Abstract
This is a visual representation of the abstract.
Introduction
Infectious illnesses are among the most common diseases and largest motive of mortality. 1 In recent years, the use of medicinal plants in alleviating various diseases, including infectious diseases have considerably increased. 2 Among these plants is Daniellia oliveri Rolfe a well-known plant of the Fabaceae family. In Africa, D. oliveri is very important in wooded savannas. It has a conical crown whose shape is generally tapered. It has up to 18.29 m-24.38 m tall with about 3.66 m in circumference, and the bark is pale gray and thick.3,4 This African tree, is extensively found in Benin, Nigeria and Gambia. 5
The stem, bark and leaves of D. oliveri are commonly used in the treatment of dysentery, syphilis, ring worms, wounds, typhoid fever, heart diseases, high blood, pains, asthma, skin infection and malaria.5,6 In Nigeria the root is considered diuretic and a decoction is taken to treat veneral diseases, absence of menses, anxiety, jaundice, food poisoning, and skin diseases.5,6 In northern Nigeria, leaves of D. oliveri are used to treat diabetes, gastrointestinal problems, diarrhea, as a diuretic and an aphrodisiac, the bark and the resin are used as a mosquito repellent. 7 In the Adamawa region of Cameroon, D. oliveri is used in the treatment of skin infections, menstrual problems, wounds, diabetes and yellow fever. Previous phytochemical and pharmacological studies performed on D. oliveri led to the identification of triterpenoid, phenol, anthraquinone oxalate with antioxidant, antibacterial and antifungal compounds,5,6 alkaloids with anti-inflammatory and antiparasitic potential,5,6 tannin, phylate, diterpenoid, quinine, polyphenols, anthraquinone oxalate, saponins and steroid with antirheumatic, antidiabetic, antiseptic, diuretic, antiglycation and hypotensive properties.8,9 In our permanent search for secondary metabolites with antimicrobial value from Cameroonian medicinal plants,10–13 the investigation of the methanol extract of the root bark of D. oliveri was undertaken. Moreover, the extract, fractions and isolated compounds were investigated for their antimicrobial activity against Pseudomonas aeruginosa, Escherichia coli, Staphylococcus aureus, Streptococcus sp., Candida albicans and Candida tropicalis.
Results
General Information of the Isolated Compounds
Compounds

Structures of the isolated compound
The UV spectrum denoted a maximum absorption at 235 nm, while IR spectrum revealed absorption bands, indicating the existence of a γ-substitute furan ring (ether C-O at 1070 cm−1 and aromatic C = C at 1470 cm−1), unsaturated vinyldiene (-C = CH2 at 1664 cm−1) and carboxylic acid (C-O, C = O and O-H vibrating respectively at 1270, 1720 and 2960 cm−1) functional groups. The 1H and 13C NMR spectrometric properties were globally superimposable onto those of polyalthic acid (
The 1HNMR (CDCl3, 500 MHz)δ(ppm): 1.56/1.31 (2H, m, H-1a, H-1b), 1.53/1.43 (2H, m, H-2a, H-2b), 1.90/1.65 (2H, m, H-3a, H-3b),1.31 (1H, m, H-5), 1.96/1.89 (2H, m, H-6a, H-6b), 2.14/2.12 (2H, m, H-7a, H-7b),1.72(1H, m, H-9),1.61 (2H, m, H-11),2.55 (2H, m, H-12),6.24 (1H, s, H-14),7.33 (1H, m, H-15),7.18 (2H, s, H-16), 4.88/4.56 (2H, s, H-17a, H-17b),1.22 (3H, s, H-19),0.59 (3H, s, H-20) (Fig. S1); 13C NMR (CDCl3, 125 MHz)δ(ppm): 40.4 (CH2, C-1),19.8 (CH2, C-2),37.2 (CH2, C-3),44.2 (C, C-4),56.2 (CH, C-5),24.3 (CH2, C-6),38.7(CH2, C-7),147.8 (C, C-8),55.2 (CH, C-9),39.0 (C, C-10),28.9 (CH2, C-11),26.0 (CH2, C-12),125.4 (CH, C-13),111.0 (CH2, C-14),142.6(CH2, C-15),138.7 (CH2, C-16),106.5 (CH, C-17),184.5 (C, C-18),28.9 (CH3, C-19),12.7(CH3, C-20) (Fig. S2).
The above mentioned information referred to diterpene type labdane derivative.9,15 The molecular weight was 316.2 corresponding to the molecular formula C20H28O3 corresponding to polyalthic acid and its stereo-isomer daniellic acid. 15
Finally, the absolute stereochemistry of compound

Molecular structure of the crystals of compound
All the other isolated compounds
Antimicrobial Assay
The crude extract demonstrated selective antimicrobial activity against the tested micro-organisms. It showed interesting activity on the tested strains of E. coli with MIC value of 32 μg/mL. The evaluated fractions (D1–D5) showed various degrees of antimicrobial activity (8 ≤ MIC ≤ 1024 µg/mL) with the best MIC value observed for fraction D5 on E. coli (MIC = 16 µg/mL). On yeasts, D1 and D2 appeared to be the most active fractions with MIC value of 512 µg/mL on C. albicans which showed more sensitivity compared to C. tropicalis (Table 1). As far as compounds are concerned, they showed various antimicrobial activities. Polyaltic acid
Minimum Inhibitory Concentrations (MIC) and Minimum Fungicidal Concentrations (MFC) of Crude Extract, Fractions, and Isolated Compounds in μg/mL.
Crystal Structure Report for Compound 1
Compound
Structure Feature
The crystal structure of title compound
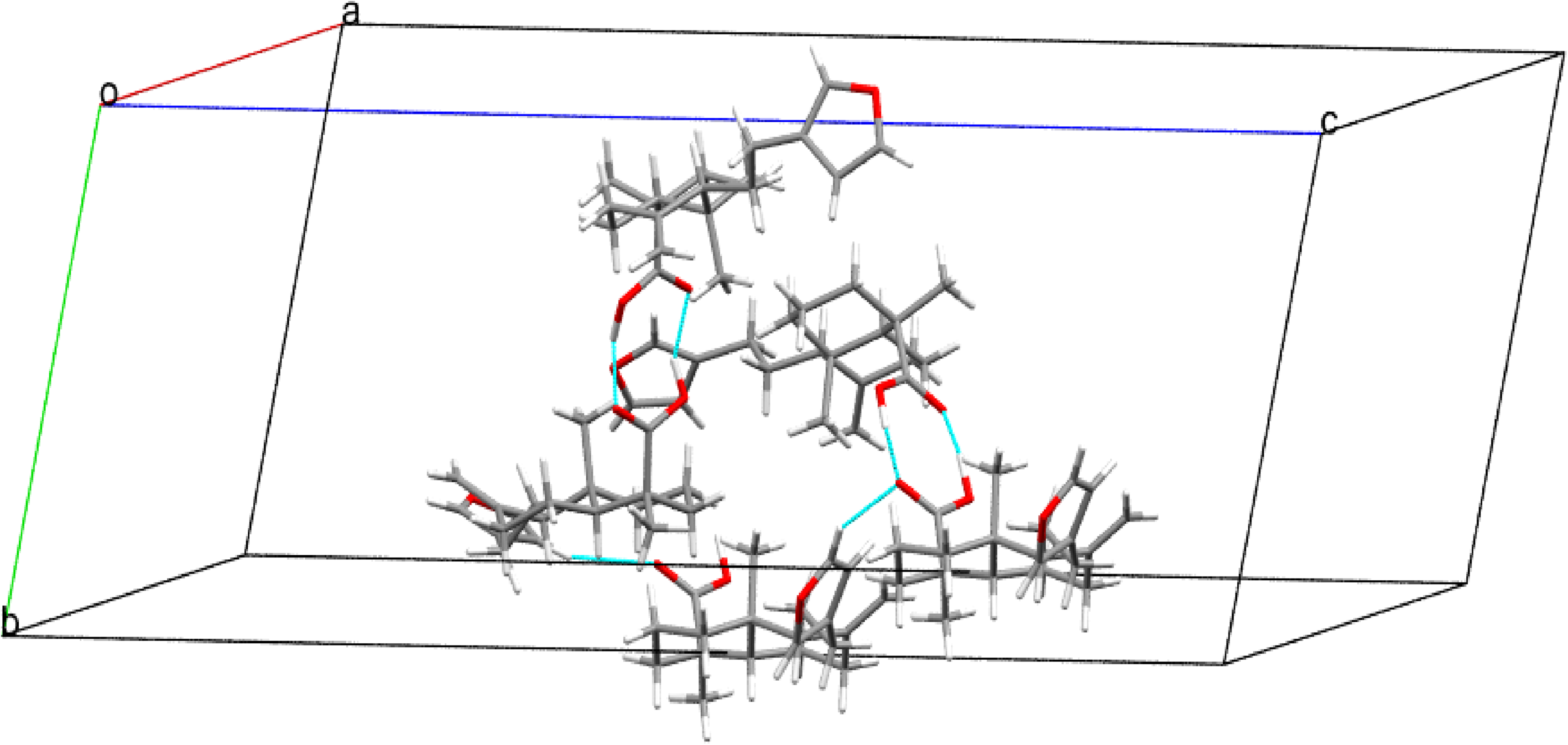
The unit cell packing diagram of compound
Crystallographic Data of Compound

Hydrogen Bonding Table for Compound

Symmetry Codes: x-1,y,z#1, x + 1, y, z#2.
Discussion
The crude extract showed selective antimicrobial activity against the tested micro-organisms. According to Kuete, 1 the activity of extracts is classified as significant when MIC < 100 µg/mL, as moderate when 100 < MIC <625 µg/mL and as weak when (MIC > 625 µg/mL). Based on this classification, the crude extract showed significant activity on the tested strains of E. coli. Previous studies revealed the antimicrobial activities of Daniellia oliveri. In fact, it was shown that the leaf essential oils of D. oliveri showed marginal antibacterial activity against Bacillus cereus, Cutibacterium acnes, Staphylococcus aureus, Staphylococcus epidermidis, Streptococcus pyogenes, Pseudomonas aeruginosa, and Serratiamarcescens, and antifungal activity against Aspergillus fumigatus, Aspergillus niger, Cryptococcus neoformans, Microsporum canis, Microsporum gypseum, Trichophyton mentagrophytes, Trichophyton rubrum, and Candida albicans while D. oliveri bark essential oil showed notable antifungal activity against Aspergillus niger and Trichophyton rubrum with a minimum inhibitory concentration (MIC) of 78.1 µg/mL for each. 7 These findings are different from the obtained results and could be justified by the difference in the extract type. 20
The tested fractions (D1–D5) showed various degrees of antimicrobial activity (8 ≤ MIC ≤ 1024 µg/mL) with the significant MIC value observed for fraction D5 on E. coli. These results are different from previous findings which revealed that four chromatographic fractions of the aqueous ethanolic extract of the leaves of Daniellia oliveri showed activity against Staphylococcus aureus while one chromatographic fraction showed significant activity against the fungus Tricophyton rubrum with an inhibition diameter of 10.5 mm.
21
Moreover, to the contrary of our findings, they showed no activity against Staphylococcus aureus, Escherichia coli, Pseudomonas aeruginosa. This difference in activity could be astribed to the difference in the testing methods because they used disk dilution methods against broth microdillution method used in this study. It could also be due to the difference in the type of extraction solvent which can modify the composition of the extract hence the biological activity.
20
On Bacteria strains, D5 showed a significant MIC value on E. coli. Compounds showed various antimicrobial activities. According to Kuete,
1
the activity of pure compounds was classified as significant when (MIC < 10 µg/mL), as moderate when (10 < MIC <100 µg/mL) and as weak when (MIC > 100 µg/mL). Polyaltic acid
Limitations
The tested compounds were not in sufficient quantity to allow the study of their activities on more strains of microorganisms.
Conclusion
The results obtained in this research revealed the presence of antimicrobial phytochemical components from the root bark of D. oliveri. Single-crystal-X Ray was used to determine the absolute stereochemistry of compound
Experimental
Plant Material
The root barks of Daniellia oliveri Rolfe were collected in October 2021 from Adamawa region of Cameroon. The plant was identified by Mr Eric Ngansop, a botanist at the Cameroon National Herbarium in Yaoundé by comparison with an existing voucher specimen HNC10672/SFR/CAM.
Chemicals and Instruments
TLC was performed on silica gel 60 F254 (Merck) plates, after what they were sprayed with H2SO4 (10%) reagent, heating and spots visualized on 254-364 nm UV. Silica gel mesh 60- 120 (Merck) was used for Column chromatography while a JASCO 7300 FTIR spectrometer helped in recording IR spectra as KBr pellets. The NMR (1D &2D) hetero- and homonuclear spectra were obtained on a Bruker spectrometer (AMX-500) with TMS (δ(ppm)) as the reference at 500 and 600 MHz; while Hertz (Hz) is the unit of coupling constants (J). HRESIMS (high resolution electrospray ionization mass spectrometry) spectrograms were recorded on a Varian MAT 311A spectrometer X-ray analysis was performed on Bruker D8 Quest Photon 100 diffractometer equipped with an Incoatec IμS high brilliance Cu Kα (1.54178 Å) X-ray.
Extraction and Chromatographic Separation
The powder obtained from air-dried D. oliveri root barks (1.0 kg) was extracted with 4L MeOH at room temperature for 3 days. After filtration, the extract was concentrated using a rotary evaporator to yield 80.5 g of a viscous dark yellow methanolic crude extract (ME). 60.0 g of ME was subjected to a silica gel column (70-230 mesh) and eluted with n-hexane then n-hexane-EtOAc (100:0→0:100) and EtOAc-MeOH (100:0→0:100) in increasing polarity to yield 110 subfractions (150 mL each) combined into five main fractions (D1–D5) based on their TLC profiles. Fraction D1 (19.7 g) was applied to column (70-230 mesh) and eluted with n-hexane then n-hexane-EtOAc (90:10-50:50) to give 03 sub fractions(D1A1 – D1A3). D1A1 (12.1 g) was assessed on silica gel column (70-230 mesh) eluted with n-hexane-EtOAc (60:40) isocratic to afford a crystalline polyalthic acid (
Polyaltic acid (
Evaluation of in Vitro Antimicrobial Activity
Bacteriological assay. The antimicrobial activity of the tested stuffs was carried out on six microbial strains including four bacterial strains (P. aeruginosa 27853, E. coli 5, S. aureus 25923 and Streptococcus sp. 9619) and two strains of yeast (C. albicans14053 and C. tropicalis 018). These strains were obtained from the Research Unit of Microbiology and Antimicrobial Substances of the University of Dschang-Cameroon. The choice of these micro-organisms was motivated by their impact in human health. In fact, Pseudomonas aeruginosa, can cause infections in the blood, lungs (pneumonia), or other parts of the body after surgery, 24 E. coli is a group of bacteria that can cause infections in gut, urinary tract and other parts of the body. 25 Staphylococcus aureus acts as a commensal of the human microbiota, but can also become an opportunistic pathogen, being a common cause of skin infections including abscesses, respiratory infections such as sinusitis, and food poisoning. 26 Streptococcus species are responsible for many cases of pink eye, meningitis, bacterial pneumonia, endocarditis, erysipelas, and necrotizing fasciitis. 27 Candida albicans and C. tropicalis are usually commensal organisms, but can become pathogenic to immunocompromised individuals under a variety of conditions. Species of the genus Candida cause the human infection candidiasis, which results from an overgrowth of the fungus.28,29
Microorganisms were activated for one or two days respectively for bacteria and yeasts growth before being used in the experiment. Bacteria and yeasts inocula were separately prepared from 24 h Muller Hinton agar and 48 h Sabouraud Dextrose Agar cultures respectively for bacteria and yeasts. Colonies from each microorganism were diluted in 0.9% NaCl to have a turbidity matching the 0.5 of Mc Farland standard turbidity scale equivalent to about 1.5 × 108 CFU/mL. The microorganism suspensions were diluted to match the absorbance of 0.1 for bacteria and 0.09 for yeast at 600 nm (Jenway 6105 UV/Vis spectrophotometer, 50 Hz/60 Hz) equivalent to about 2.5 × 105 spores/mL for yeast and 106 CFU/mL for bacteria. 30 The broth microdilution method,31,32 was employed to evaluate the MIC as well as the fungicidal concentration (MFC) of the tested stuffs utilizing 96 well microplates.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241282853 - Supplemental material for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid
Supplemental material, sj-docx-1-npx-10.1177_1934578X241282853 for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid by Larissa M. Magnibou, Dabole Bernard, Peron B. Leutcha, Guy S.S. Njateng, Larissa Y. Chimi, Maurice F. Tagatsing, Sammer Yousuf, Atia-tul-Wahab, Muhammad I. Choudhary and Emmanuel Talla in Natural Product Communications
Supplemental Material
sj-docx-2-npx-10.1177_1934578X241282853 - Supplemental material for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid
Supplemental material, sj-docx-2-npx-10.1177_1934578X241282853 for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid by Larissa M. Magnibou, Dabole Bernard, Peron B. Leutcha, Guy S.S. Njateng, Larissa Y. Chimi, Maurice F. Tagatsing, Sammer Yousuf, Atia-tul-Wahab, Muhammad I. Choudhary and Emmanuel Talla in Natural Product Communications
Supplemental Material
sj-docx-3-npx-10.1177_1934578X241282853 - Supplemental material for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid
Supplemental material, sj-docx-3-npx-10.1177_1934578X241282853 for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid by Larissa M. Magnibou, Dabole Bernard, Peron B. Leutcha, Guy S.S. Njateng, Larissa Y. Chimi, Maurice F. Tagatsing, Sammer Yousuf, Atia-tul-Wahab, Muhammad I. Choudhary and Emmanuel Talla in Natural Product Communications
Supplemental Material
sj-docx-4-npx-10.1177_1934578X241282853 - Supplemental material for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid
Supplemental material, sj-docx-4-npx-10.1177_1934578X241282853 for Phytochemical Constituents, Antimicrobial Activity of Daniellia oliveri, and X-ray Crystal Structure of Polyalthic Acid by Larissa M. Magnibou, Dabole Bernard, Peron B. Leutcha, Guy S.S. Njateng, Larissa Y. Chimi, Maurice F. Tagatsing, Sammer Yousuf, Atia-tul-Wahab, Muhammad I. Choudhary and Emmanuel Talla in Natural Product Communications
Footnotes
Acknowledgments
Authors are grateful to TWAS- ICCBS for the fellowship award.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article since our study does involve neither animals nor humans.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Third World Academy of Science (TWAS)- International Center for Chemical and Biological Sciences (ICCBS) fellowship (N0: 3240311212). The recipient was Dr Larissa M. Magnibou.
Statement of Human and Animal Rights
This information is not relevant.
Statement of Informed Consent
This information is not relevant.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
