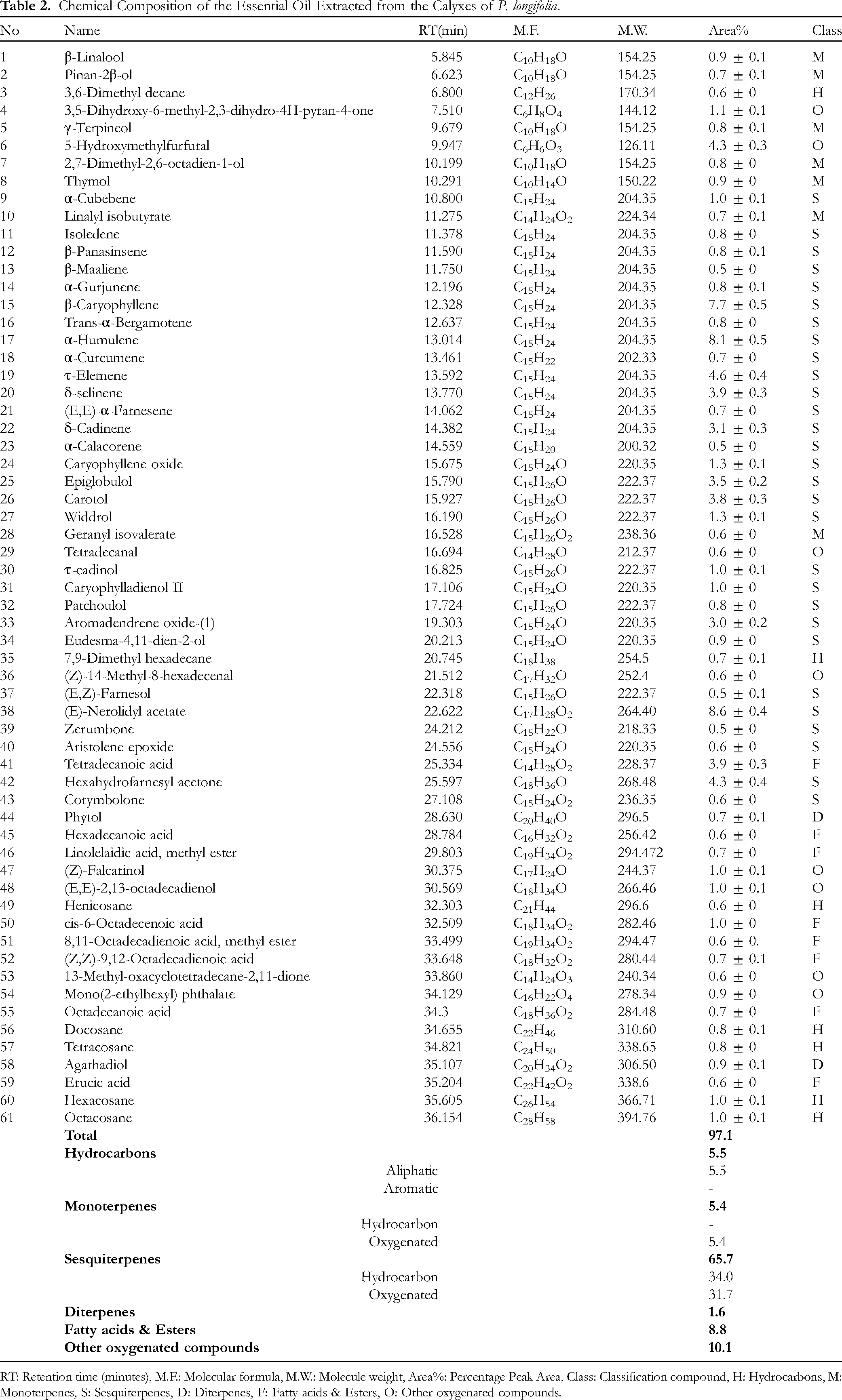
RT: Retention time (minutes), M.F.: Molecular formula, M.W.: Molecule weight, Area%: Percentage Peak Area, Class: Classification compound, H: Hydrocarbons, M: Monoterpenes, S: Sesquiterpenes, D: Diterpenes, F: Fatty acids & Esters, O: Other oxygenated compounds.
Chemical Composition of the Essential Oil Extracted from the Leaves of P. longifolia.

RT: Retention time (minutes), M.F.: Molecular formula, M.W.: Molecule weight, Area%: Percentage Peak Area, Class: Classification compound, H: Hydrocarbons, M: Monoterpenes, S: Sesquiterpenes, D: Diterpenes, F: Fatty acids & Esters, O: Other oxygenated compounds.
The major compounds in the flower's essential oil were: widdrol (29.8%), β-caryophyllene (9.7%), and E-nerolidyl acetate (5.8%). The mass spectra of the major compounds are shown in Fig. S2. Where the major compounds in the calyx's essential oil were: (E)-nerolidyl acetate (8.6%), α-humulene (8.1%), and β-caryophyllene (7.7%). The mass spectra of the major compounds are shown in Fig. S3. Whereas the major compounds in the leave's essential oil were: (E)-nerolidyl acetate (11.4%), β-caryophyllene (9.5%), α-amorphene (8.8%), caryophyllene oxide (6.7%), α-humulene (5.3%), and 3,5-Dihydroxy-6-methyl-2,3-dihydro-4H-pyran-4-one (5.2%). The mass spectra of the major compounds are shown in Fig. S4.
In general, isoprenoids constitute the majority of the chemical composition of essential oils. This was shown by the chemical composition of the essential oils extracted from P. longifolia's aerial parts in this work. Where the chemical composition was dominated by sesquiterpene compounds (77.3% for flowers, 65.7% for calyxes, and 77.1% for leaves). In the flower's essential oil, the highest content was the share of oxygen sesquiterpenes (51.9%), represented by the major compounds: widdrol (29.8%), (E)-nerolidyl acetate (5.8%), caryophyllene oxide (2.7%), and corymbolone (2.6%). While the hydrocarbon sesquiterpene compounds were found in (25.4%), they were represented by the following major compounds: β-caryophyllene (9.7%), and α-humulene (4.0%). While in the calyx's essential oil, hydrocarbon and oxygen compounds were found in close proportions (34.0% and 31.7%, respectively). The oxygen sesquiterpene compounds were represented by the following major compounds: (E)-nerolidyl acetate (8.6%), hexahydrofarnesyl acetone (4.3%), carotol (3.8%), epiglobulol (3.5%), and aromadendrene oxide-(1) (3.0%), whereas hydrocarbon sesquiterpenes were represented by the following major compounds: α-humulene (8.1%), β-caryophyllene (7.7%), τ-elemene (4.6%), δ-selinene (3.9%), and δ-cadinene (3.1%). As for the leaves's essential oil, the highest value of oxygenic sesquiterpenes was (43.9%) and was represented by the following major compounds: (E)-nerolidyl acetate (11.4%), caryophyllene oxide (6.7%), aromadendrene oxide-(1) (4.5%), 3-hydroxy-5,6-epoxy-β-ionone (4.2%), globulol (4.1%), hexahydrofarnesyl acetone (3.8%), and caryophylladienol II (2.5%), while the hydrocarbon sesquiterpenes (33.2%) were represented by the following major compounds: β-caryophyllene (9.5%), α-amorphene (8.8%), α-humulene (5.3%), and δ-cadinene (2.4%). The monoterpenoid and diterpenoid were found in small amounts, and the compounds with the greatest value from these classes were: β-linalool (1.7% in flowers, 0.9% in calyxes, and 1.0% in leaves) for the monoterpenoid, and phytol (0.4% in flowers, 0.7% in calyxes, and 1.6% in leaves) for the diterpenoid. Furthermore, fatty acids & esters, and non-terpene hydrocarbon compounds were also present in small amounts. Octacosane had the highest value for these compounds in the hydrocarbon class (0.3% for flowers, 1.0% for calyxes, and 1.3% for leaves); as for the class of fatty acids and their esters, tetradecanoic acid had the highest value (1.7% for flowers, 3.9% for calyxes, and 2.1% for leaves).
Additionally, the amount of non-terpene oxygen compounds in essential oils was low, and the major compounds in this class are: 3,5-dihydroxy-6-methyl-2,3-dihydro-4H-pyran-4-one (2.0% for flowers, 1.1% for calyxes, 5.2% for leaves), and 5-hydroxymethylfurfural (3.5% for flowers, 4.3% for calyxes, 1.1% for leaves).
Comparing the chemical composition of the oils demonstrates how similar the chemical content is among the essential oils extracted from the aerial parts of the P. longifolia plant. The majority of the compounds were the same but differed in concentration. For example, the main compound widdrol was found with a high value of (29.8%) in the flowers, while it was found with low values in the leaves and calyxes (0.9% and 1.3%, respectively). The compound α-amorphene was found as a major compound in the leaves (8.8%), while its value decreased in the flowers (1.9%), while it was not observed in the calyxes.
Several investigations have been conducted on the chemical composition of essential oils from different species of Phlomis spp. The studies have included the following species growing in Turkey (P. linearis, P. russeliana, P. grandiflora var. grandiflora, P. lunariifolia, P. amanica, P. monocephala, P. sieheana, P. armeniaca Willd, P. bourgaei, P. leucophracta, and P. chimerae),22–27 Iran (P. lanceolata, P. anisodonta, P. bruguieri, P. olivieri),28–30 Greece (P. fruticose, P. cretica, P. samia),
31
Cyprus (P. brevibracteata, P. cypria),
32
Tunisia (P. crinite),
33
Algeria (P. bovei).
34
Although the yield of essential oils varies greatly (0.05-1%), as observed in these investigations, sesquiterpenes dominate in their chemical composition. As Table 4 indicates, both compounds, β-caryophyllene and germacrene D, were found to be major compounds in large amounts in the majority of the Phlomis species plants that were investigated. Among the sesquiterpenes found as major compounds were farnesene in the oils of P. linearis (6.6%),
22
P. lunariifolia (6.5%), P. amanica (8.3%), and P. sieheana (23.4%),
24
P. armeniaca Willd (7.24%),
25
P. bourgaei (15.88%),
26
P. lanceolata (10.5%),
28
and P. samia (20.7%),
31
caryophyllene oxide in the oils of P. linearis (9.2%),
22
P. russeliana (8.1%),
23
P. armeniaca Willd (13.35%),
25
P. chimerae (4.8%),
27
P. samia (3.2%),
31
and P. brevibracteata (26.0-7.2%),
32
and bicyclogermacrene in the oils of P. grandiflora var. grandiflora (4.9%), P. amanica (10.7%),
24
P. lanceolata (5.9%), P. bruguieri (4.1%),
28
and P. olivieri (10.2%).
30
In addition to sesquiterpenes, monoterpenes were found as major compounds in some of the Phlomis species oils. Among these compounds are α-pinene in the oils of P. leucophracta (19.2%) and P. chimerae (11.0%),
27
P. lanceolata (8.7%),
28
P. bruguieri (6.8%),
29
P. olivieri (11.7%),
30
P. fruticose (12.6%), and P. cretica (9.4%).
31
limonene in the oils of P. grandiflora var. grandiflora (2.7%), P. leucophracta (11.0%), P. chimerae (5.5%),
27
and P. cretica (7.1%).
31
Despite the fact that sesquiterpene compounds constitute the majority of the essential oil composition in most Phlomis species, it is evident that these species differ chemically from one another. The main compounds in the oil of P. grandiflora var. grandiflora collected from the northern and southern regions of Turkey were found to be
Phlomis Spp. Containing β-Caryophyllene and Germacrene D.

The chemical composition of essential oils is influenced by a variety of factors, both internal and external. One of these important internal factors is the variation in the plant parts because different plant parts have various secretory structures that distribute irregularly throughout the plant body. 35 This could explain the quantitative and qualitative diversity in chemical composition between flowers, calyxes, and leaves. The physiological stage of the plant during which the plant samples were taken (the flowering period), when the plant is susceptible to attack by pollinators and diseases, is one of the external factors that influences the chemical composition of essential oils. Furthermore, the genotype of the plant has the greatest influence on the production of secondary metabolites, as plants subjected to harsh environmental stresses and ongoing external conditions can change their genotype and allow for alternative biosynthesis pathways, which results in a diversity of secondary metabolite production that is both qualitatively and quantitatively varied. 36 This could account for the variations in the chemical composition of essential oils extracted from various Phlomis plant species.
Evaluation of the Biological Efficacy of the Identified Compounds by GC-MS
Table 5 shows the biological activity of the identified compounds in the essential oils from the aerial parts of P. longifolia. Among the (63, 61, and 48) identified compounds, (50, 45, and 41) compounds with different biological activities were found, according to the published literature. Which represent (79.4%, 73.8%, and 85.4%) of the total compounds for flowers, calyxes, and leaves, respectively. (43, 37, and 34) antimicrobial active compounds were found, representing (86.0%, 82.2%, and 82.9%) of all active compounds. Furthermore, (30, 26, and 24) compounds with antioxidant activity were found, representing (60.0%, 57.8%, and 58.5%) of the total active compounds. Also, it was found that (27, 26, and 20) compounds have anti-cancer activity, representing (54.0%, 57.8%, and 48.8%) of all active compounds. Moreover, it was also found (17, 18, and 15) compounds that have anti-inflammatory activity, which represent (34.0%, 40.0%, and 36.9%) of the total active compounds for each of the flowers, calyxes, and leaves, respectively. In addition to the presence of several compounds with activities related to the nervous system and the cardiac system, several compounds have also been found to be effective as insecticides and herbicides.
Literature Biological Activity of the Identified Active Compounds in Essential Oils Extracted from the Aerial Parts of P. longifolia.

Compounds identified in flower's essential oil, bcompounds identified in the calyx's essential oil, ccompounds identified in the leave's essential oil.
Essential oils are a significant and abundant source of biologically active compounds that can be used in various pharmaceutical science fields. The primary biological function attributed to essential oils is antioxidant activity. The second biological function is antimicrobial, followed by insecticide and repellent activity. While the least active are anti-cancer, anticonvulsant, anti-inflammatory, etc. 167 However, in this work, the results of the research in the published literature on the biological activity of the essential oil components of the aerial parts of P. longifolia shown in Table 5 were interesting. Besides the richness of these essential oils in compounds that have antioxidant and antimicrobial activities, compounds that have anti-cancer and anti-inflammatory activities were found in an appreciable percentage. While a few compounds were found to have insecticide and insectifuge efficacy as well as activity related to the nervous system, heart, and skin. Therefore, it is possible to forecast the potential of essential oils extracted from the aerial parts of P. longifolia as a potential preventive or therapeutic agent for various diseases brought on by microbes or induced by oxidative stress. Furthermore, it may be used as a natural preservative instead of synthetic preservatives.
To our knowledge, there have been no previous chemical or biological studies on essential oils extracted from the aerial parts of P. longifolia. Consequently, we were unable to compare the chemical composition of our study's results with those of previous investigations. particularly considering that, given the capabilities at hand, we only used one method to verify the chemical composition (by comparing mass spectra for compounds with those in the device library). Therefore, this study is considered an initial investigation that may be used to support future chemical research on this plant and from various locations to verify its chemical composition and investigate its biological activities with the aim of developing new drugs as well as employing it in possible pharmaceutical and food applications.
Conclusions
In this work, the chemical composition of essential oils extracted from the aerial parts of P. longifolia was determined for the first time by GC-MS. Essential oils were rich in phytochemical compounds, especially sesquiterpenes, which were characterized by a diversity of biological activities. Therefore, such compounds provide leads for future research on biological activities of commonly used medicinal plants for their nutritional and therapeutic purposes.
Materials and Methods
Instrumentation and Chemicals
GC-MS (Agilent −6890, USA), clevenger-type apparatus (Shamlab, Syria), laboratory heating (Heraeus Wittmann, Germany), sensitive scale (Sartorius, Germany), rotary evaporator (Heidolph Laborota 4000, Germany), laboratory glassware (Isolab, Germany), hexane (Honeywell, Germany), sodium sulphate (ANH.) (TITAN BIOTECH LD, India), filter paper (Whatman no.1, USA).
Plant Material
The aerial parts (flowers, calyxes, and leaves) of the P. longifolia Boiss. & C.I. Blanche. wild plant were collected during the flowering period in June 2022 from a mountainous area located at an altitude of about 900 m (Latakia Province-Syria). A voucher specimen (No. HFC 950) has been deposited in the herbarium of the faculty of science (Tishreen University, Latakia, Syria). Plant specimens were identified by Prof. Dina Haddad from that faculty. After washing and drying in the shade, they were ground well and stored in an opaque glass bowl until extraction time.
Essential oil Extraction
The essential oils were extracted by hydrodistillation of dried and ground plant material (flowers, calyxes, and leaves) for 5 h (100 g of each sample in 500 ml of distilled water) using a Clevenger-type apparatus.
168
The fractions of oils were collected in hexane and dried using anhydrous sodium sulfate. After filtration, the solvent was removed by passing a stream of nitrogen, and the oils were stored at +4 °C until analysis. The weight of the obtained oils was
GC-MS Analysis
GC-MS analysis was carried out using an Agilent −6890 gas chromatograph coupled to a Hewlett Packard-5975 mass spectrometry. A HP-5 column (30 m × 0.25 mm, film thickness 0.25 μm), stationary phase: 5% phenyl methyl siloxane, was used. The column temperature was programmed at 70 °C, then increased to 280 °C at 4 °C/min, and held for 10 min. Helium was used as a carrier gas, at a flow rate of 1.2 ml/min. The injections were performed in splitless mode at 250 °C. Mass spectra were recorded with an ionization energy of 70 eV and an interface temperature of 230 °C.
Identification of Components
The compounds were identified based on the interpretation of the mass spectra of GC-MS using the databases of NIST and WILEY, where the mass spectra of the unknown components were compared with the spectra of the known components stored in the NIST 05a. L, NIST 02. L, and WILEY 7.1 libraries.
Statistical Analysis
All the experiments were performed in triplicate. For each result, the data were summarized as the mean ± standard deviation (SD). Statistical analysis was performed using Microsoft Excel (version 2013).
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241282866 - Supplemental material for Essential Oils from Phlomis longifolia Boiss. & C.I. Blanche. Aerial Parts (Flowers, Calyxes, Leaves): GC-MS Analyzes and Biological Properties
Supplemental material, sj-docx-1-npx-10.1177_1934578X241282866 for Essential Oils from Phlomis longifolia Boiss. & C.I. Blanche. Aerial Parts (Flowers, Calyxes, Leaves): GC-MS Analyzes and Biological Properties by Muhannad Hasan, Imad Hwija and Yaseer Mossa in Natural Product Communications
Footnotes
Acknowledgments
We thank Dr. Fadi Younis (Friedrich-Schiller-University Jena) for review the manuscript.
Author Contributions
Conceptualization, M.H., I.H. and Y.M.; methodology, M.H. and I.H.; validation, M.H. and I.H.; formal analysis, M.H. and I.H.; investigation, M.H, I.H. and Y.M.; data curation, M.H..; writing—original draft preparation, M.H.; writing—review and editing, M.H., I.H. and Y.M.; supervision, I.H. and Y.M. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
