Abstract
Background
Camphor has a long-term application history in the control of insect pests and their mixture with kerosene is not an exception in the control of mosquitoes.
Objectives
Due to differences in larval biology and positioning in breeding sites, this study was designed to reassess the potency of camphor mixed with kerosene against the emergence of Anopheles mosquitoes in order to ascertain that efficacy of a treatment may not differ greatly with different mosquito species.
Methodology
Larvae and pupae of Anopheles mosquitoes were collected from Ndokwa West Area of Delta State, Nigeria using standard World Health Organization method and left to acclimatize for 6 h in the Insectary before exposure. Mosquitoes were confirmed as Anopheles gambiae. Camphor (2 g) and its mixture with kerosene in equal ratio were introduced into 100, 200, and 400 mL of water which equated 0.02%, 0.01%, and 0.005%, respectively. Anopheles mosquitoes exposed to only water served as control for the experiment. Twenty larvae and pupae in their third stage of larval development was first sorted into 1000 mL glass beaker and then introduced into various treatment concentrations in 3 replicates.
Results
Highest mortality was recorded in larvae exposed to 0.02% of camphor mixed with kerosene. Pupae exposed to same concentration compared favorably. Lethal dosage (LD50 and LD95) of camphor exposed to Anopheles larvae and pupae ranged from 0.002% to 0.004% and 0.021% to 0.491%, respectively. More so, values were 0.002% and range from 0.010% to 0.021% in camphor mixed with kerosene, respectively.
Conclusion
Therefore, the intensification in the use of camphor mixed with kerosene among the population in mosquito breeding sites would reduce the mosquito biting activities and diseases in the locality.
Introduction
Worldwide, vector control interventions (herein as VCIs) is a medical concern adopted to fight mosquito bites which result in high seasonal transmission of malaria in individuals nearly in all ages, especially children and pregnant women. Malaria, according to the World Health Organization (WHO) and Centers for Disease Control and Prevention, is considered one among the top 5 infectious diseases in Africa and the major disease in Nigeria prompting public health action. 1 Global malaria statistics a year ago implicates the disease burden in about 250 million individuals and death at approximately 620 000 persons. 1 Malaria will continue to thrive with repeated bites of Anopheles mosquitoes amid mosquito control intervention due to prevailing resistance in species or without application of intervention. Vector control intervention is primarily aimed at targeting all stages of mosquitoes.
Vector control interventions adopt recommended chemical insecticides of different classes such as pyrethroid, organochlorine, organophosphate, and carbamates in the fight against mosquitoes. 2 Over the years, some of these insecticides have been used to treat mosquito bed nets and to kill mosquitoes in households by spraying. Yet, insecticide resistance due to unprescribed use and overuse of recommended insecticides remains the issue threatening the successful interventions using these methods. 3 This has necessitated the need for research on improving existing formulations of insecticides or developing newer insecticides especially of plant and other viable origin. With insecticide resistance thriving in almost all WHO regions, several innovations in VCIs included the combination of recommended insecticide classes, 4 synergizing insecticides, 5 genetic transplant, 6 application of insecticidal microbes; Wolbachia, 7 techniques applying male mosquitoes exposed to irradiations 8 and introduction of predacious fish species in mosquito breeding site.9,10 In insecticide resistant species, 2 primary mechanisms have been implicated; up regulating insecticide-detoxifying enzymes and altering the insecticide target site to decrease the affinity between the agent and the binding site.11,12 Pyrethroids and organochlorines are commonly used insecticides that target the voltage-gated sodium channel (VGSC). In Anopheles, Culex and Aedes mosquitoes exposed to pyrethroids, knockdown resistance (kdr) has been linked to the mutation of the VGSC gene at codon 1014. 13 Similarly, in mosquitoes exposed to organochlorides, a Gly to Ser amino acid change occurs at codon 119 of the acetylcholinesterase gene 1 (Ace-1) due to mutation, which results in cross-resistance to carbamates and organophosphates. 14 The determination of resistance genes is quite pricy. Thus, the search for cost-effective alternatives becomes necessary to cushion the upregulating resistance in species as well as the altered binding sites. Temephos resistances in larval of Anopheles stephensi was due to CYP450, esterase, alpha, and beta esterase genes. 15
Essential oils derived from leaf, seed and seed pod of Acacia nilotica and other plants have been reported to cause mortality that compared favorably with some chemical insecticides.16,17 For instance, oil extract of Eucalyptus globulus was reported to cause high larval mortality compared to temephos in mosquitoes. 18 Similarly, Acacia nilotica caused favorable larval, pupal, and adult toxicity. 16 The use of native plants and plants based products in mosquito control has gain much importance recently. Other product from petroleum such as petrol, kerosene (Kero here in), engine oil, and waste oil have been traditionally used as insecticides with remarkable success.19,20 Kero acts as larvicide by triggering inflammatory responses that affect the insect physiological balance in their habitat. 21 Camphor, on the other hand, is a natural occurring and affordable organic substance in synthetic form. Unlike kero, camphor and some chemical insecticides activate cytochrome P450 enzyme in neuronal channel which can cause acute mortality of neural cells,15,22 Thus its insecticidal tendencies. Camphor has successfully been tried as an insecticide on other insects.17,23,24 The mixture of camphor with kero as an insecticide has been tested on insects, like termites 4 and even on culicine mosquitoes (Aedes aegypti) and was effective in causing high mortality at high concentrations. 5 In order to knock off the agitations for studies directed at testing same substance on different species, this study was conducted to ascertain the prospective differences in mortality with same treatment as reported in an earlier study by Ojianwuna and Enwemiwe. 25 This study used these test materials because they are available, toxic, and relative affordable. To our knowledge, there are no reports on the efficacy of camphor on Anopheles mosquitoes. Therefore, this study reassessed the potency of camphor and its combination with kero at various concentration against the emergence of Anopheles mosquitoes.
Result and Discussion
Mortality of Anopheles gambiae
Recommendations and guideline developed by WHO 26 in order to test the larvicidal efficacy of insecticidal materials have been used in a previous study by Ojianwuna and Enwemiwe 25 to report the efficacy of camphor and their mixture with kero against the emergence of Aedes aegypti mosquitoes. The bioactivity of toxic materials on specific group of insects is sometimes not location bound, and this present study tends to establish that testing a toxic substance on a close related species is not species bound as this may not cause a separate study in future correspondence whereby the differences in mortality may be close to the prior test. Adequate care is required when designing a study to reassess the potency of a test material already established as toxic in closely related species.
This present study reassessed the potency of camphor mixed with kero as adult emergence inhibition of Anopheles gambiae mosquito larvae and pupae in Abbi, Ndokwa West LGA, Delta State, Nigeria. Larvae exposed to camphor mixed with kero at 0.02% concentration recorded highest mortality. The highest mortality recorded in the best concentration equates the observation in earlier study by Ojianwuna and Enwemiwe. 25 The finding of this study is in tandem with studies by16,18 where oil extracts of plants caused highest mortalities in Anopheles mosquitoes. However, mortality recorded in larvae exposed to 0.02% camphor compared favorably. The lowest mortality was recorded in pupae of Anopheles exposed to 0.005% of camphor (Table 1). All concentrations of the treatment caused mortalities in larvae and pupae. There were notable significant differences in the mortality recorded from various concentrations of treatments exposed to pupa and larva of An gambiae (P < .05). These toxic substances were observably floating on the water of mosquito breeding containers, and the active components in these substances could be the reason for the higher mortality in larvae as they could have caused inflammatory and physiological responses in species as they move to the water top surface to filter feed. Camphor and their mixture with kero has been adopted in several studies with high reports of insect mortality.4,20 More so, their mortality on embedded sand fleas in tungiasis infested individuals has been reported.23,24 A study by Benjamin et al. 27 reported the potency of adopting the steam of camphor mixed with other valid substances in the cure of malaria. The finding of this present study is in agreement with the similar finding of these studies conducted. Mortality of camphor in individuals due to oral routes has equally been reported. 28 As well, oral mortality of these substances can be highly fatal in individuals especially children. 29
Mortality of Anopheles gambiae Exposure to Camphor Mixed With Kero at Various Concentrations.

Mortality with similar adjoining letters do not differ significantly between treatments (P < .05) using Tukey's test. Low and high bounds are 95% CI.
Interestingly, the lowest concentration of these test materials caused above 50% mortality in the mosquitoes (Table 1). It predicts that incorporating these substances in nano-formulated insecticides will increase mortality in parts per millions. Studies on definite mechanism of action of mosquitoes exposed to camphor or their combination with kero are limited.21,22 Thus, this paves a way for novel study in this aspect. Notwithstanding, other aspects of camphor use have been outlined. A study by Sucker et al. 30 has reported that camphor can cause mild acute inflammatory irritation of eyes and nose characterized by redness and swollen eyes as well as severe nasal mucus in industrial workers constantly exposed to inhaling it. Fu et al.31 opined that they can cause high mortality in insects with reduce exposure time. This was observed in this present study as mortality in both life stages increased with increasing time.
Mortality Time of Anopheles gambiae
In this present study, significant mortalities occurred within a short time. The mean time mortality of An gambiae mosquitoes exposed to camphor mixed with kero at different concentrations is presented in Tables 2 and 3. Mortality of both life stages generally increased with time. It was observed that larval mortality was high at lower time compared to pupal mortality. Within the pupal exposure group, mortality was highest in 0.01% kero and camphor combination at 60 min post exposure time. This shows that the mixture of kero with camphor acted as synergists that cause mortality with more reduced time than in camphor exposure alone. This observation was equally parallel to the study of Ojianwuna and Enwemiwe. 25 The mortality records of pupae exposed to camphor mixed with kero compared favorably with the larval exposure. Interestingly, mortality recorded in 0.01% camphor and kero at 15 min equates 0.02% kero and camphor at 60 min. More so, no mortality was recorded in camphor exposure from 10 to 20 min. The reduced mortality could be ascribed to that they were in the quiescent stage and occasionally thrust to the surface. The mild sclerotization of the pupal cuticle could have favored reduced mortality in this group. This observation was equally similar to the report of Ojianwuna and Enwemiwe. 25 The mortality recorded in 0.01% camphor at 30 min equates 0.02% kero and camphor at same time and 0.02% camphor and kero at 15 min. Mortality recorded in 0.01% camphor at 20 min also equated 0.02% of camphor at same time. Within the larval group, no mortality was recorded in 0.005% of kero and camphor at 10 min, and also at 0.005% and 0.01% camphor. The highest time mortality in larval exposure was recorded in 0.01% camphor at 60 min. The differences between time mortalities were significant (P < .05) (Tables 2 and 3). The efficacy of kero alone has been tried on An gambiae in previous studies by Refs.4,19,32 Kero was efficacious against the emergence of An gambiae.
Mortality Time of Anopheles gambiae Pupae Exposed to Camphor Mixed With Kero at Different Concentrations.
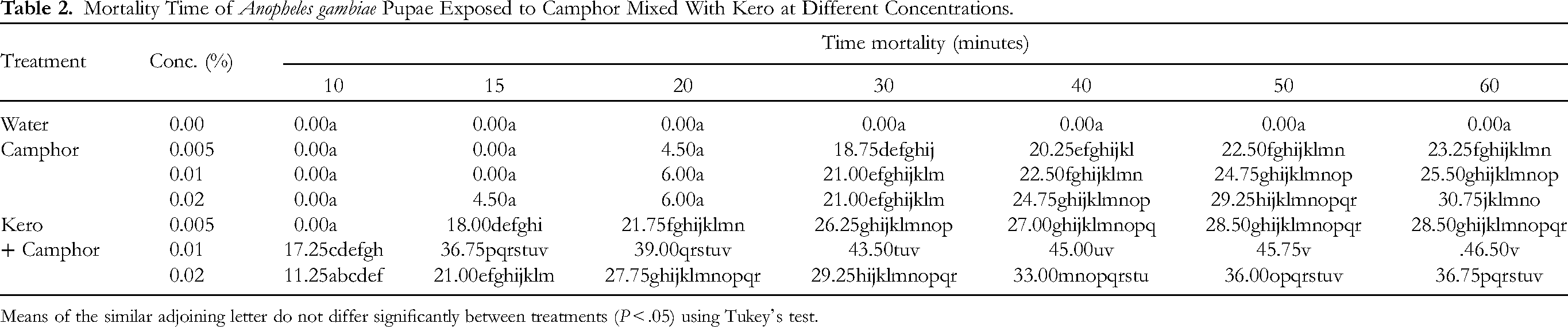
Means of the similar adjoining letter do not differ significantly between treatments (P < .05) using Tukey's test.
Mortality Time of Anopheles gambiae larvae Exposed to Camphor Mixed With Kero at Different Concentrations.

Mean time mortality for all ±2.35; Means of the similar adjoining letter do not differ significantly between treatments (P < .05) using Tukey's test.
Regression Analysis for Time Mortality
Regression square (R2) for larval exposure contributed 73% of the variability to the mortality with time and the difference was highly significant (<0.001). The equation for the larval mortality model is y = 1.07 + 0.44x. The effect size measure (f2) for larval mortality is 2.697 (Figure 1A). The R2 for pupal exposure contributed 33% of the variability to the mortality with time and the difference was also highly significant (<0.001). The equation for the pupal mortality model is y = 11.02 + 0.274x. The effect size measure (f2) for larval mortality is 0.489 (Figure 1B). Regression analysis between the larval and pupal mortality shows that it contributed 42% of the variability to the mortality with time. The difference was also highly significant (<0.001). The equation for the mortality model is y = 1.42 + 0.696x. The effect size measure (f2) for the mortalities is 0.719 (Figure 1C). In this present study, larval exposed to the treatment recorded the highest regression square and effect size measure compared to the pupa and this may be linked to the larval cuticle that allowed for easy penetration of treatment substances.

Regression analysis in larval and pupae of Anopheles gambiae exposed to camphor and its combination with kero.
Lethal Dose Analysis
Probit model analysis of An gambiae mosquitoes exposed to camphor, and camphor with kero is shown in Table 4. Anopheles larvae and pupae exposed to camphor had LD50 and LD95 values between 0.002% and 0.004%, and 0.021% to 0.491%, respectively. Similarly, LD50 and LD95 values of camphor with kero were 0.002, respectively, and 0.010% to 0.021%. The dose of camphor applied alone was insignificant to with their mixture in kero. This dosage was slightly different from those reported with other insects4–5 and mosquitoes. 25
Probit Determination Model of Anopheles gambiae Exposed to Camphor, and its Combination With Kero.

N equates mosquitoes assayed; LD50 and LD95 represents 50% and 95% lethal dosage in grams for camphor and g/mL for treatment mixture.
Adult Emergence and Inhibition
Immature stage of Anopheles exposed to camphor, and camphor with kero at different concentrations is shown in Figure 2. Total adult emergence was observed in the control group. Adult emergence was observed in Anopheles mosquitoes exposed to all concentrations of camphor. Lower concentrations of camphor had more adult emergence, and the differences between the adult emergence were significant (P < .05). No adult emergence was recorded in mosquitoes exposed to camphor and kero mixture. The predicted emergence inhibition for 50% and 95% of the mosquito is −0.006 and −0.040. This corresponded as reported previously, Ojianwuna and Enwemiwe. 25 Complete adult mosquito emergence was observed in the untreated group. The lower concentrations of camphor had more adult emergence. This shows that the concentration left patches of treatment substance that allowed emergence. The observation of mortality in adult mosquitoes that emerged in best concentration occurred in this study. The adoption of these substances in larval management systems is encouraged but with some limitations needing prior attention. It is recommended to measure, before applying this prospecting intervention: (1) the ecological safety of these substances on population of natural enemies of mosquitoes in their natural breeding habitats; (2) intensify collaboration between researcher, community heads, and households on the readiness for intervention; (3) appropriate minimal dosage that will cause complete mortality in larger water quantities; (4) mortality to plants and bioaccumulation in run offs; (5) appropriate time to apply, probably at the onset of dry season when there is lesser downpour.

Adult emergence in pupa exposed to camphor, and camphor with kero at different concentrations. (Means with similar letters are not significantly different using Tukey's test). CP, camphor; PUP, pupae; CP + KS, camphor and kero.
Conclusively, this study has shown that camphor and their mixture with kero caused mortalities in An gambiae with time. The best concentrations significantly inhibited the emergence of adult mosquitoes. Therefore, the intensification in the use of camphor mixed with kero among the population in mosquito breeding sites would reduce the mosquito biting activities and diseases in the locality. However, we recommend that the mechanism of action of these toxic substance adopting molecular diagnostics be addressed as well as the careful redress of ecological safety.
Materials and Methods
Experimental Area
Experimental set up was done in the Entomology unit, of the Department of Animal and Environmental Biology, Delta State University, Abraka. Immature stages of mosquitoes for this study were collected from Abbi, (5.6782° N, 6.2137° E) in Ndokwa West Local Government Area, Delta State.
Anopheles Mosquito Sampling
Wild strain of Anopheles mosquitoes was collected from the natural breeding site using 350 mL dippers and scooping spoons in the early hours of the morning, transported to the Insectary and left to acclimatize following the previous study by Ojianwuna and Enwemiwe. 25 The larvae were fed a mixture of biscuit and yeast before exposure to the various treatment concentration. The emerged adult mosquitoes in control were identified using identification key by Coetzee. 33
Essential Oil Assay
Camphor was grounded into powder and 2 g was used for the exposure in single form. In the combined form, camphor and kero were mixed in the ratio of 1g:1 mL. The stock solution was emptied into 100, 200, and 400 mL of water which corresponded to 0.02%, 0.01%, and 0.005%, respectively. Twenty-third stage larvae and pupae were introduced into the various treatment concentrations in 1000 mL glass beakers. Readings of mortality were taken from 10 min, 15 min, 20 min, 30 min, 40 min, 50 min, and 60 min using the WHO standard manual for larval testing. 26 The exposure was replicated into 3.
Statistical Analysis
Mean mortality and inhibition of emergence were analyzed using XL Stat version 2023. Tables and bar graph were used in result presentation. Means of mosquito mortality and emergence were separated using Tukey's test and analysis of variance was used to determine significant differences at P < .05. Lethal doses for 50% and 95% (LD50 and LD95) of mosquitoes were determined using Probit model analysis. Generalized linear regression analysis was used to monitor the relationship between mortality and time of exposure. The effect size measure (f2) was also recorded. Adult emergence was computed adopting the formula in WHO
34
:
Footnotes
Authors’ Contributions
CC and VN conceptualize the study. All authors collected the field data and experimented. VN analyzed the data. CC supervised the work. All authors interpreted the analyzed data, wrote, and reviewed the manuscript. All authors read and approved the final manuscript.
Availability of Data and Material
All the data are analyzed and presented in the article.
Consent for Publication
The authors have declared the publication of this manuscript in your journal. Please contact any of us via email when necessary.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
