Abstract
Objectives
The present study focused on exploring the chemical composition of essential oil and corresponding hydrolate obtained by steam distillation of industrial hemp (Cannabis sativa L.) cultivar “Helena” (low THC content).
Methods
Chemical characterization of industrial hemp essential oil and hydrolate was performed by gas chromatographic and gas chromatographic-mass spectrometric analysis, while biological activities included antimicrobial and antioxidant tests. Antimicrobial activity was determined by measuring diameters of the inhibition zone by using a disc-diffusion method with nine microbial strains from ATCC culture. Moreover, minimal inhibitory concentration (MIC) as well as time-kill kinetic studies, antiadhesion, and antibiofilm formation potential were also evaluated. Antioxidant activity was evaluated through three different antioxidant assays: 2,2-diphenyl-1-picrylhydrazyl (DPPH●), 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS●+), and reducing power (RP).
Results
The gas chromatography mass spectrometry analysis showed that the main compound in the essential oil was trans-caryophyllene (37.4%), followed by caryophyllene oxide (12.4%) and α-humulene (11.0%), while in the hydrolate it was 1,8-cineole (11.5%). Results showed that industrial hemp essential oil and hydrolate exhibited no antimicrobial activity against gram-negative bacteria, yeasts, and fungi, while gram-positive bacteria were sensitive. Therefore, in the following step, MIC was determined by microdilution method. The lowest MIC for essential oil (12.5%) was obtained for Enterococcus faecalis and Staphylococcus aureus, while this value doubled for Listeria monocytogenes. Conversely, the MIC for hydrolate was 100% for all gram-positive bacteria. Antioxidant activity showed that industrial hemp essential oil and hydrolate have potential as natural sources of antioxidants.
Conclusion
This research confirmed the previously proven antimicrobial and antioxidant activities of industrial hemp essential oil. The novelty lies in the antimicrobial and antioxidant activity of hydrolate, which is practically waste, but has great potential to be a useful by-product.
Introduction
Cannabis sativa L. is considered a single species of the isolated primary gene pool within the Cannabis genus (family Cannabaceae). 1 Controversial taxonomy within the genus is based on morphological characteristics, chemotype, ecotype, or cultivation purpose.2,3 Industrial hemp is traditionally grown as a fiber crop, although hurds are also used in many industries. 4 Hemp seed, which is a source of protein and oil, finds its valorization on the food market. 5 The industrial hemp flower represents a relatively unexplored area for the potential exploration, with different biomolecules, primarily cannabinoids, a group of C21 terpenophenolic compounds, exhibiting a broad spectrum of activity. 6 Moreover, terpenes, volatile compounds from essential oil, are responsible for the aromatic qualities, and thus for its market value.7,8,9
Reports indicate that industrial hemp essential oil possesses antimicrobial,10,11,12,13 antioxidant,12,13 as well as anti-inflammatory properties.14,15 Additionally, antiparasitic and antileishmanial activities have also been noted,16,17 along with hepatoprotective and antidiabetic properties.18,19 This indicates that industrial hemp essential oil can be considered a potential source of natural bioactive compounds suitable for use as functional food, supplements, or natural preservatives in food and cosmetic products. Furthermore, it may have applications in topical therapy for inflammation and wound healing.20,21
Nowadays, zero-waste managements is gaining importance due to the environmental preservation, the potential for additional incomes and the increased demand for healthy food supplements in everyday nutrition. 22 Previous studies have explored the extraction of phenolic compounds and terpenes from industrial hemp by-products, such as waste material remaining after seed herb, leaf and flower extraction.23,24 However, this is the first study to focus on the hydrolate. During steam distillation, the essential oil (the final primary product) is collected in the Florentine flask, along with the hydrolate (which is considered waste). 25 However, a minor part of the volatile compounds from the plant material passes into the hydrolate (usually less than 0.1%), mainly the highly polar, water-soluble fraction such as monoterpene alcohols, aldehydes, and ketones, as well as sesquiterpene alcohols, which are responsible for the specific scent of the hydrolate. 26
According to previous studies, hydrolates possess antimicrobial, antioxidant, and anti-inflammatory effects, and therefore have potential applications in the food industry (for flavoring, and preservation), in the production of functional (non-alcoholic) beverages, for household products (for disinfection and aromatherapy), and in cosmetic formulations (replacement for water phase, natural fragrance, stabilizers, and preservatives).26,27,28,29 Additionally, hydrolates have potential applications in organic agriculture as biopesticides.30,31
The purpose of this research was to characterize volatile organic compounds from essential oil and hydrolate obtained by steam distillation of dry plant material of industrial hemp cultivar “Helena” (low THC content). In vitro antimicrobial and antioxidant activity testing was also conducted to find additional value for these products.
Materials and Methods
Plant Material
In 2020, the industrial hemp crop cv “Helena” (Voucher No 2-0024, BUNS Herbarium) was cultivated at the Institute for Field and Vegetable Crops Novi Sad (IFVCNS)—Vegetable and Alternative Crops Department in Bački Petrovac, Serbia (N502138 E395689). Sowing was conducted on April 15, and throughout the growing season, agrotechnological measures recommended for commercial production were implemented. On September 14, 2020, fresh biomass samples were collected from randomly selected plants. Industrial hemp raw material samples were generated by cutting the top 45 cm of monoecious plants. Samples were then hung in a shady area (tobacco dryer) until a constant air-dried weight was achieved and then further processed.
Essential oil and Hydrolate Preparation and Chemical Characterization
The dried plant material was processed by steam distillation (in a small distillation unit at IFVCNS) for 4 h (the process is previously described in detail).32,33 The obtained essential oil, which floated on the hydrolate in the Florentine flask, was decanted, placed in a separation funnel overnight, and sodium sulphate was added the next day. After filtering through filter paper, the essential oil was stored in dark glass bottles until further analysis. The remaining hydrolate in the Florentine flask was collected through filter paper into sterile plastic bottles for further experiments. However, before the chemical characterization of the volatile compounds from the hydrolate, the procedure of simultaneous distillation and extraction with dichloromethane via a Likens–Nickerson apparatus was applied, in order to obtain a secondary essential oil.
The volatile profiles was characterized using a gas chromatographic (GC) analysis system using a HP 5890 GC equipped with a flame ionization detector, HP 5973 mass selective detector, and a capillary column HP-5MS (the terms and conditions are described in detail).32,33 The identification of isolated components was based on their linear RI in relation to homologous series of n-alkanes (C8–C32), and compared with the Wiley and NIST databases. The relative percentage of identified compounds was calculated from the chromatogram peak area.
Antimicrobial Activity
Test Microorganisms
In this study, a total of nine microorganisms were used for different analyses: gram-negative bacteria Escherichia coli ATCC 25922, Pseudomonas aeruginosa ATCC 27853, Salmonella Typhimurium ATCC 13311; gram-positive bacteria Staphylococcus aureus ATCC 25923, Enterococcus faecalis ATCC 19433, Listeria monocytogenes ATCC 19115; yeasts Saccharomyces cerevisiae ATCC 9763, Candida albicans ATCC 10231, and fungi Aspergillus brasiliensis ATCC 16404. These microorganisms are stocked at 80 °C with glycerol as cryoprotectant and are part of the culture collection of the Laboratory of Microbiology, Faculty of Technology Novi Sad, Serbia. Prior to microbiological analysis, the bacteria were transferred to Müller-Hinton Agar (HiMedia, Mumbai, India) and incubated at 37 °C for 24 h, while eukaryotes were transferred to Sabouraud Maltose Agar (HiMedia, Mumbai, India) and the following incubation conditions were implemented: S. cerevisiae was grown at 30 °C for 48 h, C. albicans at 37 °C for 24 h and A. brasiliensis was incubated at 25 °C for 5 days.
Antimicrobial Assessment
In order to determine the antimicrobial potential of industrial hemp essential oil and hydrolate, two methods were conducted. As a preliminary test, the disc-diffusion method was chosen, while for the sensitive microorganisms, minimal inhibitory concentration (MIC) was further determined. Both methods were previously described in detail.34,35
Disc-Diffusion Method
In the disc-diffusion method, in addition to the tested samples of industrial hemp essential oil and hydrolate, positive and negative controls were used. Sterile distilled water and 5% DMSO served as the negative control, while tetracycline 30 μg/ml (HiMedia, Mumbai, India) was used for all bacterial strains and actidione 30 μg/ml (Acros Organic, New Jersey, USA) for eukaryotes was chosen as the positive control. Each experiment was performed in triplicate (n = 3).34,35
Microdilution MIC Method
MIC was determined for all sensitive microorganisms using the microdilution method in sterile flat-bottom 96-well microtiter plates. The experiments were performed in triplicate, and MIC was calculated as follows:
Time-Kill Kinetic Study
To monitor changes during the contact of microbial cells and MIC × 1 and MIC × 2 values of essential oil, as well as MIC × 1 value of hydrolate, time-kill kinetic study was conducted as described. 36 The above-mentioned MIC values of essential oil and hydrolate were brought into a contact with L. monocytogenes, E. faecalis, and S. aureus cell suspensions (prepared in nutrient medium up to concentration of approx. 6 logCFU/ml) and the sampling was performed in the following time intervals: 0, 2, 3, 4, 5, 6, 12, 24, and 48 h. The number of cells was determined by spreading an aliquot of 100 μL of appropriate dilution onto Plate Count Agar (HiMedia, Mumbai, India) and counting the grown colonies after the incubation at 37 °C for 24 h. All results were subjected to mathematical modelling and the four-sigmoidal kinetics were determined using the described method. 37
Antiadhesion and Antibiofilm Formation Potential
The antiadhesion and antibiofilm formation potentials of the tested essential oil and hydrolate were determined, and all results from triplicate repetition were presented as percentage reduction in viable count by using the methods described with some modifications. 38 The control experiment involved growth without the addition of industrial hemp essential oil or hydrolate under the same conditions.
For the antiadhesion test, sterile 96-well plates were used as a test surface. In each well, 200 μL of Müller-Hinton Broth (HiMedia, Mumbai, India) was added, along with 10 μL of a freshly prepared suspension of the tested microorganism to give a final inoculum size of approximately 6 log units per well. Tested essential oil and hydrolate were then added to each well, and the plate was incubated at 37 °C for 24 h for all bacteria. Following the incubation, the antiadhesion activity of the tested extracts was determined using the viable counting method.
The antibiofilm formation potency was determined using 96-well plates. An aliquot of 100 μL of previously prepared suspensions of test microorganisms in Müller-Hinton Broth supplemented with 2% w/v sucrose (Sigma-Aldrich, Ottawa, Canada, USA), in the presence or absence of the tested essential oil or hydrolate, was added to the plates. The incubation conditions of plates were the same as for the antiadhesion test. All other steps in this method were performed according to previously described method. 38
Antioxidant Activity
To investigate its antioxidant potential, a prepared and diluted sample of industrial hemp essential oil and its hydrolate were subjected to three different antioxidant assays: 2,2-diphenyl-1-picrylhydrazyl (DPPH●), 2,2′-azinobis-3-ethylbenzothiazoline-6-sulphonic acid (ABTS●+), and reducing power (RP). The prepared industrial hemp essential oil was diluted with an ethanol:acetone solution (50:50) to a concentration of 100 mg/ml, while its by-product, hydrolate, was used directly. The procedures for these tests have been described in detail previously.39,40 The absorbance was measured by spectrophotometry on different wavelengths (515, 414, and 700 nm) and antioxidant capacity was expressed as μmol of Trolox equivalent (TE) per 1 mL of solvent.
Statistical Analysis
All in vitro tests were performed in triplicate, and results are expressed as mean value ± standard deviation (SD). The time-kill kinetics approach was performed using sigmoidal numerical model suitable for biological systems, as described in detail previously. 35 The coefficient of determination (R2) and residual sum of squares (RSS) were calculated to test the adequacy of the developed models. All data were statistically processed using the software package STATISTICA 10.0 (StatSoft Inc., Tulsa, OK, USA).
Results and Discussion
Chemical Composition of Industrial Hemp Essential oil and Hydrolate
There are 43 compounds in the industrial hemp essential oil, with trans-caryophyllene (37.4%) being the main compound, followed by caryophyllene oxide (12.4%) and α-humulene (11.0%). Conversely, in the hydrolate, 1,8-cineole (11.5%) was the predominant compound among a total of 34 compounds (Table 1). As shown in Table 1, sesquiterpenes accounted for 66.7% of the essential oil, while oxygenated monoterpenes constituted the most abundant class of compounds in the hydrolate, with 70.9%.
Chemical Composition of Industrial Hemp Essential Oil (EO) and Hydrolate (HY).

Abbreviations: R.T. (min), retention time; RIexp, retention index determined experimentally by C8–C36 n-alkanes on a HP-5MS non-polar capillary column; tr, trace (less than 0.05%); nd, not detected; *, aliphatic hydrocarbons, aldehydes, alcohols, acids, as well as their esters, and alkyl-aromatic alcohols.
Similar chemical compositions of industrial hemp essential oil have been reported by other authors as well.41,42 Moreover, it is established that the composition of hemp essential oil depends on genotypes/cultivar, climatic conditions, phenological stage, postharvest processing, and extraction method.41,43,44,45,46 However, to the authors’ knowledge this is the first reference to the chemical composition of the secondary essential oil re-extracted from industrial hemp hydrolate. The chemical composition of essential oils from other industrially interesting plants, such as lavender, thyme, oregano, savory, mint, sage, rosemary, rose, citrus, bay laurel, and others, has been extensively studied, along with their biological activity and potential applications. 25 Bearing in mind that only a small amount of hemp essential oil is obtained during the distillation process, usually between 0.085 and 0.262 mL per 100 g dried plant, 47 and that the amount of the obtained hydrolate is limited by the capacity of the distillation system and is usually significantly larger,48,49 the hydrolate can be a very interesting by-product due to its specific fragrance and potential biological activities.
Screening of Antimicrobial Potential
The antimicrobial potential of the industrial hemp essential oil and hydrolate was assessed by measuring the diameters of the inhibition zones using the disc-diffusion method. The results are presented in Figure 1. Analysis of all controls used in this assessment (Tetracycline, actidione, DMSO, and sterile distilled water) is presented in Supplemental Table 1. According to the obtained results, it is evident that the tested essential oil and hydrolate have no antimicrobial activity against gram-negative bacteria and the tested yeasts, and fungi. Moderate activity of the essential oil against tested gram-positive bacteria was observed, as well as the low activity of the tested hydrolate. The highest antimicrobial activity was determined against E. faecalis (16.33 mm in the case of industrial hemp essential oil and 8.0 mm for hydrolate). The antibiotic tetracycline formed a halo zone above 26 mm for all tested bacteria, while the antimycotic actidione formed a halo zone above 21 mm for all tested eukaryotes. Negative controls (sterile distilled water and DMSO) showed no activity against the tested microorganisms.

Antimicrobial potential of industrial hemp cultivar “Helena” essential oil and hydrolate with determined minimal inhibitory concentrations.
Determination of Minimal Inhibitory Concentration
In the following step, the MIC value for sensitive gram-positive bacteria was determined using the microdilution method. As shown in Figure 1, the lowest MIC (12.5%) was obtained for the essential oil in the cases of E. faecalis and S. aureus, while this value doubled for L. monocytogenes. The MIC value of industrial hemp hydrolate was 100% for all gram-positive bacteria, consistent with its low activity previously observed in the disc-diffusion method.
The antimicrobial activity of industrial hemp essential oils is primarily attributed to the presence of various terpenes. In the investigated samples, trans-caryophyllene, caryophyllene oxide, and α-humulene have been identified as significant components. For instance, trans-caryophyllene has been shown to damage the cell membrane integrity of bacteria, leading to leakage of cell contents and eventual cell death. 11 Caryophyllene oxide is noted for its efficacy in inhibiting fungal cell growth and is also suggested to interfere with cell wall synthesis. 11 Moreover, antibacterial and antibiofilm effects of α-humulene have also been noted. 50
Additionally, the concentrations of the essential oils and their components were chosen based on preliminary studies that indicated their MIC effectiveness against the target microorganisms. The MIC for E. faecalis and S. aureus was found to be 12.5% for the essential oil, suggesting a potent antimicrobial effect sufficient to warrant the use of these concentrations in our disc-diffusion assays. This study also notes that the antimicrobial activity of industrial hemp essential oil is more pronounced against gram-positive bacteria, which can be attributed to the differences in cell wall composition between gram-positive and gram-negative bacteria. The outer membrane in gram-negative bacteria often serves as a barrier to many substances, including essential oils. Generally, essential oils are more effective against gram-positive bacteria, likely due to the lack of an outer membrane that is present in gram-negative bacteria, which provides an additional barrier to antimicrobial agents.51,52,53
In summary, the obtained results successfully demonstrated the antimicrobial properties of industrial hemp essential oil samples, particularly highlighting their efficacy against gram-positive bacteria. The predominant compounds, such as trans-caryophyllene, caryophyllene oxide, and α-humulene, might play a critical role in the antimicrobial mechanism, primarily through the disruption of microbial membrane integrity and inhibition of vital cellular processes. Additionally, a synergistic effect of these components is possible. Overall, the findings from this study confirm the potential of industrial hemp essential oil as a natural antimicrobial agent, suitable for applications where reduction of gram-positive bacterial populations is desired.
Time-Kill Kinetics Study
As a supplement to evaluation of antimicrobial activity, a time-kill kinetic study was conducted using MIC × 1 and MIC × 2 of essential oil and MIC × 1 of hydrolate against the tested gram-positive bacteria. Figure 1 illustrates the kinetics of control samples (non-treated bacterial suspensions) and the time-kill kinetics for the treated bacterial suspensions. The aim was to determine the influence of control and MICs values of industrial hemp essential oil on sensitive bacteria. The first step was to obtain the bacterial growth profile without the addition of essential oil or hydrolate. For all three tested bacteria, three dominant growth phases were observed—lag, log, and stationary phase. The lag phase lasted for the first few hours and was followed by the log phase, which ended after 24 h, when the stationary phase began. Upon contact with bacterial cells, the MIC level of hydrolate led to a decrease in the number of bacteria, reaching zero log CFU/ml after 48 h for all sensitive bacteria. A similar trend is observed in the case of MIC level of industrial hemp essential oil. However, the number of bacteria sharply decreased compared to the hydrolate. Conversely, the double MIC value of the essential oil significantly reduced the initial number of cells, and after 12–24 h no bacteria in the suspension could be detected.
Estimation of kinetic models’ parameters (a—lower asymptote, b—upper asymptote, c—inflection point, and d—slope at the inflection point) was successfully completed for each tested combination from Figure 2. The calculated kinetic parameters provide insights into the growth characteristics under each condition. The standard errors provide an indication of the precision of the parameter estimates. The model appears to fit well for the control condition but shows some variability in the treatment conditions, as indicated by the standard errors. This variability might be due to the biological nature of the data or the specific response to treatments.

Time kill kinetics evaluation through four-parameter sigmoidal models.
To evaluate the goodness-of-fit for the four-parameter sigmoidal models to the time-kill kinetics study (Figure 2), coefficients of determination (R2) were calculated (Table 2). These provide a measure of how well the observed bacterial behavior is replicated by the model. Additionally, the RSS was defined for each kinetic experiment in order to see how much of the variance in the dependent variable is not explained by the model. Based on the obtained results, for the control and hydrolate MIC conditions, R2 values were very close to 1, suggesting that the model fits the data very well. The essential oil MIC and essential oil 2 × MIC conditions had slightly lower R2 values, indicating a good but not perfect fit compared to the control and hydrolate MIC conditions. The low RSS values across all conditions also indicate a good fit of the model to the data.
Variability and Discrepancy of the Obtained Kinetic Models.

R2 value of 1 indicates perfect fit and vice versa.
Lower RSS indicates a better fit.
The in vitro antimicrobial activity of industrial hemp essential oil and hydrolate was assessed based on the inhibition zone diameter, MIC and pharmacodynamic potential of biocide effects (Figure 2, Supplemental Table 2). Regarding the obtained results, the antimicrobial activity of the examined essential oil and hydrolate may be qualified as low to moderate only in the case of gram-positive microorganisms. All other tested microorganisms appear to be resistant to essential oil and hydrolate. Generally, gram-positive bacteria were more susceptible to the bioactive substance, which is related to their cell wall structure. Gram-positive bacteria have high membrane permeability due to the presence of peptidoglycans that are linked to teichoic and teichuronic acids by covalent bonds. 37 These findings are consistent with the previously reported research.4,10 Specifically, other researchers have also emphasized the antimicrobial activity of industrial hemp essential oil against gram-positive bacteria, while there is limited information on the activity of industrial hemp hydrolate. The antimicrobial activity of industrial hemp essential oil is strongly influenced by variety, sowing time, and the chemical composition.10,54 Although, the time-kill kinetics has become an inevitable method when testing new substances with antimicrobial potential, it has not been previously used for industrial hemp essential oil or hydrolate. The only available research based on pharmacodynamic focuses on synergism of industrial hemp essential oil with different antibiotics. The results showed a significant decrease of MICs values when the essential oil was added to the antibiotics. 12 This study demonstrated that double MIC concentration of industrial hemp essential oil rapidly reduced the microbial population. This result may represent the first step in a strategy to reduce the intense use of synthetic antibiotics.
In summary, the study observed moderate antimicrobial activity of industrial hemp essential oil against gram-positive bacteria, while the hydrolate exhibited low activity. No significant activity was noted against gram-negative bacteria, yeasts, or fungi. This is consistent with the known differences in membrane permeability between gram-positive and gram-negative bacteria. In addition, the essential oil showed a lower MIC for gram-positive bacteria, notably for E. faecalis and S. aureus. In contrast, the hydrolate exhibited a 100% MIC value for all gram-positive bacteria, reflecting its lower efficacy. To obtain a complete picture of the antimicrobial assessment, the time-kill kinetics study provided insights into the dynamics of bacterial population reduction upon treatment with the essential oil and hydrolate. The essential oil demonstrated a more rapid decrease in bacterial numbers compared to the hydrolate, especially at double MIC values. All obtained kinetic models fit well for the control conditions but showed variability under treatment conditions, which could be attributed to the biological nature of the data or specific treatment responses.
Antiadhesion and Antibiofilm Potential
The ability of industrial hemp essential oil and hydrolate to inhibit the adhesion of cells to surfaces as well as the potential of biofilm formation were assessed at MIC and MIC × 2 levels for essential oil and MIC level for hydrolate (Figures 3 and 4). It was found that the hydrolate had minimal or no influence on the both tested capabilities. Conversely the essential oil was considerably efficient, especially when double MIC value was introduced in cell suspension. Industrial hemp essential oil significantly reduced the potential of initial cell attachment in the case of E. faecalis (about 40%) (Figure 3), as well as biofilm formation (about 60%) (Figure 4). Similar results were also obtained for S. aureus, while the examined potentials were at the lowest level for L. monocytogenes. According to the results presented in Figures 3 and 4, it can be concluded that industrial hemp essential oil was more efficient in antibiofilm formation than in inhibiting cell attachment to surfaces.

Antiadhesion potential of industrial hemp cultivar “Helena” essential oil and hydrolate.
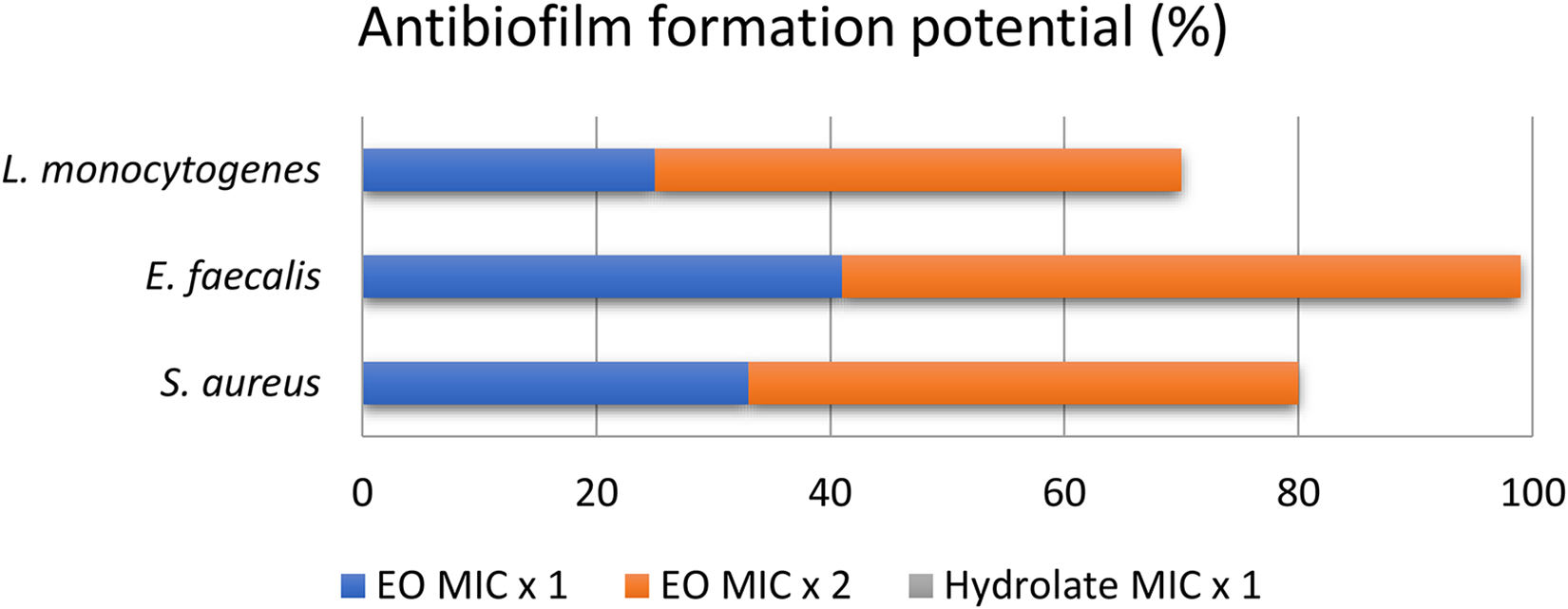
Antibiofilm potential of industrial hemp cultivar “Helena” essential oil and hydrolate.
In addition to antimicrobial resistance, the problem of bacterial attachment to surfaces and biofilm formatting became a global issue, especially in food industries. To date, various physical and chemical methods have been employed to combat bacterial colonization, but these methods are usually expensive, unstable, difficult to perform, and can even contribute to microbial resistance, as well pose a risk to consumer health. 55 From this perspective, researchers are currently making efforts to find natural compounds with antibiofilm and antiadhesion properties. Considering substances of natural origin, most are phytochemicals such as carvacrol, phytol, curcumin, ginkgolic acid, etc. 56 Therefore, essential oils have become valuable sources of bioactive compounds with possible antibiofilm potential. For example, there are studies which proved the high efficiency of cassia, Peru balsam, and red thyme essential oils in controlling biofilm formation of Pseudomonas spp. and S. aureus. 57 Similarly, Mexican oregano and thyme essential oils both have antibacterial and antibiofilm activity on E. coli and S. aureus. 58 In this study, it was demonstrated that industrial hemp essential oil significantly reduces the possibility of initial cell adhesion to surfaces (between 25% and 40%) as well as the potential of forming biofilm (between 25% and 60%) in cases of L. monocytogenes, E. faecalis, and S. aureus. To our knowledge these are pioneer results related to industrial hemp essential oil ability to inhibit cell attachment to surfaces and biofilm formation.
As main highlights, the study assessed the potential of industrial hemp essential oil and hydrolate to prevent bacterial adhesion and biofilm formation. The essential oil was found to be more effective, particularly at double MIC concentrations, in reducing initial cell attachment and biofilm formation, with varying efficacy among different bacteria. The findings align with previous research highlighting the antimicrobial potential of industrial hemp essential oil against gram-positive bacteria. The study indisputably contributes new insights into the antimicrobial action of industrial hemp hydrolate, a lesser-studied substance, and provides pioneering data on its antiadhesion and antibiofilm properties. Furthermore, the study's comprehensive approach, from evaluating antimicrobial activity to understanding the kinetics and mechanisms of action, offers valuable information for further research and potential applications in controlling microbial growth, particularly in the context of increasing antibiotic resistance.
This pioneer study in antiadhesion and antibiofilm potential of industrial hemp can be crucial for further combating bacterial infections, particularly in the era of increasing antimicrobial resistance. Industrial hemp essential oil's effectiveness in preventing bacterial adhesion and biofilm formation presented in this study might be linked to its specific chemical composition (Table 1). The identified compounds are well-known molecules that interfere with bacterial communication systems (quorum sensing), which are vital for biofilm formation. This mechanism is distinct from traditional antibiotics, making them potentially valuable in addressing antibiotic-resistant strains. These findings contribute to the promotion of the use of natural products in controlling biofilms, which is a major challenge in healthcare settings. Given the increasing prevalence of antibiotic-resistant infections, the potential of such substances in supplementing or enhancing existing treatments is highly relevant.
In Vitro Assessment of Antioxidant Activity
The results of in vitro antioxidant activity of industrial hemp essential oil and its hydrolate are presented in Table 3. These results showed positive but notably different antioxidant activity for both essential oil and hydrolate, across all three tests performed. Both samples exhibited the highest scavenging activity against ABTS•+ (189.26 and 40.61 μmol TE/mL), followed by DPPH• (157.62 and 33.82 μmolTE/mL) and RP (127.45 and 26.57 μmol TE/mL).
Antioxidant Activity (IC50) of Industrial Hemp Essential Oil and Hydrolate (μM TEAC/ml).

Results are expressed as mean ± standard deviation (n = 3).
Abbreviations: DPPH
Literature reviews have shown that DPPH● and ABTS●+ antioxidant assay results fluctuated from 8.63 to 11.70 mg TE/g and 13.76 to 137.23 mg TE/g, respectively, showing higher scavenging activity of ABTS assay, as well. 59 Even though in literature, there is still a scarcity of results on this topic, the potential of industrial hemp essential oil is peaking the interest of some researchers.12,59
Limitations of the Study
The results of this research provide information about the chemical composition of industrial hemp essential oil and hydrolate. Additionally, the study is dealing with their antimicrobial and antioxidant potential. The significance of this study is the presentation of the chemical composition of volatile compounds in industrial hemp hydrolate and its biological activity. However, it is known that the profile of the hydrolate is related to the corresponding essential oil. Therefore, the main limitations can be considered that this is a case report of one cultivar of industrial hemp whose essential oil dominates sesquiterpenes (trans-caryophyllene, caryophyllene oxide, and α-humulene), while volatile compounds from hydrolate are mainly oxygenated monoterpenes (1,8-cineole, 1,8-menthadien-4-ol, terpinen-4-ol, trans-pinocarveol, and others). It is well known that oxygenated monoterpenes are water-soluble compounds, while sesquiterpenes are practically insoluble in water, therefore it can be considered a relevant hydrolate profile for other industrial hemp cultivars as well. Moreover, hemp expresses wide ranges of essential oil profiles depending on genotypes/cultivar, climatic conditions, phenological stage, postharvest processing, and extraction method, all of these certainly have a significant impact on the hydrolate profile as well. Additional limitations are that monoterpenes and sesquiterpenes have different biological potential, and the differences in chemical composition can significantly affect the change in biological activity. In order to reduce limitations and provide a more complete overview of this issue, it is necessary to expand research to different genotypes and cultivars that are of economic importance, but also to the application of new extraction technologies, especially techniques which can be utilized as pre-extraction media prior to the hydro-distillation such as natural deep eutectic solvent, microwave, etc. Additionally, in vivo studies are desirable to confirm the obtained results.
Conclusion
This research confirmed the previously proven antimicrobial and antioxidant activity of industrial hemp essential oil. Conversely, the novelty is the antimicrobial and antioxidant activity of hydrolate, which is considered waste, but has great potential to be a useful by-product. For this reason, further investigation and discovery of the biological activities of industrial hemp hydrolates would be of great importance due to the increasing public interest in this plant, as well as zero waste technologies.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X241264712 - Supplemental material for Exploring (Un)Covered Potentials of Industrial Hemp (Cannabis sativa L.) Essential Oil and Hydrolate: From Chemical Characterization to Biological Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X241264712 for Exploring (Un)Covered Potentials of Industrial Hemp (Cannabis sativa L.) Essential Oil and Hydrolate: From Chemical Characterization to Biological Activities by Olja Šovljanski, Milica Aćimović, Vladimir Sikora, Anamarija Koren, Anja Saveljić, Ana Tomić and Vele Tešević in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is financially supported by the Ministry of Science, Technological Development and Innovation of the Republic of Serbia, under grant numbers: 451-03-47/2024-01/200134 (Faculty of Technology, University of Novi Sad), 451-03-66/2024-03/200032 (Institute of Field and Vegetable Crops Novi Sad), and 451-03-68/2024-14/200168 (Faculty of Chemistry, University of Belgrade).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

