Abstract
Introduction
Nowadays, synthetic pesticides are the main method used in crop protection. However, their excessive use have increased pesticide resistance, environmental contamination, and toxicity and affected human health. 1 This problem has led to restrictions on the use of synthetic pesticides being implemented worldwide. 2 One solution lies in developing the use of pesticides of natural origin, known as biopesticides. 3 These can be essential oils, non-volatile extracts, essential oil hydrolates, or pure natural molecules.4–7
However, since forests require maintenance through periodic pruning, a large mass of residues is generated, which constitutes a sustainable natural resource of interest as a supply of biopesticide ingredients. 8 Juniper forests, mainly composed of Juniperus phoenicea or Juniperus thurifera, can be found throughout the Mediterranean basin such as in Spain, Morocco, Algeria, Turkey, or Greece.9–11 The largest juniper forest in Europe is located in Spain, with an area of about 30 000 hectares. 12 J. phoenicea is a shrub up to 8 meters high while J. thurifera is a tree that reaches 20 m in height. 9 These two species have medicinal applications and have been reported to alleviate diseases such as diarrhea, arthritis, diabetes, 13 and to cure ulcers.14,15 In addition, essential oils (EOs) of J. phoenicea and J. thurifera have shown antimicrobial, antifungal, and cytotoxic activity,9,15–17 and EOs from leaves of J. thurifera have been reported to be used for pest control. Thus, these oils were reported to be active as insecticide against Acanthoscelides obtectus, Tribolium castaneum, and Sitophilus oryzae. They also presented antifungal activity against Alternaria alternata, Fusarium oxysporum, Fusarium solani, Rhizoctonia solani, Verticillium dahlia, 17 Penicillium digitatum, Penicillium expansum, and Aspergillus niger 18 or anti-tick activity against Hyalomma aegyptium. 17
The chemical composition of the different aerial parts (organs) of J. phoenicea and J. thurifera has been previously reported. The major components from the EO of the leaves of J. phoenicea leaves are the monoterpenes α-pinene and phellandrene.11,15,19,20 When considering the European and African varieties of J. thurifera, a remarkable change in the composition was noticed. Thus, the essential oil from the leaves of the European variety is rich in limonene (>50%),21,22 whereas the African variety contains more than 40% of sabinene and α-pinene.19,23
With regard to the composition of the arcestides, the EO of J. phoenicea arcestides is rich in α-pinene with a proportion ranging between 33% and 88%,15,20,24 whereas the EO of J. thurifera arcestides presents a significant disparity, with the European variety being rich in limonene, 25 and the African variety containing mostly mentha-6,8-diene, β-pinene, elemol, and 4-terpineol. 16
Significant differences are also observed in the wood, with the EO of the wood of J. thurifera (European variety) being abundant in sesquiterpenes such as cedrol (>40%), thujopsene, widdrol and α-cedrene (>10% each), 9 while J. phoenicea (var africana) is rich in α-pinene (over 50%) and δ−3-carene (14.5%). 26
In this work, the potential forestry use of J. phoenicea and J. thurifera pruning waste is studied by analyzing the composition of the woods and testing the biological activities of the corresponding components against some pests, with the ultimate target of finding new biopesticide ingredients.
Results and Discussion
Essential Oils, Hydrolates, and Extracts of Sabinas Wood
Essential Oils
The different parts obtained from the pruning of Sabinas, namely, arcestides (berries), leaves, stems, and branches were separated. The branches–stems of each species are finely crushed, and then the sawdust is subjected to steam distillation to provide the corresponding EOs and hydrolates. The latter are extracted over activated charcoal and then eluted with ethyl acetate (EtOAc) to obtain the hydrolate extracts. The resulting plant biomass residues were subjected to Soxhlet extraction with hexane (H) and EtOAc successively to obtain the corresponding extracts. The yield of the essential oil obtained from J. phoenicea was higher than that produced collected from J. thurifera (0.52% vs 0.31%), Considering non-volatile extracts, the quantities extracted from J. thurifera doubled those obtained from J. phoenicea (Table 1). On the other hand, the yields of the hydrolate extracts proved to be very low.
GC-MS Analysis of J. phoenicea EO and Fractions.

Components with percentages higher than 1%.
Each essential oil was then fractionated into two parts by fast elution column chromatography, firstly with hexane to obtain a hydrocarbon fraction (HF), and then with diethyl ether to obtain an oxygenated fraction (OF) (Scheme 1).

Fractionation of J. phoenicea and J. thurifera branches wood. Weights and yield are given in parentheses.
The chemical composition of EOs and of the resulting HF and OF fractions was analyzed by GC-MS (Tables 1 and 2). The chromatograms show that in EO of J. phoenicea wood (
GC-MS Analysis of J. thurifera EO and Their Fractions.
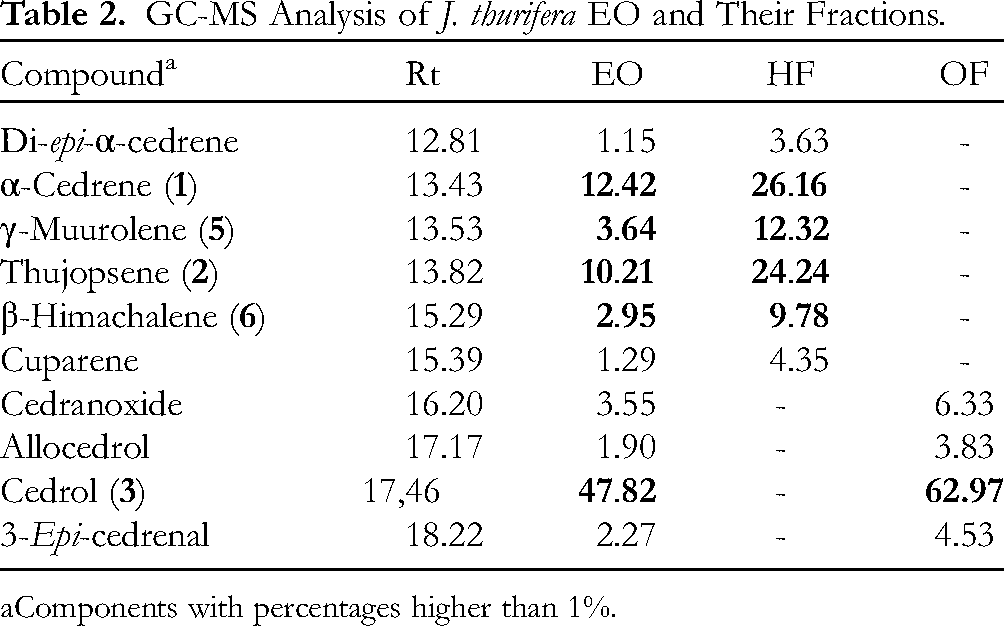
Components with percentages higher than 1%.
The EO of J. thurifera (
Noteworthy, there are three major compounds for both EOs, namely, cedranes

Major constituents identified.
Cedrene (
Cedrol (
Finally, nootkatone (
Hydrolates
The composition of the hydrolates of both species presents a higher proportion of oxygenated terpene components (Table 3). The sesquiterpenes cedrol
GC-MS Analysis of Juniperus species Hydrolates Organic Fractions.

Components with percentages higher than 1%.
Extracts
We continued this study by analyzing the volatile components of the hexane and EtOAc extracts of these species by GC-MS. (Table 4). A significant presence of the sesquiterpene cedrol (
Volatiles of J. phoenicea and J. thurifera from Organic Extracts (Hex and EtOAc). Estimated According to CG-MS.

Components with percentages higher than 1%.
Totarol (

Aromatic diterpenes identified by GC-MS from
Considering that, to the best of our knowledge, the composition of the hexane extracts of the branches wood of these species has not been studied, we addressed the phytochemical study of these hexane extracts by separating their main components using column chromatography and confirming the structures of the isolated natural products by nuclear magnetic resonance (NMR). Compound
The hexane extract J. thurifera (

Compounds isolated from the hexane extracts from J. phoenicea (
The diterpenes cis and trans-communic acids (
Biological Activity
The antifungal effects (spore germination inhibition against A. niger) are shown in Table 5. The essential oil
Antifungal Activity Against A. niger.

The EOs and extracts were tested against the tick Hyalomma lusitanicum (Table 6). Among the EOs, the most active was
Ixodicidal Effects of J. phoenicea and J. thurifera Extracts (EOs and Fractions, Organic Extracts, Hex and EtOAc) Against H. lusitanicum larvae.

Not tested. bFrom Galisteo et al. 6
As previously mentioned, J. phoenicea essential oil (
There are reports on the antifungal effects of Juniperus spp. essential oils, including J. phoenica (rich in α-pinene) against Botrytis cinerea mycelium 50 and hexane, ethanol, and methanol extracts (rich in cedrol, thujopsene, and widdrol) active on wood-rot fungi.51,52
Juniperus essential oils have been described as ixodicidal against several tick species,53,54 including J. thurifera var. Africana (rich in sabinene, α-pinene, and γ-terpinene) against H. aegyptium
17
and J. phoenica (rich in α-terpinyl acetate, α-pinene, and germacene D) against Ixodes ricinus.
55
None of these oils were chemically similar to the ones studied here. Essential oils rich in some of these components have been described as ixodicidal. Cupressus funebris wood essential oil characterized by α-cedrene (
Additionally, some of these compounds (
Conclusion
J. phoenicea essential oil (
We have found that the hexane extracts from the branches wood of the two studied Juniperus species constitute an important source of cedrol, totarol, and nootkatone, respectively; with these natural products presenting an abundance ranging from 50% to 14% with respect to the total weight of the extract.
The different samples obtained from the pruning of both Juniperus species showed antifungal and ixodicidal effects against A. niger and H. lusitanicum. The essential oil
Further work is already in progress, such as the study of the chemical composition of the EtOAc extracts of both species or the biological studies of all the isolated natural products.
All in all, this work proved that Juniperus prunings are renewable and easily accessible sources of pure sesquiterpenes cedrol, thujopsene or nootkatone, and diterpenes such as totarol, all of them presenting interesting bioactivities.
Part Experimental
The wood of Juniperus phoenicea and Juniperus thurifera was collected in the Sierra de Albarracin, Teruel (Spain).
The wood from each plant was air-dried at room temperature for 48 h. Once dried, they were crushed and subjected to hydrodistillation with a Clevenger apparatus for 8 h.
9
After this time, the essential oil (
Each essential oil was partitioned by filtration through column chromatography of silica gel to afford a hydrocarbonated fraction (

EO analysis
The EOs and dichloromethane fraction of the hexanic and EtOAc extracts were analyzed by gas chromatography-mass spectrometry (GCMS) using a Shimadzu GC-2010 gas chromatograph coupled to a Shimadzu GCMS-QP2010 Ultra mass detector (electron ionization, 70 eV). Sample injections (1 μl) were carried out by an AOC-20i and equipped with a 30 m × 0.25 mm i.d. capillary column (0.25 μm film thickness) Teknokroma TRB-5 (95%) Dimetil-diphenylpolisiloxane (5%). Working conditions were as follows: split ratio (20:1), injector temperature 300 °C, temperature of the transfer line connected to the mass spectrometer 250 °C, initial column temperature 70 °C, then heated to 290 °C at 6 °C/min and a Full Scan was used (m/z 35-450). Electron ionization mass spectra and retention data were used to assess the identity of compounds by comparing them with those found in the Wiley 229 and NIST 17 Mass Spectral Database. All extracts (4 μg/μl) were dissolved in 100% DCM for injection.
Flash column chromatography of Jp-Hex: - - - -
A 2 g portion of
Compound
Compound
Compound
Compound - • Flash column chromatography of Jt-Hex - -
A 3.5 g portion of
Compound
Compound - - -
Compound
Compound
Compound - Bioassay - -
Compound
Footnotes
Acknowledgment
The authors thank engineer Victor Marco Gómez for his help for plant harvest (Valdecuenca, Teruel, Spain) and the laboratory assistant Jose Luna.
Author Contributions
JFQM, AFB and AG-C wrote the first draft of the manuscript and formatted the last version. JFQM edited the manuscript. AFB and MFA revised the manuscript. AFB, JFQM, AG and HZ performed the analysis of the composition of the woods, including the isolation of pure compounds. JRL-M, MFA and AG-C performed the biological experiments. All authors have read and agreed to the published version of the manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to the article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work has been supported by grants MINISTERIO de CIENCIA e INNOVACIÓN, PID2019-106222RB-C31/SRA, PID2019-106222RB-C32/SRA (State Research Agency, 10.13039/501100011033) and Unidad Asociada UGR-CSIC Bioplaguicidas: Biotecnología, Síntesis y Diversidad química.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
