Abstract
Fungal resistance to the limited existing antifungal agents has become a considerable public health problem in the past few decades. However, natural products with multiple bioactivities are important sources for developing new therapeutic agents against fungi. Consequently, there is an urgent need to grasp the antifungal mechanism of natural products. In this review, the primary antifungal mechanisms of natural products derived from plants, including targeting the cell wall, cell membrane, mitochondria, biofilm and other mechanisms, are elaborated. This article critically describes the compounds with high antifungal activity, revealing opportunities for future research such as clinical trials or utilization as environmental disinfectants.
Introduction
Fungi are a group of eukaryotic microorganisms with complete cell walls, nuclei, and organelles. It is evaluated that there exist more than 500 species of human pathogenic fungi, 1 and new species of pathogenic fungi have been continuously being reported, with several of them considered as emerging pathogens to human. 2 To date, widespread fungal infections are a worldwide public health problem. Fungi can cause not only superficial infections but also invasive infections, especially in immune-deficient populations. According to an estimated data, more than 1.6 million people are die of fungal diseases, and about a billion people have suffered by fungal infections each year. 3
Currently, azoles, polyenes, echinocandins, and pyrimidine analogs are the four main classes of antifungal agents used in patients with fungal infections. 4 Azoles, including fluconazole, itraconazole, voriconazole, and others, can inhibit lanosterol 14-α-demethylase and thus block ergosterol biosynthesis, resulting in the accumulation of toxic sterol intermediates that inhibit fungal growth. 5 Polyenes (amphotericin B) that bind to ergosterol can change the permeability of the cell membrane, causing cell death. 6 Echinocandins, including caspofungin, anidulafungin, and micafungin, target the fungal cell wall enzyme β-(1,3)-D-glucan synthase (GS), leading to impaired cell wall integrity. 7 Pyrimidine analogs, known as 5-flucytosine, work by impeding DNA and protein synthesis. 8 Fortunately, new antifungal compounds like echinocandin rezafungin as well as the enfumafungin-derivative ibrexafungerp have recently obtained approval from the Food and Drug Administration (FDA), and the inhibitor of glycosylphosphatidylinositol synthesis, namely fosmanogepix is currently under clinical study.9,10
Over the past few decades, the wide use of antifungals has overwhelmingly resulted in the development of drug resistance in many Candida, Aspergillus, Cryptococcus, and other fungal species. A study of 350 clinical Candida auris isolates collected from 10 hospitals in India showed that 90% of them exhibited resistance to fluconazole. 11 Furthermore, an epidemiological analysis of 41 C. auris isolates from Pakistan, India, South Africa, and Venezuela showed that 41% of isolates were resistant to two classes of antifungal agents, and approximately 5% of isolates were resistant to three classes of agents. 12 Similarly, a study in a U.S. hospital found that the resistance rate to echinocandin in Candida glabrata increased from 4.9% to 12.3% over a 10-year period. 13 Interestingly, two voriconazole-resistant strains (TR46/Y121F/T289A mutation) and one itraconazole-resistant strain (TR34/L98H/S297 T/F495I mutation) were isolated from 144 soil Aspergillus fumigatus samples collected from China, suggesting a strong relationship between agricultural triazoles use and antifungal drug resistance. 14
Currently, several antifungal drug resistance mechanisms have been identified, including drug target alterations, overexpression of multidrug transporters, modulation of stress responses, genomic modifications, and intrinsic antifungal resistance. 15 Among those mechanisms, drug target alterations and overexpression of multidrug transporters occurred most frequently. As an example of drug target alterations, antifungal agents such as azoles invariably exhibited lower drug-binding affinity to the drug target Erg11 as amino acid substitutions occurred in the Erg11 open reading frame. 16 In fungi, overexpression of multidrug transporters, the ATP-binding cassette (ABC) superfamily and the major facilitator superfamily (MFS), is another major mechanism that governs antifungal drug resistance. 17
An extremely limited number of antifungal agents and toxic side effects lead to high mortality rates in patients with invasive fungal infections. Therefore, the discovery or chemical synthesis of new antifungal agents is an urgent need. Natural products (NPs) extracted from traditional medicinal plants or fungi have great potential as antifungals. It is a fact that many antimicrobials in clinical use today were derived from NPs. 18 Medicinal plants, especially Chinese traditional herbals, have demonstrated their potential value for fungal inhibition over the millenniums. In the past several decades, large quantities of NPs with application value as antifungals have been discovered. 9 This review describes the possible antifungal mechanisms of NPs derived from plants.
NPs Targeting the Cell Wall
Since human cells lack a cell wall, this fungi-specific structure is a promising target for antifungal agent development with selective effects and low toxicity. 19 The fungal cell wall includes two layers: the inner layer which is composed of β-(1, 3)-D-glucan, β-(1, 6)-D-glucan, and chitin; and the outer layer, which mainly includes glycosylated proteins. 20 During fungal infection, the cell wall, which acts as the primary structure in contact with the host cell membrane, is essential for fungal pathogenicity.21–23 Presently, the commercial antifungal agents in clinical use that target the cell wall GS are echinocandins and include caspofungin, micafungin, and anidulafungin. Interestingly, because the cell wall composition varies between different fungal species, Candida and Aspergillus spp. are susceptible to echinocandins, but Zygomycetes, C. neoformans, and Fusarium species are less susceptible. 24
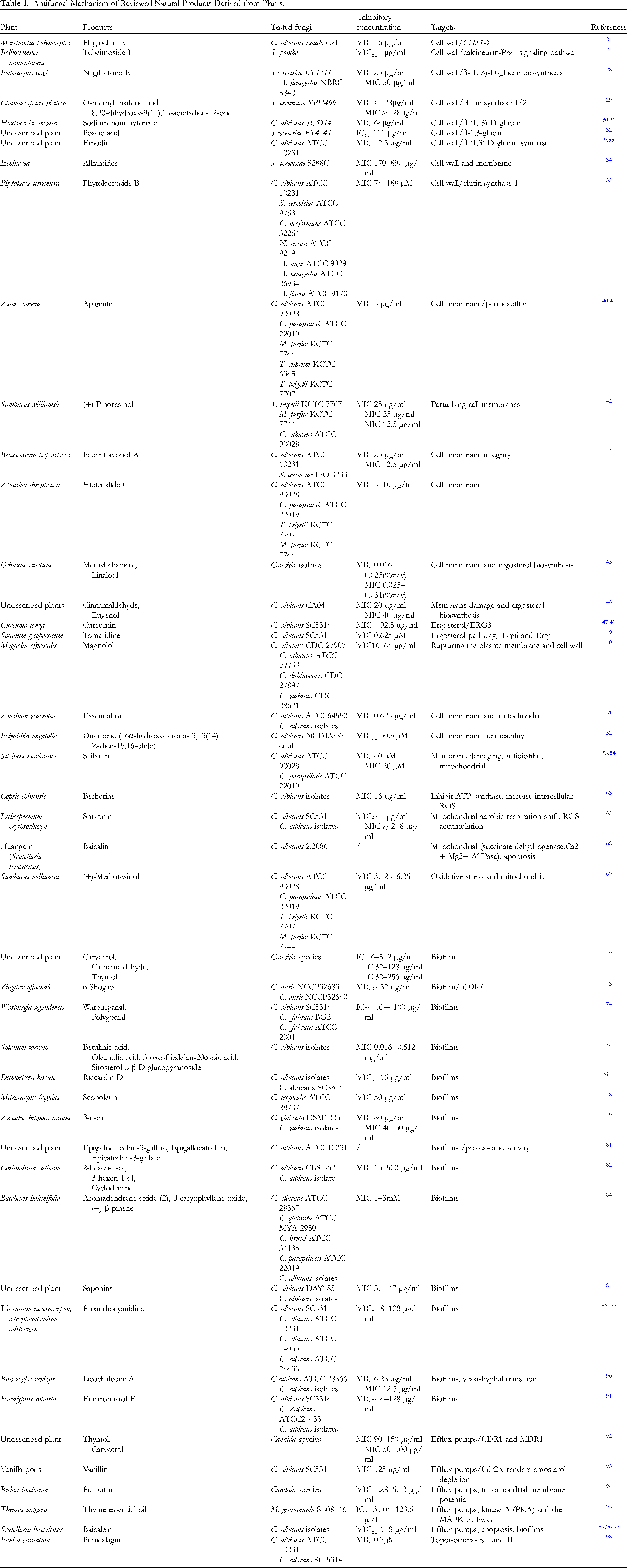
Plagiochin E, a phenolic macrocyclic bis(bibenzyl) isolated from the Chinese liverwort Marchantia polymorpha L., has demonstrated antifungal activity against C. albicans in vitro.25,26 Transmission electron microscope (TEM) observation under plagiochin E treatment showed that the structure of the C. albicans cell wall was severely damaged. Further transcriptional analysis revealed that chitin synthetase gene CHS1 was significantly downregulated, while CHS2 and CHS3 were upregulated upon plagiochin E treatment, suggesting that plagiochin E disrupted the cell wall integrity, possibly by targeting the expression of chitin synthetase genes. 25
Tubeimoside I, a natural compound extracted from the Chinese medicinal herb Bolbostemma paniculatum, was discovered to actively suppress Schizosaccharomyces pombe and clinical isolates of pathogenic fungi by targeting the cell wall. 27 This study further reveals that the treatment of tubeimoside I impacts a portion of genes related to cell wall organization or biogenesis, including stimulating the calcineurin/Prz1 signaling pathway. 27
Nagilactone E is a norditerpene dilactone isolated from the root bark of Podocarpus nagi. In the model organism Saccharomyces cerevisiae, nagilactone E was able to inhibit β-(1, 3)-D-glucan biosynthesis, which caused cell wall fragility and thus resulted in antifungal activity. 28 Additional investigations also confirmed the ability of nagilactone E to produce morphological changes in the pathogenic fungus A. fumigatus. 28
O-methyl pisiferic acid and 8,20-dihydroxy-9(11),13-abietadien-12-one, which was isolated from the leaves of Chamaecyparis pisifera, were also shown to be potent chitin synthase inhibitors of S. cerevisiae. 29 These compounds showed significant inhibition to chitin synthase 1/2 but no inhibitory activity to chitin synthase 3. 29
Sodium houttuyfonate, a derivative of houttuynin that was extracted from Houttuynia cordata Thunb, was demonstrated to induce β-glucan exposure and thus induce intestinal macrophages, eliminating C. albicans through recognition of Dectin-1 and TLR2/4. 30 Further investigation showed that sodium houttuyfonate can physically interact with β-(1, 3)-D-glucan and remodel the C. albicans cell wall. 31 In contrast, a hydrolase test illustrated that β-(1, 3)-D-glucan inhibited sodium houttuyfonate from entering into fungal cells by non-specific absorption with β-(1, 3)-D-glucan. 31 Similar to sodium houttuyfonate, poacic acid, a compound found in lignocellulosic hydrolysates of grasses, directly bound to β-(1, 3)-D-glucan, disrupting cell wall integrity. 32
Emodin (6-methyl-13,8-trihydroxyanthraquinone), a natural anthraquinone derivative found in numerous plants, reduced β-(1,3)-D-glucan synthase activity and increased cell wall damage.9,33
Currently, most reported plant-derived compounds mainly target chitin synthase and glucan synthase; however, alkamides isolated from Echinacea were found to disrupt the fungal cell wall and membrane complex, which represents a potential target for fungal pathogen inhibition. 34 In contrast to chitin or glucan synthase activity inhibition, phytolaccoside B, an antifungal monodesmoside triterpenoid glycoside isolated from Phytolaccaceae, notably enhanced chitin synthase 1 activity and concomitantly increased chitin content, which subsequently altered the fungal cell wall morphology. 35
NPs Targeting the Cell Membrane
In addition to inhibiting the fungal cell wall, targeting the cell membrane is another successful antifungal strategy. Cell membranes of fungi are composed of diverse lipids, including glycerophospholipids, sphingolipids, and sterols. 36 Cell membranes contain lipids that play various roles in biochemical processes: lipids regulate membrane fluidity and the activity of membrane proteins; lipids form a barrier between the inside and outside of the cell or organelle, which provides a place for biochemical reactions; lipids act as secondary messengers; and lipids are a form of energy. 37
In contrast to cholesterol, which functions as the major sterol in animal cells, the fungal cell membrane primarily contains ergosterol. 38 In fungi, ergosterol determines the fluidity, permeability, and activity of membrane-associated transporters or other proteins of the cell membranes, the deficiency of which causes various defects and thus limits cellular growth or adaptation to stress. 39 Based on this fact, the agents used for antifungals in clinical settings (such as amphotericin B and azoles) mainly target ergosterol biosynthesis. However, long-term studies have shown that amphotericin B administered intravenously often induced nephrotoxicity or side effects. 8 Therefore, isolating antifungal agents from NPs that target the fungal cell membrane is a promising approach.
Apigenin, a flavonoid compound found in the chamomile plant, has been reported to possess antifungal activity.40,41 One study found that apigenin altered the cell membrane potential, induced membrane dysfunction, and increased cell permeability, which subsequently led to cell leakage of intracellular components. 41 (+)-Pinoresinol, a biphenolic compound isolated from the traditional medicine herb Sambucus williamsii, revealed significant antifungal activities by perturbing cell membranes in C. albicans. 42 Papyriflavonol A, a prenylated flavonoid isolated from Broussonetia papyriferra, revealed its activity against C. albicans and S. cerevisiae by disrupting cell membrane integrity. 43
Hibicuslide C derived from Abutilon theophrasti acts against fungi through the formation of pores within the plasma membrane with radii between 2.3 nm and 3.3 nm. 44 Ocimum sanctum, which contains two main constituents, methyl chavicol and linalool, showed inhibitory activity against clinical and laboratory isolates of Candida and even azole-resistant strains. 45 Investigation of the antifungal mechanism suggested that the above two main constituents action against fungi was mostly caused by extensive lesions to the plasma membrane and ergosterol biosynthesis inhibition. 45 Similarly, phenyl aldehyde and propanoid treatment resulted in membrane damage and ergosterol biosynthesis inhibition in C. albicans. 46 Phenyl aldehyde and propanoid also can bind with ergosterol. 46
Curcumin showed significant effects against fungi, with a possible antifungal mechanism in which curcumin downregulated ERG3 (Δ5,6 desaturase), leading to decreased ergosterol production, which resulted in the accumulation of biosynthetic precursors of ergosterol and thus led to cell death.47,48 Tomatidine, which originates from Solanum lycopersicum, exhibits strong antifungal activity against Candida species without cytotoxicity to mammalian cells. Transcriptional analysis revealed that the key enzyme of ergosterol pathway, including Erg6 (C-24 sterol methyltransferase) and Erg4 (C-24 sterol reductase), are targeted by tomatidine. An study of tomatidine in S. cerevisiae showed that 2 nonsynonymous mutations in ERG6 (amino acids D249G and G132D) confer resistance to tomatidine, further validating that ERG6 is the target of tomatidine. 49
A few compounds not only target the cell membrane but also attack the fungal cell wall or increase cellular reactive oxygen species (ROS) production. It was demonstrated that magnolol, an antifungal polyphenol from Magnolia officinalis, acted by rupturing the cell wall and plasma membrane, causing cell wall swelling and intracellular contents to release. 50 Dill seed essential oil (DSEO) was shown to have anti-Candida activity and targeted the cytoplasmic membrane and mitochondria. 51 In one study, upon DSEO treatment, lesions were formed in the plasma membrane of C. albicans and thus increased penetrability. Interestingly, DSEO significantly reduced the ergosterol content. Furthermore, DSEO treatment also increased the mitochondrial membrane potential, causing ROS accumulation, which is an important mechanism for inhibiting fungi. 51 A diterpene (16α-hydroxycleroda-3,13(14)Z-dien-15,16-olide) isolated from Polyalthia longifolia var. Pendula revealed antifungal activities against C. albicans, C. neoformans, and Neurospora crassa. 52 The antifungal mechanisms of this diterpene were due to compromised cell membrane permeability, increased intracellular ROS, and probably disruption of the cell wall. 52 Silibinin derived from Silybum marianum exerts a membrane-damaging and antibiofilm effect in C. albicans. 53 Another study showed that silibinin triggered yeast apoptosis related to mitochondrial Ca2+ influx. 54
NPs Targeting the Mitochondria
Mitochondria are the energy factories of almost all fungal cells and produce energy through oxidative phosphorylation. Metabolic intermediates produced by mitochondria are important sources of amino acid and lipid biosynthesis, which are indispensable for fungal survival and growth. 55 Studies of mitochondria have demonstrated that they have multiple functions in fungal growth, pathogenesis, antifungal susceptibility, yeast-to-hypha transition, and host immune responses; consequently, the fungal mitochondrion is a promising drug target for therapy. 56
Studies in Candida, Aspergillus, and other fungi have demonstrated that mitochondrial dysfunction altered the susceptibility to azoles, likely by activating transcription factors that regulate efflux pumps, such as ScPdr3 (S. cerevisiae) and CgPdr1 (C. glabrata).57,58 In addition to azoles, mitochondrial mutants and mitochondrial dysfunction also altered the susceptibility to the echinocandins, which inhibit β-(1, 3)-D-glucan synthase, and polyene drugs that disrupt membrane integrity. 58 It was revealed that mitochondria are sensitive to caspofungin mainly because it caused mitochondrial dysfunction that disturbed lipid homeostasis. 59 Deletion of CHO1 (phosphatidylserine synthase) and of PSD1 and PSD2 (phosphatidylserine decarboxylases) resulted in mitochondrial dysfunction and sensitivity to caspofungin. 60 In contrast, the respiratory-deficient mutant or disruption of cytochrome biosynthesis significantly increased tolerance to amphotericin B.61,62
Berberine is an alkaloid isolated from Coptis chinensis. Functional investigation revealed that berberine treatment alone was able to decrease intracellular ATP production, inhibit ATP-synthase, and increase intracellular ROS in fluconazole-resistant strains. 63 Treatment with berberine and fluconazole caused the mitochondrial membrane potential to increase significantly, and the synergistic activities of ATP production, ATP-synthase inhibition, and reactive oxygen species accumulation became more pronounced. 63
Shikonin is a red pigment isolated from the plant Lithospermum erythrorhizon. 64 A study found that shikonin exhibited antifungal activity against almost all of the tested C. albicans isolates, with a mechanism of ROS accumulation and a mitochondrial aerobic respiration shift. 65 In addition to endogenous ROS augmentation, nitric oxide (NO) accumulation induced by shikonin also played an important role against C. albicans. 64
Baicalin is an active ingredient isolated from Huangqin, a traditional Chinese herb, which has important antibacterial and antifungal activities.66,67 Upon baicalin treatment, the activities of succinate dehydrogenase and Ca2+-Mg2+-ATPase in C. albicans mitochondria were significantly inhibited. 68
(+)-Medioresinol, is a furofuran-type lignan derived from Sambucus williamsii. A study demonstrated that (+)-medioresinol treatment arrested the cell cycle and increased intracellular ROS, which induced oxidative stress and mitochondria dysfunction, and subsequently resulted in apoptosis. 69
NPs Targeting Biofilms
During invasive infections, fungal cells are capable of adhering to the skin or mucous surfaces to form biofilms. Biofilms are rich in polysaccharides and difficult to eradicate, which confers biofilms with drug resistance and immune defense functions. Although current agents are generally able to inhibit biofilm formation, there are only a few drugs available, and most of them have certain toxic effects.70,71 Consequently, the original active compounds derived from plants are of interest, especially traditional Chinese herbs with antibiofilm effects.
A study of carvacrol, cinnamaldehyde, and thymol against C. planktonic showed that cinnamaldehyde effectively inhibited biofilm adhesion, while carvacrol and thymol significantly reduced both mature biofilm biomass and metabolic activity. 72 6-Shogaol derived from ginger Zingiber officinale showed antibiofilm activity by blocking biofilm formation and eradicating the preformed biofilms in C. auris. 73 Interestingly, this study also showed that 6-shogaol downregulated the expression of the efflux pump-related CDR1 gene of C. auris.
Warburganal and polygodial isolated from Warburgia ugandensis showed significant inhibition against biofilms of C. albicans, and in combination treatment with the isolated compounds and azoles, the effective concentration inhibited biofilm formation approximately tenfold decrease compared to individual compounds. 74
Bioactive triterpenoids, including betulinic acid, oleanolic acid, 3-oxo-friedelan-20α-oic acid, and sitosterol-3-β-D-glucopyranoside, isolated from Solanum torvum were demonstrated to harbor considerable antibiofilm formation activity. 75 Riccardin D isolated from Dumortiera hirsute was shown to prevent biofilm formation of C. albicans in a dose-dependent manner both in vitro and in vivo.76,77
Scopoletin, a natural coumarin isolated from Mitracarpus frigidus, acts as a biofilm inhibitor on Candida tropicalis, not only for the growth rate of preexisting biofilms but also the formation of elongated fungal forms. 78 β-Escin derived from the seeds of Aesculus hippocastanum L. also showed the ability to inhibit or destroy biofilm formation, possibly due to the downregulation of some crucial genes including ALS3, CDR1, ERG11. Furthermore, the combination of escin with azoles presented synergistic activity against C. glabrata biofilm.79,80 Epigallocatechin-3-gallate, which is the most abundant ingredient of tea polyphenols, displayed a significant inhibition on either biofilm formation or established biofilms. Epigallocatechin and epicatechin-3-gallate demonstrated similar role in biofilm inhibition. Interestingly, it was found that the tea polyphenols also exhibits an inhibitory effect on the proteasome activity of C. albicans. 81
The crude oil of Coriandrum sativum has the capacity to impact the biofilm formation in C. albicans, with chemical analysis identifying the most active compounds as 2-hexen-1-ol, 3-hexen-1-ol and cyclodecane. 82 Similar activity of Coriandrum sativum essential oil extract, principally consisting of alcohols and aldehydes, in regard to Candida spp. biofilm formation were described by D H X Barbosa et al. 83 A study of the essential oil of Baccharis halimifolia with major component of aromadendrene oxide-(2), β-caryophyllene oxide, and (±)-β-pinene also displayed significant activities against the maturation phase and preformed 24 h-biofilm of C. albicans. 84
Saponins, both the arginoside type and those of the barrigenol family, showed excellent antibiofilm formation activity at concentrations below the minimum inhibitory concentration, which was comparable to the level of caspofungin. 85 Proanthocyanidins obtained from Vaccinium macrocarpon and Stryphnodendron adstringens,86–88 all displayed antibiofilm activity against the tested fungi. Some flavonoids, including baicalein present in Scutellaria baicalensis 89 and licochalcone A present in licorice, 90 have demonstrated significant inhibition of the formation of C. albicans biofilms.
In a recent study, Liu RH et al discovered that when treated with Eucarobustol E, a natural formyl-phloroglucinol meroterpenoids from Eucalyptus robusta, there was a significant inhibition of biofilm, along with a reduction in hyphal growth and the transcription of cell surface-related genes. 91
Other Mechanisms
In fungi, the activation of efflux pumps is an important mechanism of multidrug resistance. Consequently, screening for NPs with efflux pump inhibition activity presents an attractive strategy to combat multidrug fungal pathogen. Thymol and carvacrol, the principal chemical components of thyme oil, demonstrated significant synergistic activity against C. albicans when combined with fluconazole. This was due to the inhibition of efflux pumps and the decreased transcripition of CDR1 and MDR1 genes that encode efflux pumps. 92 Similar to thymol and carvacrol, vanillin, a major constituent of vanilla pods, specifically suppresses the expression of C. albicans Cdr2p and synergizes with fluconazole and amphotericin B. 93 Purpurin, a natural red anthraquinone pigment from Rubia tinctorum L., also exhibited energy-dependent efflux pumps inhibition in the Candida isolates. 94 In mycosphaerella graminicola, thyme essential oil isolated from Thymus vulgaris exhibited significant antifungal activity by regulating efflux pumps, kinase A (PKA) and the MAPK pathway. 95 Baicalein, a compound that has been established to possess the ability of antibiofilm formation, 89 not only exhibited the inhibition of efflux pumps, but also accelerated apoptosis accompanied by an increase of reactive oxygen species when treated in combination with baicalein and amphotericin.96,97 In addition, punicalagin isolated from Punica granatum L. showed a strong inhibitory activity against C. albicans topoisomerases I and II. 98 Other antifungal mechanisms of NPs derived from plants have also been reported. 99
Conclusion
This article reviewed the antifungal mechanisms of NPs derived from plants with defined ingredients (Table 1). Although the crude water or alcohol extracts of many plants have demonstrated with good antifungal activity, the active constituent are unknown and thus not included in this article. NPs derived from plants are an invaluable resource for antifungal drug development, especially with the current state of microorganism drug resistance. Through the millenniums, traditional Chinese medicine has demonstrated its great importance to humans on fungal inhibition. However, the detailed molecular targets of NPs are still limited. It would also be interesting to validate the in vitro and vivo antifungal activities of NPs either alone or in combination with other antifungal agents by conducting clinical trials,100,101 as well as to investigate their potential use as environmental disinfectants. 102 In the future, structural modifications or directly using NPs as precursors for new antifungal drug development is of great interest.
Antifungal Mechanism of Reviewed Natural Products Derived from Plants.
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by National Natural Science Foundation of China (grant 32000107), Hunan Provincial Natural Science Foundation of China (2024JJ7510) to N. B. Long; National College Students’ innovation and entrepreneurship training program (202210547017) to F. Li. The funders had no role in study design, data collection and interpretation, or the decision to submit the work for publication.
