Abstract
Background/Purpose
Some health-beneficial foods, including adzuki beans (Vigna angularis), can show antihyperglycemic effects. Adzuki bean-derived polyphenols, such as (+)-catechin 7-O-β-
Methods
Six-week-old male ICR mice were orally administered ABP (100 or 250 mg/kg), C7G or E7G (15 or 40 mg/kg), and sucrose (2 g/kg). Blood samples were taken from the lateral tail vein to measure the blood glucose levels at various times over 0–120 min. Cultured caco-2 cells, an intestinal model, were treated with ABP, C7G, or E7G. The glucose concentration in the medium was measured to examine the activity of α-glucosidase.
Results
Administration of adzuki polyphenols decreased blood glucose levels, especially E7G administration significantly showed antihyperglycemic effects in vivo. In Caco-2 cells, α-glucosidase activity was significantly decreased by ABP, C7G, or E7G addition in a dose-dependent manner without cytotoxicity.
Conclusion
These results suggest that C7G and E7G in adzuki bean extract inhibited sucrose-degrading enzymes to elicit antihyperglycemic effects in vivo, which supports the results of previous studies using adzuki beans, and may contribute to health promotion.
Introduction
Type 2 diabetes (T2D) is a systemic disorder that is characterized by abnormally increased blood glucose levels, which can lead to serious symptoms, including nephropathy and cardiovascular disease.1,2 Hyperglycemia is a representative characteristic of T2D and is responsible for many diabetic complications. An effective approach for preventing hyperglycemia is the inhibition of the important carbohydrate degrading enzymes, α-amylase and α-glucosidase.3,4 α-Amylase breaks down carbohydrates into disaccharides in the digestive system, and α-glucosidase degrades disaccharides into glucose, which then enters the bloodstream. Both these enzymes are essential for homeostasis in animals; however, their activation can cause acute rises in blood glucose levels after meals. Therefore, inhibiting α-amylase and α-glucosidase can suppress carbohydrate digestion, delay glucose uptake, and reduce blood glucose levels.
Some medicines and food-derived compounds can retard the digestion of carbohydrates and suppress the rate of glucose uptake in the small intestine to prevent acute rises in blood glucose. Acarbose strongly inhibits the activity of α-amylase and α-glucosidase, which results in reduced glucose generation. 5 Antihyperglycemic medicines can be effective for the treatment of diabetes; however, some of these medicines can cause serious side effects in patients, including liver and intestinal damage. 6 In terms of safety and ease of use, the inhibition of α-amylase and α-glucosidase by natural compounds in the daily diet may be effective in the prevention of T2D. 7 From this view point, in recent years, polyphenols have attracted attention because of their protective role against T2D. 8 For instance, some polyphenols play a significant role in carbohydrate metabolism by inhibiting key enzymes responsible for the digestion, 9 exhibit increased insulin secretion in pancreatic β-cells, 10 and enhance glucose uptake in the muscles and adipocytes. 11
Several natural inhibitors found in edible plants, including in adzuki beans (AB, Vigna angularis), have the potential for preventing hyperglycemia. AB have been traditionally used as a health-promoting food for a long time, are easy to prepare, and can be consumed in our daily meals. AB are known to contain abundant polyphenols, and most of these polyphenol compounds have been identified.12,13 The administration of a hot water extract of AB significantly suppressed increases in blood glucose levels as indicated by an oral glucose tolerance test.14,15 The AB extract suppressed blood glucose levels after sucrose, maltose, or starch administration in vivo. In addition, AB extracts have been reported to have several health-beneficial effects, for instance oxidative stress-lowering and cholesterol-lowering activities.16,17 In the recent trend for healthy lifestyles and health-beneficial foods, AB have attracted attention and demand, not only in Asia, but worldwide. However, the specific bioactive components in AB that are responsible for the beneficial effects are still under investigation.
Recently, we isolated and identified (+)-catechin 7-O-β-
The elucidation and analysis of the activity of adzuki polyphenols would contribute to the development, and promote the use, of adzuki materials and products with respect to their health beneficial effects. Therefore, in the present study, we investigated the antihyperglycemic effects of adzuki bean extract containing polyphenols (ABP) in vivo. To assess the effects on postprandial blood glucose levels in mammalian body, we measured blood glucose levels after the administration of sugar with ABP, and purified C7G or E7G in mice. In addition, we evaluated the cytotoxicity and α-glucosidase inhibiting activity of these polyphenols using Caco-2 cells as an intestinal model, 19 which are derived from human colon cancer and known to show intestinal features, such as disaccharidase activity. 20
Results
HPLC Analysis of ABP and Polyphenols
We prepared adzuki bean extract containing polyphenols (ABP; total polyphenol content: 70.0%, average yield: 155.8 mg from 100 g of adzuki beans), and each polyphenol was determined as follows: peak I, C7G; peak II, procyanidin; peak III, catechin; peak IV, E7G; peak V, tryptophan; peak VI, epicatechin; and peak VII, rutin (Figure 1A). For determination, ion peaks were observed in the mass spectra at [M-H]−= 451 and 451 for peaks I and IV by LC-MS analysis, respectively. We previously reported and isolated the peaks I and IV as C7G (MW: 452) and E7G (MW: 452), respectively. 18 Ion peaks were observed in the mass spectra at [M-H]−= 577 for peak II, and MW of peak II was detected as 578. Thus, we estimated peak II as procyanidin based on previous reports.21,22 Peaks III, V, VI, and VII were analyzed and estimated by LC-MS analysis, and these four peaks were determined by commercially available standards using HPLC. Among them, C7G was the most abundant polyphenols in ABP (16.5%), on the other hand, E7G was only a small amount that exists in ABP (3.8%). However, E7G is known to be converted from C7G by heating during cooking, this study focused on C7G and E7G (Figure 1B).

HPLC chromatograms of ABP. (A) ABP was analyzed by HPLC using a C18 column at 210 nm (1.0 mg/mL of ABP, 10 μL injected). Peak I: catechin 7-O-β-
Oral Glucose Tolerance Test in Mice After Administration of ABP
The effect of ABP on postprandial blood glucose levels was examined in sucrose-loaded mice. When 250 mg/kg of ABP was orally administered simultaneously with sucrose to the mice, the postprandial blood glucose levels were significantly decreased 15 and 120 min after the administration, compared with those of the control (CNT; saline) group (P < 0.05, Figure 2A). The area under the curve (AUC) value for the total increase in the blood glucose level was significantly decreased after administration of 250 mg/kg of ABP (P < 0.05, Figure 2B). These results indicated that the ABP had antihyperglycemic effects against sucrose loading in vivo, and we predicted C7G and E7G as potential antihyperglycemic compounds in AB.
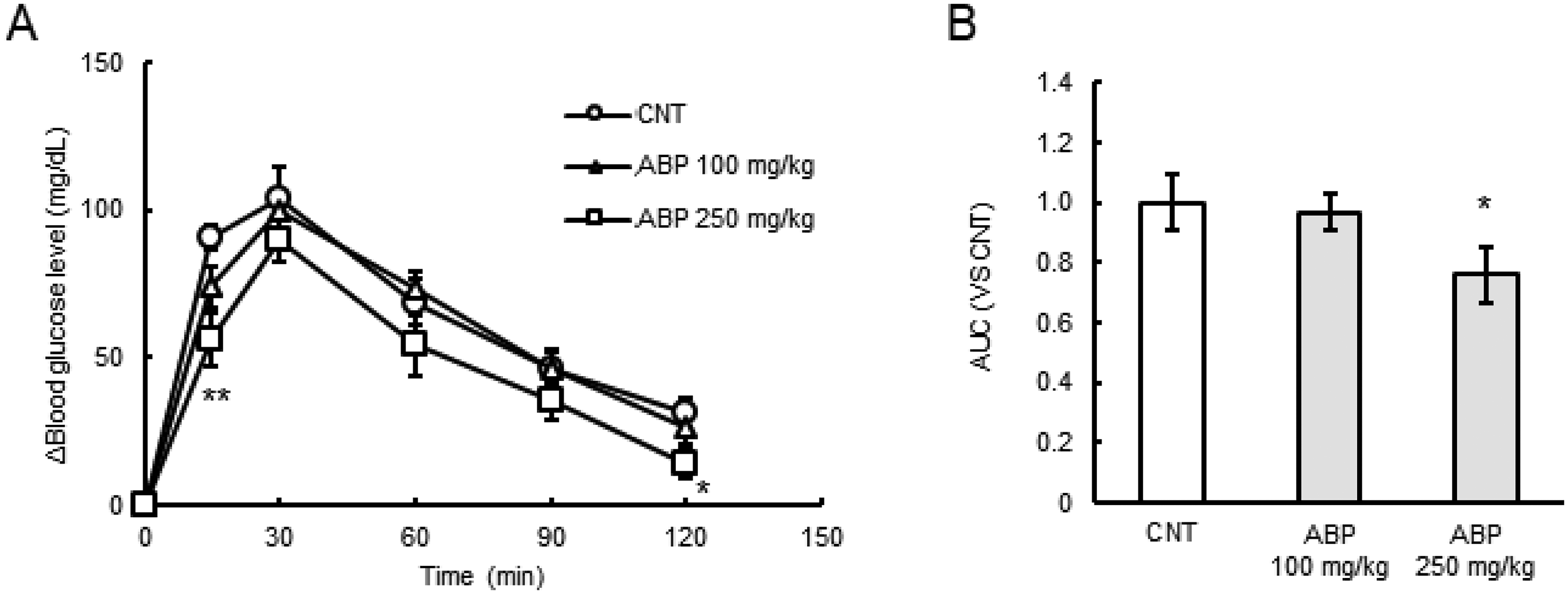
Effects of ABP on the postprandial blood glucose levels in sucrose-loaded mice. (A) ABP and sucrose (2 g/kg) were administered (n = 7-8 per group). Blood samples were taken at the indicated times (0, 15, 30, 60, 90, and 120 min). ○, CNT (saline); △, 100 mg/kg ABP; □, 250 mg/kg ABP. (B) Area under the curve (AUC) of blood glucose levels after sucrose loading. All values represent the mean ± SE. *P < 0.05, **P < 0.01 compared with the CNT group by Dunnett's test.
Oral Glucose Tolerance Test in Sucrose-Loaded Mice
Antihyperglycemic effects of C7G and E7G on postprandial blood glucose levels were examined in sucrose-loaded mice. Experimental conditions are the same as that of ABP, and the applied doses of C7G correspond to the percentage in ABP (considered as 15%). When 15 or 40 mg/kg of C7G was orally administered simultaneously with sucrose to the mice, the postprandial blood glucose level was shown a decreasing trend, however, there were no significant differences compared with CNT group (Figure 3A). The AUC value for the total increase in the blood glucose level was shown a decreasing trend by 15 or 40 mg/kg of C7G administration (Figure 3B). When 40 mg/kg of E7G was orally administered, the blood glucose level was significantly lower 30 min after administration compared with that of the CNT group (P < 0.05, Figure 3C). The AUC value was also significantly decreased after administration of 40 mg/kg of E7G, compared with the CNT group (P < 0.05, Figure 3D). These results showed that E7G had antihyperglycemic effects in sucrose-loaded mice, and the effect of C7G was partially in sucrose-loaded mice. Despite their low concentrations, oral administration of purified polyphenol (40 mg/kg) showed an inhibitory effect similar to that of oral administration of high concentrations of ABP (250 mg/kg).

Effects of C7G and E7G on the postprandial blood glucose levels in sucrose-loaded mice. (A) C7G and sucrose (2 g/kg) were administered (n = 10 per group). Blood samples were taken at the indicated times (0, 15, 30, 60, 90, and 120 min). ○, CNT (saline); △, 15 mg/kg C7G; □, 40 mg/kg C7G. (B) AUC values of blood glucose levels (C7G). (C) E7G and sucrose (2 g/kg) were administered (n = 10-12 per group). ○, CNT; △, 15 mg/kg E7G; □, 40 mg/kg E7G. (D) AUC values of the blood glucose levels (E7G). All values represent the mean ± SE. *P < 0.05, **P < 0.01 compared with the CNT group by Dunnett's test.
Oral Glucose Tolerance Test in Maltose- or Soluble Starch-Loaded Mice
To investigate the further potential of antihyperglycemic effects of C7G and E7G, maltose-loaded mice were gavaged with C7G or E7G. When 15 or 40 mg/kg of C7G was orally administered, the postprandial blood glucose levels were significantly lower 30 min after administration compared with those of the CNT group (P < 0.05, Supplementary Fig. 1A), and the AUC values were shown a little decreasing trend (Supplementary Fig. 1B). When E7G was orally administered, there were no significant differences in the levels of postprandial blood glucose or the AUC values, compared with those of the CNT group (Supplementary Fig. 1C, D).
Next, the effects of C7G and E7G on postprandial blood glucose levels were examined in starch-loaded mice. When 15 mg/kg of C7G was orally administered, the blood glucose levels were significantly lower 60 min after administration, and when 40 mg/kg of C7G was orally administered, the blood glucose levels were significantly lower 30 and 90 min after administration, compared with those of the CNT group (P < 0.05, Supplementary Fig. 2A). The AUC values were significantly decreased after administration of 15 or 40 mg/kg of C7G (P < 0.05, Supplementary Fig. 2B). In contrast, when E7G was orally administered, there were no significant differences in the levels of postprandial blood glucose or the AUC values, compared with those of the CNT group (Supplementary Fig. 2C, D). These results showed that C7G had potential antihyperglycemic effects in starch-loaded mice.
Inhibition of α-Glucosidase Activity in Caco-2 Cells
To investigate the decreasing mechanism of blood glucose levels, we evaluated whether ABP, C7G or E7G showed inhibitory activity toward α-glucosidase in an intestinal model. ABP, C7G, or E7G treatment resulted in a dose-dependent decrease in the amount of glucose generated from sucrose degradation in Caco-2 cells (Figure 4A, B). Compared with vehicle control group (100%), amount of glucose was significantly decreased to 20.5% ± 2.3% when 500 µg/mL of ABP was added (P < 0.05). Treatment of 5, 50, or 500 µg/mL of C7G decreased amount of glucose to 55.1% ± 4.6%, 38.9% ± 2.0%, 16.5% ± 1.8%, respectively (P < 0.05). Treatment of 5, 50, or 500 µg/mL of E7G decreased amount of glucose to 50.0% ± 10.1%, 35.9% ± 5.8%, 16.7% ± 2.7%, respectively (P < 0.05). Decreased glucose generation because of the inhibition of α-glucosidase by acarbose, an α-glucosidase inhibitor, was observed in a dose-dependent manner (Figure 4C). Both C7G and E7G significantly reduced α-glucosidase activity in Caco-2 cells, and significant differences or marked differences between C7G and E7G were not observed. Only the high concentration of ABP (500 µg/mL) significantly suppressed α-glucosidase activity, while low concentration of purified C7G or E7G (5, 50, or 500 µg/mL) also significantly suppressed α-glucosidase activity.
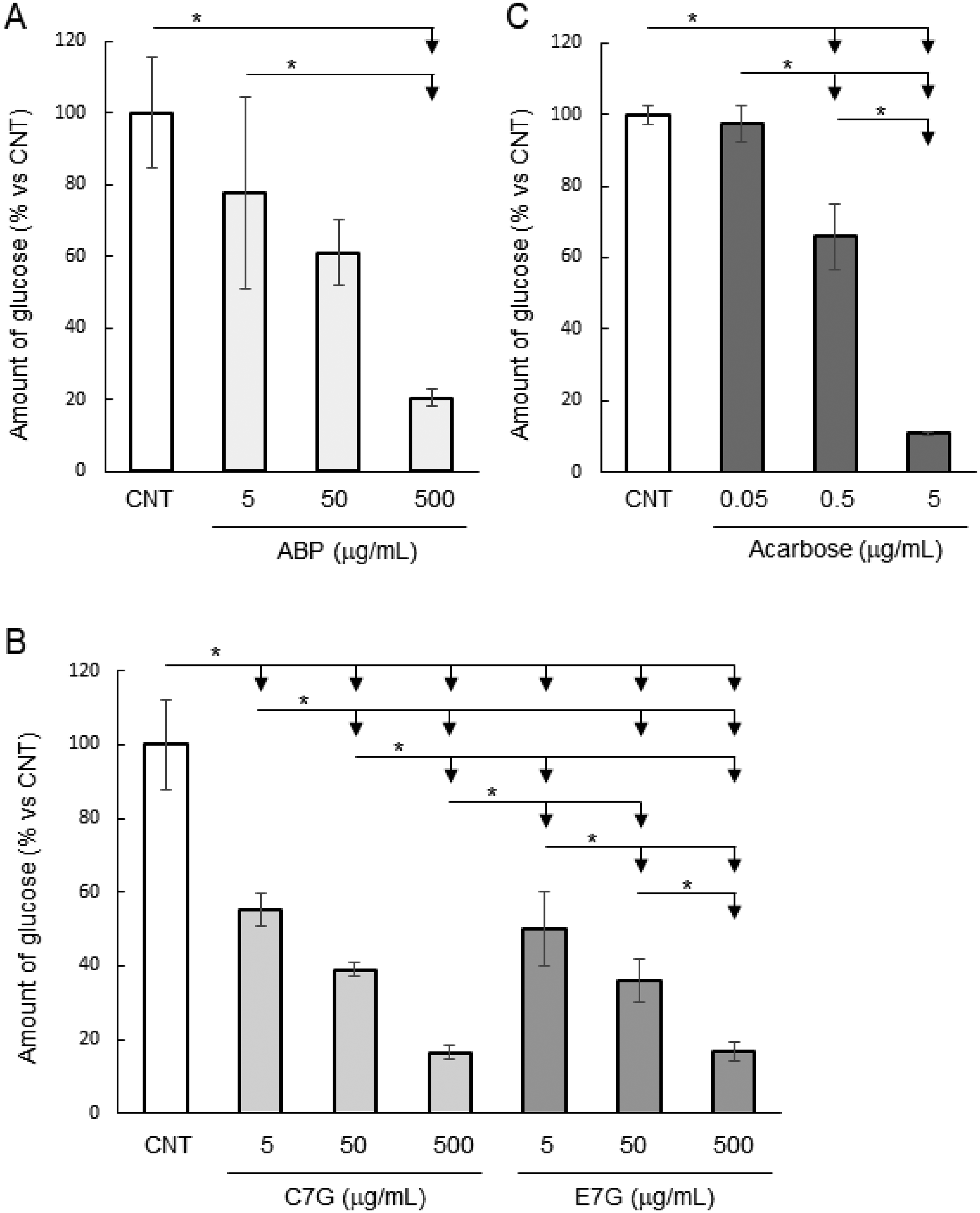
Inhibition of sucrose degradation by adzuki polyphenols in Caco-2 cells. (A, B) The inhibitory activity against sucrose degradation of ABP, C7G, and E7G was measured at final concentrations of 5–500 µg/mL on day 19 in Caco-2 cells. (C) Acarbose was also used at final concentrations of 0.5–5 µg/mL as a positive control. The generated glucose was measured by the glucose oxidase method using Amplex™ Red reagent. The data are representative of three independent experiments conducted in triplicate and are expressed as mean ± SD. *P < 0.05 by Tukey–Kramer test.
In addition, to evaluate the cytotoxic effects of ABP, C7G, and E7G under the experimental conditions, we investigated cell viability. As a result, neither compound showed inhibitory activity, thus, there was no cytotoxicity toward Caco-2 cells at the indicated concentrations (Supplementary Fig. 3A, B).
Stability of C7G and E7G Under Gastrointestinal pH Conditions
Finally, we investigated the stability of C7G and E7G under gastrointestinal conditions. C7G and E7G significantly suppressed blood glucose levels at 30 min after oral administration; thus, it was likely that C7G and E7G were stable under gastric conditions to reach and act in the intestine. Both C7G and E7G were stable (C7G: pH 3, 101.0% ± 0.5%; pH 5, 101.2% ± 1.4%; and pH 7, 100.0% ± 0.9% and E7G: pH 3, 99.3% ± 1.4%; pH 5, 100.6% ± 0.2%; and pH 7, 100.8% ± 3.1%; compared with before incubation) under all the tested gastrointestinal pH conditions (Figure 5A–C). Catechin and epicatechin, which are unglycosylated forms of C7G and E7G, were also stable (catechin: pH 3, 101.7% ± 1.8%; pH 5, 100.4% ± 0.6%; and pH 7, 100.0% ± 0.6% and epicatechin: pH 3, 100.7% ± 0.3%; pH 5. 100.9% ± 2.0%; and pH 7, 100.8% ± 0.7%) under the same conditions.

Stability of C7G, E7G, catechin, and epicatechin at indicated pH conditions. The percentage remaining after incubation in sodium phosphate buffer at pH 3 (A), pH 5 (B), or pH 7 (C) at 37 °C for 30 min was measured by HPLC. The degree of degradation of C7G, E7G, catechin, and epicatechin at gastrointestinal pH was compared with that of the unincubated compound. The data are shown as the mean ± SD from triplicate determinations, and there were no significant differences between the groups by Tukey–Kramer test.
Discussion
Functional foods and food components that can reduce α-amylase or α-glucosidase activity have attracted attention for their potential to prevent hyperglycemia. 23 In the present study, we investigated the antihyperglycemic effects of ABP and the major polyphenols in ABP, C7G and E7G, on blood glucose levels in sucrose-loaded mice. Administration of ABP or E7G significantly suppressed blood glucose levels in sucrose-loaded mice (Figure 2A, B and 3C, D). In addition, administration of C7G, but not E7G, significantly suppressed blood glucose levels in soluble starch-loaded mice (Supplementary Fig. 2). The above results showed that C7G had antihyperglycemic effects in starch-loaded mice, in contrast, E7G had antihyperglycemic effects in sucrose-loaded mice. Some compounds have been shown to have different activities against maltose and sucrose degradation 24 similar to how E7G showed inhibitory activity against sucrose degradation but not against maltose or starch degradation. Then, to investigate the antihyperglycemic mechanism of adzuki polyphenols, we evaluated α-glucosidase inhibitory activity using Caco-2 cells. In accordance with previous studies, Caco-2 cells were grown for 19 days, which exhibits characteristics as an intestinal model compatible with the purpose of this study.25,26 ABP, C7G, and E7G significantly reduced glucose generation by suppressing α-glucosidase activity in a dose-dependent manner (Figure 4A, B). These results indicated that both C7G and E7G have potential inhibiting activity toward α-glucosidase and reduced the degradation of disaccharides, which can indirectly lead to a suppressed intake of glucose.
In a previous study, the postprandial blood glucose levels of normal mice were suppressed by oral administration of a hot water extract of AB. 14 The AB extract significantly suppressed blood glucose levels when sucrose or soluble starch was loaded. It was presumed that these effects were associated with the polyphenols in the AB, which affected the postprandial glucose levels in vivo. The suppression of the postprandial blood glucose levels by AB polyphenols was thought to be caused by the inhibition of intestinal enzymes, although there was no effect on glucose absorption.14,15 Some flavonoids, including catechins, are known to inhibit α-amylase by direct binding to the enzyme mediated by hydrogen bonding and hydrophobic interactions.27,28 Thus, the inhibitory activity can be explained by the high affinity between the flavonoids and the enzyme. Considering our results of HPLC analysis, oral glucose tolerance test, and Caco-2 cell experiments, C7G is one of the most important antihyperglycemic compounds in AB, and its heat conversion epimer E7G is also a potential antihyperglycemic compound.
C7G and E7G showed similar antihyperglycemic effects with slightly different activities. In addition to environmental effects on the reaction, the differences in the observed inhibitory activity between C7G and E7G might be derived from their structural differences. As shown in Figure 1B, the structures of C7G and E7G are different, similar to the case with catechin and epicatechin, which may cause differences in the inhibitory activity. Some catechin-related compounds have been reported to inhibit α-glucosidase activity through direct binding to the enzyme, and the structure was critical for the binding efficiency. 29 Thus, the differences in the structures of C7G and E7G may be important for enzyme inhibition and may contribute to the observed differences in their activities. Given Caco-2 experiments in Figure 4, C7G and E7G could inhibit α-glucosidase activity through direct binding to the enzymes in the intestine.
Both C7G and E7G, as well as catechin and epicatechin, were shown to be stable under gastrointestinal pH conditions (Figure 5). In a previous study, several catechins, including catechin and C7G, were ≥ 90% stable under various pH conditions for at least 1 h. 30 We obtained similar results to this study, and showed that C7G and E7G can reach the intestine after administration and suppress blood glucose levels under gastrointestinal conditions. Thus, C7G and E7G were stable under gastric pH conditions, and these results support the hypothesis that suppressing α-glucosidase activity in the small intestine.
Several of the agents that are used for the treatment of hyperglycemia can cause a variety of serious side effects; therefore, the development of new therapeutics with low treatment burdens is desirable. Toward this purpose, mild α-amylase and α-glucosidase inhibitors that could be consumed in a daily diet without toxicity would be useful. 31 Our results indicated that C7G and E7G are the main bioactive components in AB, and might act as α-glucosidase inhibitors in vivo. There's a possibility that these adzuki polyphenols show suppressive effects on postprandial blood glucose levels in human body.
However, there are still several limitations to this research. Firstly, only one intestinal cell line was used, and this study does not cover late-stage process of digestion, such as glucose uptake or regulation of insulin secretion. In vitro models such as Human Pancreatic Normal Cell Culture- T25 Plated Cells, a more relevant model for T2D and glucose metabolism, can be used in our future studies to further verify the effects and deeply elucidate the mechanisms of adzuki polyphenols on regulating glucose metabolism. Secondly, experimental results in mice do not necessarily lead to the same results in humans. Therefore, we recommend clinical studies to further validate the health-beneficial effects of adzuki polyphenols. Lastly, we performed single oral administration in this study, thus, the continuous administration of these polyphenols needs to be investigated, focusing on the systemic effects in vivo. For example, previous reports have shown that C7G suppressed inflammation in rats 32 and suppressed antigen-stimulated degranulation in leukemic cells. 33 Various health-beneficial effects have been reported after the long-term administration of adzuki extracts in vivo, and it is possible that other tissues, such as liver and muscle tissues, and not only the intestine, may be affected. Further studies are needed, and are in progress in our laboratory, to elucidate the mechanisms of action and long-term systemic effects of polyphenols from AB extracts.
Conclusions
The present study investigated the effects of the bioactive components in AB extract, focusing on the in vivo antihyperglycemic activity. Administration of ABP and E7G resulted in significant decreases in blood glucose levels in a sucrose-loaded mouse oral glucose tolerance test. C7G administration showed a decreasing trend in blood glucose levels in sucrose-loaded mice. Furthermore, ABP, C7G, and E7G significantly inhibited glucose generation from sucrose degradation by α-glucosidase in Caco-2 cells with no cytotoxicity. Overall, these results suggested that both C7G and E7G are among the key antihyperglycemic polyphenols in AB, and can decrease postprandial blood glucose levels by acting as inhibitors of sucrose-degrading enzymes in the small intestine. Our studies support the consumption of AB, and the development of adzuki polyphenols, for the prevention of acute rises in blood glucose levels.
Materials and Methods
Preparation and HPLC Analysis of ABP, C7G, and E7G
AB harvested in Hokkaido, Japan were used in this study. To obtain a polyphenol-rich AB extract, a 40% ethanol fraction was prepared according to a previous study. 14 In brief, 100 g of AB were boiled for 15 min in 500 mL of hot water, then centrifuged at 10000 × g for 10 min to obtain the supernatant. Then, the supernatant was evaporated and subjected to HP-20 open column chromatography (35ID × 300 mm; Mitsubishi Chemical Corporation, Tokyo, Japan), and the 40% ethanol fraction was collected as adzuki bean extract containing polyphenols (ABP).
High-performance liquid chromatography (HPLC; SHIMADZU, Kyoto, Japan) with a pump (LC-20AD; SHIMADZU) and an ultraviolet detector operating at 210 nm (SPD-20A; SHIMADZU) were used for analysis, and peaks were recorded by CDS-Lite software (LAsoft, Chiba, Japan). The compounds were analyzed as follows: water and methanol with 0.1% trifluoroacetic acid were used as the mobile phase (0 to 15 min, 100% water; 15 to 45 min, 100% water to 100% methanol; 45 to 60 min, 100% methanol) at a flow rate of 1 mL/min. C7G and E7G were isolated and analyzed as previously described. 18 Purity of isolated C7G and E7G was confirmed each time to be a single peak by HPLC.
LC-MS Analysis
LC-MS analysis was performed using a LCMS-2010EV (SHIMADZU, Kyoto, Japan) with a pump (LC-20AD; SHIMADZU) and a column oven (CPO-20A; SHIMADZU) in the negative ion mode. The instrument was equipped with an electrospray ionisation (ESI) source and a photodiode array detector (SPD-20A; SHIMADZU), and controlled by LCMS Solution software (SHIMADZU). Compounds were separated using an ODS column (TSKgel ODS-100 V 3 µm, 2.0ID × 75 mm; Tosoh, Tokyo, Japan).
Oral Glucose Tolerance Test in Mice
The method for the oral glucose tolerance testing was designed based on previous studies.34–36 Five-week-old male mice (ICR, Japan SLC, Hamamatsu, Japan) were kept in a pathogen-free environment and under a light schedule of 07:00–19:00 at 22 ± 1 °C. The mice were allowed free access to water and a laboratory diet (DC-8, CLEA Japan, Inc., Tokyo, Japan) for 1 week to accustom the mice to their surroundings. Then, mice at six weeks of age were randomized into groups for oral glucose tolerance tests (ABP: n = 7-8 per group; C7G or E7G: n = 9-12 per group). After overnight fasting (16 h), the mice weighing between 24 and 34 g were used, and orally gavaged with ABP (100 or 250 mg/kg body weight), C7G (15 or 40 mg/kg body weight), or E7G (15 or 40 mg/kg body weight) and sucrose, maltose, or soluble starch (2 g/kg body weight). Oral doses were set based on a previous study 14 and our results of HPLC analysis (approx. 15% of ABP). Blood samples were taken from the lateral tail vein at various times over 0–120 min. The blood glucose levels were measured by the glucose oxidase method using a Labogluco glucose analyzer (Funakoshi, Tokyo, Japan), and the values for the area under the curve (AUC) for blood glucose were calculated. ABP, C7G, and E7G were dissolved in saline (0.9% NaCl), and pure saline was used as a control. All mice were maintained at the Institute of Laboratory Animals, Mie University. All experimental procedures were performed according to the Mie University guidelines for laboratory animals, approved by the Institutional Animal Care and Use Committee of Mie University, and complied with the governmental guidelines (The Japanese Association of Laboratory Animal Facilities of National University Corporations).
Cell Culture and Reagents
Caco-2 cells (RIKEN BRC CELL BANK, Tsukuba, Japan) were grown in Dulbecco's modified Eagle's medium (Nissui, Tokyo, Japan) supplemented with 10% fetal bovine serum (Hyclone, Logan, UT, USA) at 37 °C under 5% CO2. Caco-2 cells were maintained and used in each assay within 20 passages, and cultured until 80% confluence before harvest. Trypsin/EDTA (0.25%; Nacalai Tesque) was used to harvest the cells.
The reagents sucrose (Nacalai Tesque), maltose (Nacalai Tesque), soluble starch (Wako, Osaka, Japan), glucose (Wako), and acarbose (Wako) were used in this study. Commercially available (+)-catechin (Toronto Research Chemicals, Toronto, Canada), (+)-epicatechin (Nagara Science, Gifu, Japan), rutin (Nacalai Tesque), and tryptophan (Nacalai Tesque) were used as analytical standards.
α-Glucosidase Activity Assay
The α-glucosidase activity in Caco-2 cells was measured as previously described with slight modifications.23,37,38 Caco-2 cells were seeded in a 96 well plate (1.0 × 104 cells/well) and were grown for 19 days. Then, the culture medium was replaced with a reaction mixture consisting of ABP, C7G, or E7G at final concentrations of 5–500 µg/mL with a 28 mM sucrose solution in PBS for α-glucosidase inhibitory activity determination. The assay plate was then incubated at 37 °C in 5% CO2 for 2 h. After incubation, the extracellular glucose concentration was measured by the glucose oxidase method using Amplex™ Red reagent (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacture's protocol. To confirm the applicability of this assay, acarbose was used as a control.
Cell Viability Assay
The same as α-glucosidase activity assay, Caco-2 cells were grown for 19 days and treated with ABP, C7G or E7G at final concentrations of 5–500 µg/mL for 2 h. Then, 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-tetrazolium bromide (MTT; Nacalai Tesque) solution (5 mg/mL) was added to each well, and the plate was incubated for a further 4 h at 37 °C. The absorbance of the dissolved crystals was quantitated as a percentage compared with the vehicle control using a microplate reader (CHROMATE4300; Practical Japan, Chiba, Japan) at 600 nm.
Stability Under Gastrointestinal pH Conditions
The stability of the polyphenols under gastric pH conditions was evaluated according to a previously described method. 39 The gastrointestinal transit times and pH conditions (gastric pH: pH 3; intestinal pH: pH 5 and 7) were adjusted from previous studies.40–42 Samples were diluted in phosphate buffer (100 mM NaHPO4, 100 mM Na2HPO4) at pH 3, 5, or 7 and incubated for 30 min at 37 °C. After incubation, aliquots were withdrawn and analyzed to calculate the amounts of the polyphenols by HPLC using a C18 column (Nacalai Tesque) and an ultraviolet detector operating at 210 nm.
Statistical Analysis
Data are expressed as the mean ± standard derivation (SD) of the results from three wells and were confirmed by three independent experiments. In the in vivo experiments, data are expressed as the mean ± standard error (SE) of the results from at least seven mice per group. The statistical significance of differences was evaluated by one-way analysis of variance followed by Dunnett's test or Tukey–Kramer test (BellCurve for Excel; Social Survey Research Information, Tokyo, Japan). Values of P < 0.05 were considered to indicate statistical significance.
Supplemental Material
sj-tif-2-npx-10.1177_1934578X241271734 - Supplemental material for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells
Supplemental material, sj-tif-2-npx-10.1177_1934578X241271734 for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells by Kenji Kuriya, Masahide Osumi, Mana Ishihara, Masahiro Nishio, Hirotaka Katsuzaki, Junpei Sono, Masahiro Nakamura and Hayato Umekawa in Natural Product Communications
Supplemental Material
sj-tif-3-npx-10.1177_1934578X241271734 - Supplemental material for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells
Supplemental material, sj-tif-3-npx-10.1177_1934578X241271734 for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells by Kenji Kuriya, Masahide Osumi, Mana Ishihara, Masahiro Nishio, Hirotaka Katsuzaki, Junpei Sono, Masahiro Nakamura and Hayato Umekawa in Natural Product Communications
Supplemental Material
sj-tif-4-npx-10.1177_1934578X241271734 - Supplemental material for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells
Supplemental material, sj-tif-4-npx-10.1177_1934578X241271734 for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells by Kenji Kuriya, Masahide Osumi, Mana Ishihara, Masahiro Nishio, Hirotaka Katsuzaki, Junpei Sono, Masahiro Nakamura and Hayato Umekawa in Natural Product Communications
Supplemental Material
sj-doc-5-npx-10.1177_1934578X241271734 - Supplemental material for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells
Supplemental material, sj-doc-5-npx-10.1177_1934578X241271734 for Potential Antihyperglycemic Effects of Adzuki (Vigna angularis) Polyphenols on Mice and Caco-2 Cells by Kenji Kuriya, Masahide Osumi, Mana Ishihara, Masahiro Nishio, Hirotaka Katsuzaki, Junpei Sono, Masahiro Nakamura and Hayato Umekawa in Natural Product Communications
Footnotes
Acknowledgments
We thank S. Kabeyasawa, S. Morioka, and M. Fuse (Mie University) for technical contributions to this study.
Data Availability
The data underlying this article will be shared on reasonable request to the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This study was approved by the Institutional Animal Care and Use Committee of Mie University (approval number: 2021-15, date: August 6, 2021).
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the Mie University guidelines for laboratory animals, approved by the Institutional Animal Care and Use Committee of Mie University, and complied with the governmental guidelines (The Japanese Association of Laboratory Animal Facilities of National University Corporations).
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
