Abstract
Objective
The Lauraceae plants comprised high amounts of essential oils, some of which established pharmacological potentials such as anticancer and antimicrobial activities. The Lauraceae essential oils are also used in cuisines and perfumes. The present study provides the chemical analysis of the leaf oils of Beilschmiedia tonkinensis and Lindera gracilipes, collected from Vietnam.
Method
Chemical components in the obtained oils were identified by the GC-FID/MS. The MTT and broth microdilution assays were used to evaluate cytotoxic and antimicrobial effects, respectively. The protein interactions were viewed by a docking study.
Result
Beilschmiedia tonkinensis leaf oil was characterized by sesquiterpene hydrocarbons (66.0%), in which bicyclogermacrene (23.3%), (E)-caryophyllene (21.9%), caryophyllene oxide (9.9%), and spathulenol (6.0%) were the main components. The major chemical class in L gracilipes leaf oil was still sesquiterpene hydrocarbons (64.2%) with bicyclogermacrene (32.2%) being the principal component. Both 2 oil samples (IC50 41.2-44.12 µg/mL) were actively cytotoxic against the proliferation of A-549 cancer cells. In particular, B tonkinensis leaf oil strongly controlled Hep-G2 and MCF-7 cancerous cells with the IC50 values of 20.6 and 9.36 µg/mL, respectively. Beilschmiedia tonkinensis leaf oil also strongly inhibited the Gram (–) bacterium Pseudomonas aeruginosa and the fungus Aspergillus niger with the MIC values of 16 and 32 µg/mL, respectively. By molecular docking approach, bicyclogermacrene interacted with the p38α MAPK cancer protein (PDB ID: 4FA2) with a potential binding affinity of −8.019 kcal/mol, whereas (E)-caryophyllene tends to bind 2 bacterial proteins P aeruginosa QS regulator (PDB ID: 6B8A) and glucosamine-6-phosphate synthase (GlmS) (PDB ID: 2VF5) with the better binding affinities of −6.740 and −6.521 kcal/mol, respectively. The most preferable binding mode was due to hydrophobic π-alkyl and alkyl interactions.
Conclusion
The current result can be seen as a basic foundation in the applications of essential oils of B tonkinensis and L gracilipes for anticancer and antimicrobial treatments. Further phytochemical studies and mechanisms of action are needed.
Introduction
In the laurel family, Beilschmiedia is a genus containing about 278 plants of shrubs and medium trees, which are widely distributed in tropical regions. 1 Various Beilschmiedia species have been traditionally used to cure a variety of diseases, such as uterine cancers, rheumatism, respiratory problems, infectious bacteria, malaria, and tuberculosis. 2 It turns out that Beilschmiedia plants are rich in essential oils. β-Caryophyllene (12.1%), β-eudesmol (24.1%), and δ-cadinene (28.7%) were the predominant components in the aerial part oils of Barclaya kunstleri, Byttneria maingayi, and Beilschmiedia penangiana, respectively. 1 Beilschmiedia pulverulenta aerial part oil with eugenol (45.3%) being the main component has good effects in antioxidative, antimicrobial, anti-inflammatory, antityrosinase, and anticholinesterase examinations. 3 The cytotoxic activity of Beilschmiedia erythrophloia leaf oil was due to the role of β-caryophyllene (22.6%) and α-humulene (21.9%). 4 Beilschmiedia glabra leaf and bark oils containing β-eudesmol (15.4%-19.3%) exerted good antioxidative effects. 5
Lindera is a big genus of about 100 species of shrubs or deciduous trees that are mostly native to tropical and subtropical regions. 6 The medical and therapeutic benefits of Lindera plants are well-known, in addition to their decorative and commercial values. For instance, L aggregata parts have significant effects on rheumatoid arthritis, chronic gastritis, and enuresis, as well as other illnesses of the urinary system. 6 Similar to other genera of the family Lauraceae, Lindera plants are also good reservoirs of essential oils. Lindera glauca fruit oil consisted of more than 40 components, in which (E)-β-ocimene (41.5%) and α-copaene (13.2%) were the major components. 7 L. setchuenensis leaf oil was predominated by palmitic acid (26.6%). 8
Lindera essential oils have also been paid attention to due to their cytotoxic and antibacterial properties.9–11 The leaf oil of L strychnifolia possessed IC50 values of 22.7 to 24.1 µg/mL toward 3 cancerous cell lines A-549, HeLa, and Hep-G2, whereas its root oil generated IC50 values of 23.9 to 38.0 µg/mL. 8 Lindera umbellate essential oil, also known as Kuromoji, caused apoptosis in HL-60 cancer cells. 10 Lindera nacusua leaf oil successfully inhibited the bacterium Staphylococcus aureus and the fungus Candida albicans with the inhibition zones of 23.7 and 23 mm, respectively. 11
For the first time, our current study reports the chemical analysis of essential oils of the leaves of 2 Vietnamese Lauraceae species, Beilschmiedia tonkinensis, and Lindera gracilipes, as well as the uses of these oils in cytotoxicity and antimicrobial treatment.
Results and Discussion
Hydrodistillation of B tonkinensis fresh leaves gave a yellow oil containing 45 identified components (Table 1). In a total of 96.2%, sesquiterpene hydrocarbons accounted for 66.0%, followed by oxygenated sesquiterpenes (21.5%), monoterpene hydrocarbons (8.2%), and nonterpenic components (0.5%). It was also found that B tonkinensis leaf oil was predominated by bicyclogermacrene (23.3%), (E)-caryophyllene (21.9%), caryophyllene oxide (9.9%), and spathulenol (6.0%). There have been a great number of components with more than 1.0%, such as α-humulene (3.5%), (Z)-β-ocimene (2.9%), α-pinene (2.2%), (E)-β-ocimene (2.0%), α-copaene (1.5%), and δ-elemene (1.4%).
Chemical Constituents in the Leaf Oils of 2 Vietnamese Lauraceae Plants.a
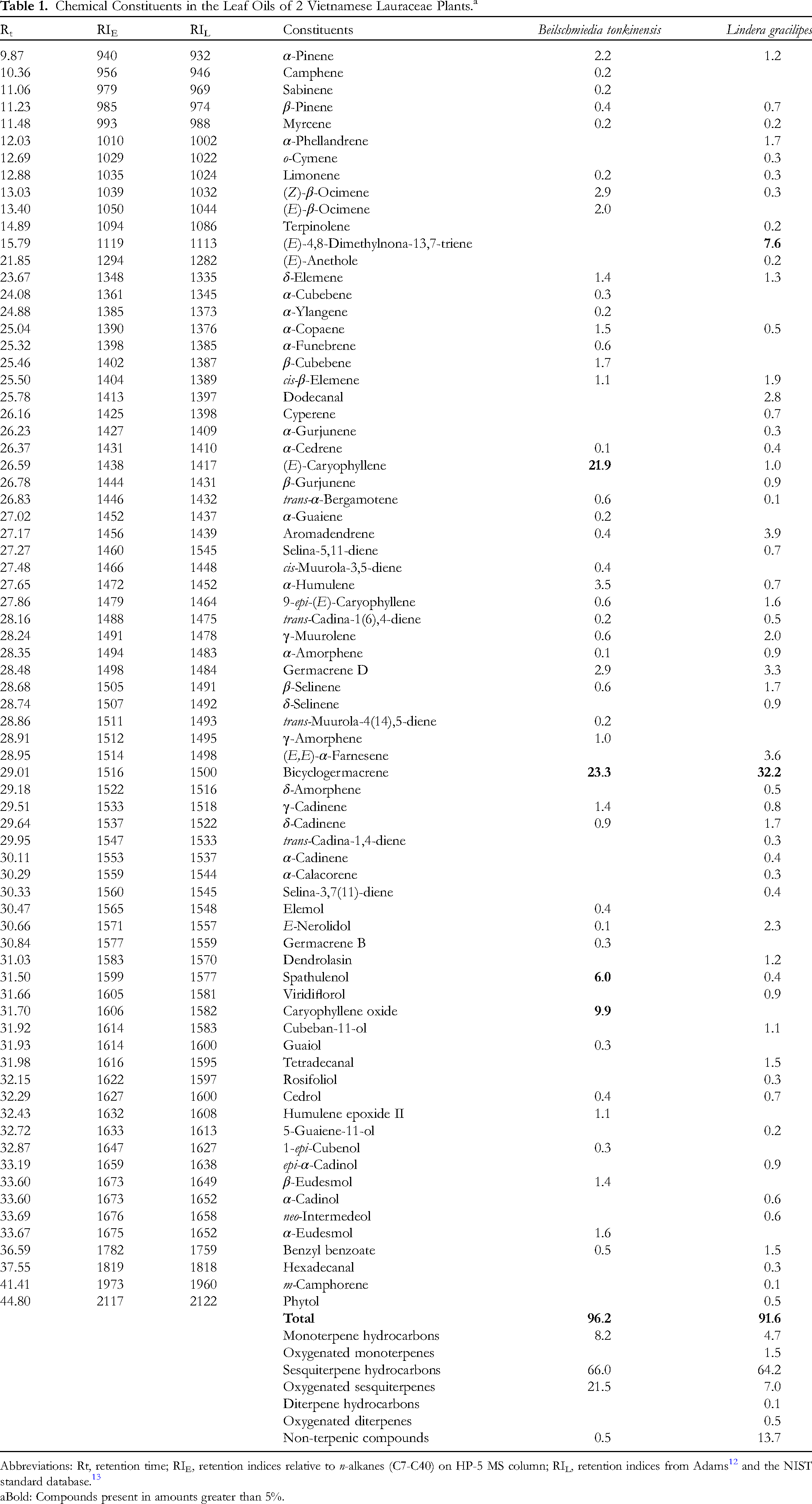
Abbreviations: Rt, retention time; RIE, retention indices relative to n-alkanes (C7-C40) on HP-5 MS column; RIL, retention indices from Adams 12 and the NIST standard database. 13
Bold: Compounds present in amounts greater than 5%.
From Table 1, it turns out that L gracilipes leaf oil contained 55 identified components, which represented 91.6%. As can be seen, the major chemical class was still sesquiterpene hydrocarbons (64.2%), whereas the remaining chemical classes consisted of nonterpenic components (13.7%), oxygenated sesquiterpenes (7.0%), monoterpene hydrocarbons (4.7%), oxygenated monoterpenes (1.5%), and diterpene derivatives (0.6%). As compared to B tonkinensis leaf oil, bicyclogermacrene in this sample was still the main component with 32.2%. However, the amount of (E)-caryophyllene was found to be only 1.0%. Lindera gracilipes leaf oil has been also characterized by the appearance of other identified components, such as (E)-4,8-dimethylnona-13,7-triene (7.6%), (E,E)-α-farnesene (3.6%), dodecanal (2.8%), E-nerolidol (2.3%), and γ-muurolene (2.0%).
Vietnamese Beilschmiedia and Lindera genera have drawn attention due to their essential oil resources. The leaf essential oils of Vietnamese Beilschmiedia plants B erythrophloia, B fordii, B robusta, and B yunnanensis were accompanied by the appearance of the principal components bicyclogermacrene (30.5%), α-pinene (45.1%), (E)-caryophyllene (22.5%), and 9-epi-(E)-caryophyllene (21.2%), respectively.14,15 β-Caryophyllene (29.2%), α-humulene (18.0%), and caryophyllene oxide (14.6%) can be seen as the major components of L glauca leaf oil. 15 The most dominant component in the other Vietnamese Lindera species was camphor (67.5%). 16 Our current results provide new evidence about chemical components analysis for 2 other perennial Lauraceae growing in Vietnam.
Both 2 oil samples were submitted to cytotoxic and antimicrobial assays. From Table 2, the leaf oils of B tonkinensis and L gracilipes exhibited cytotoxic activity against A-549 cancerous cells with IC50 values of 44.12 and 41.2 µg/mL. Significantly, B tonkinensis leaf oil successfully controlled the proliferation of Hep-G2 and MCF-7 cancerous cells with the corresponding IC50 values of 20.6 and 9.36 µg/mL. In contrast, L gracilipes leaf oil did not exhibit activity (IC50 > 100 µg/mL). The current results match well with previous reports. For instance, Su and Ho revealed that B erythrophloia leaf oil was responsible for the IC50 value of 38.8 µg/mL. 3 As mentioned above, cytotoxicity of L strychnifolia leaf oil against A-549 and Hep-G2 cells exerted the IC50 values of 23.1 and 22.7 µg/mL, respectively. 7
Cytotoxicity of 2 Tested Oil Samples.

Regarding antimicrobial assay, B tonkinensis leaf oil inhibited the proliferation of 2 Gram (+) bacterial strains S aureus and Bacillus subtilis with the same MIC value of 64 μg/mL, but the second sample did not show activity (MIC > 400 μg/mL) (Table 3). Both 2 oil samples were active against 2 Gram (–) bacteria Escherichia coli and Pseudomonas aeruginosa. Especially, B tonkinensis leaf oil showed strong activity against P aeruginosa with the MIC value of 16 μg/mL, in comparison with that of the standard tetracycline (MIC 8 μg/mL). Likewise, B tonkinensis leaf oil has good results (MIC 32-64 μg/mL) against 2 fungi Aspergillus niger and Fusarium oxysporum, in comparison with those of L gracilipes leaf oil (MIC 64-128 μg/mL). In the final case, B tonkinensis leaf oil possessed MIC values of 32 to 64 μg/mL against 2 yeasts C albicans and Saccharomyces cerevisiae, whereas L gracilipes leaf oil only exhibited activity against S cerevisiae with a MIC value of 128 μg/mL. In literature, the leaf and bark oils of B madang were moderately active against B subtilis and S aureus with a MIC value of 125 µg/mL. 17 Beilschmiedia fordii leaf oil generated remarkably strong antimicrobial activity against S aureus, B cereus, and C albicans with a MIC value of 16 µg/mL. 15 In another example, B pulverulenta aerial part oil suppressed B subtilis and S aureus with the MIC value of 62.5 µg/mL, as well as monitoring the growth of C albicans and S cerevisiae with a MIC of 125 µg/mL. 2 Collectively, Beilschmiedia essential oils are appropriate for further antimicrobial treatments (Figure 1).
Antimicrobial Activity of 2 Tested Oil Samples (MIC: μg/mL).

Molecular Docking Study
It was supposed that 2 main components, namely bicyclogermacrene and (E)-caryophyllene, established a great role in the antimicrobial activity of B tonkinensis leaf oil. Hence, they were subjected to experimental molecular docking. This procedure aimed to determine their binding affinity and optimal docking poses at the active sites of the target protein. The redocking experiments were conducted using Auto Dock Vina v1.2.3 software, resulting in the RMSD (root mean square deviation) values of 0.261688, 0.286839, and 1.24192 Å when aligning the ligands with their corresponding redocking poses (Figure 2). These findings validated the reliability of the molecular docking experiments. Additionally, the cocrystallized ligands, within the protein's crystal structure, human P38α MAPK (PDB ID: 4FA2), MvfR (PDB ID: 6F8A), and glucosamine-6-phosphate synthase (GlmS) (PDB ID: 2VF5) have been considered as reference inhibitory components, exhibiting respective binding affinities of −10.67, −10.42, and −8.43 kcal/mol, respectively.

The major compounds in the leaf oil of Beilschmiedia tonkinensis (a) and Lindera gracilipes (b).

Validation of the docking protocol through the re-docking of the co-crystallized ligands in (A) 4FA2, (B) 6B8A, (C) 2VF5 proteins, and superimposing their docked poses at the active binding pocket.
The p38 MAPK (p38 mitogen-activated protein kinase) is a crucial signal transduction pathway that activates a complex network of interacting proteins mainly responsible for various cellular performances.18–21 Various studies have indicated that p38α MAPK plays a significant role in cancer development, particularly showing substantial expression in invasive breast cancer. 22 Moreover, recent research has revealed that p38 α inhibitors can be used for the deactivation of ER-negative breast cancer and p53-mutated tumors. 23 Therefore, the p38α MAPK target was chosen for this study. To have an insight understanding of the binding mode of the studied components at the binding site of the p38α MAPK protein (PDB ID: 4FA2), molecular docking experiments were performed using the AutoDock Vina program. The binding affinity of bicyclogermacrene and (E)-caryophyllene was found to be −8.019 and −7.955 kcal/mol, respectively (Table 4). The molecular interactions of the docked poses within the active site region are depicted in Figure 3. The main interactions observed between the 2 studied components were predominantly alkyl-type interactions, focusing primarily on amino acid residues Ala51, Met109, Ala157, Lys53, Ile84, Leu167, Leu171, and Val38. Furthermore, an alkyl interaction with Leu108 residue was only observed in the complex formed by 4FA2 protein and bicyclogermacrene, but not in the complex formed by 4FA2 protein and (E)-caryophyllene. Notably, the residues Ala51, Lys53, Leu171, Leu167, and Met109 are crucial positions within the active site of the 4FA2 protein. 24 These findings demonstrate that these main bioactive components can effectively interact with and inhibit the p38 α MAPK pathway.
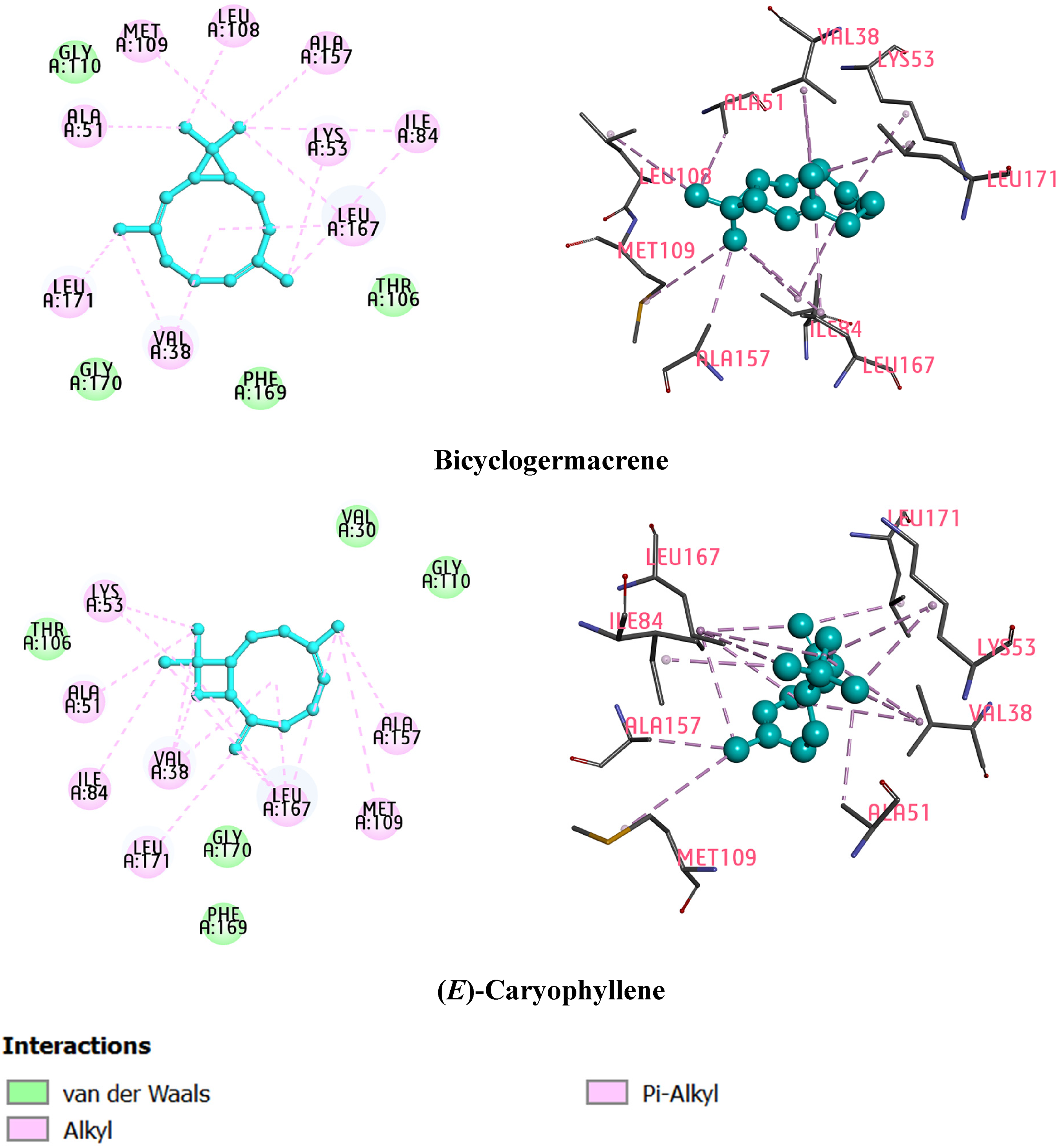
Binding interactions of compounds bicyclogermacrene and (E)-caryophyllene in the cavity of 4FA2 protein.
Docking Results of the Major Compounds of Beilschmiedia tonkinensis Leaf Oils.

Regarding antimicrobacterial activity, P aeruginosa QS regulator (or a multiple virulence factor regulator (MvfR)) (PDB ID: 6B8A) and GlmS (PDB ID: 2VF5) are the 2 protein targets selected in this study. MvfR is an important target for the control and treatment of P aeruginosa infection caused by the direct and indirect activation of a variety of virulence genes, responsible for the formation and development of biofilms. 25 Besides, GlmS played a crucial role in microbial cell membranes by transferring fructose-6-phosphate to glucosamine-6-phosphate in the presence of glutamine. 26 Therefore, in this study, docking experiments were applied to evaluate the behavior of the studied components in the active site of the 6B8A and 2VF5 proteins. From Table 4, the binding affinity of bicyclogermacrene and (E)-caryophyllene was −6.394 and −6.74 kcal/mol for 6B8A protein, and −5.964 and −6.521 kcal/mol for 2VF5 protein, respectively. (E)-Caryophyllene has been shown to inhibit proteins 6B8A and 2VF5 more effectively than bicyclogermacrene. The diagram interaction analysis shows that the main interactions between the 2 studied components were the alkyl and π–alkyl interaction types (Figure 4). In the active region of the protein 6B8A, both components formed interactions with residues Leu189, Tyr258, Trp234, Val211, Ile236, and Val170, and further (E)-caryophyllene also formed with the residue Ile263. It should be noted that Tyr258 Val211, Ile236, Val170, and Ile263 are important amino acids in the active region of the protein 6BA8. Similarly, observations of interactions between the protein 2VF5 and selected components also indicated that the main type of interaction is the alkyl type (Figure 5). Bicyclogermacrene formed these 3 interactions at the amino acid residue sites of Leu601, Leu484, and Valu605 while (E)-caryophyllene formed interactions with Leu484 and Cys300. In summary, docking results have demonstrated the antimicrobial effect of the major components of B tonkinensis essential oils.

Binding interactions of compounds bicyclogermacrene and (E)-caryophyllene in the cavity of 6B8A protein.

Binding interactions of compounds bicyclogermacrene and (E)-caryophyllene in the cavity of 2VF5 protein.
Conclusion
This is the first time that chemical components of the leaf oils of 2 Vietnamese Lauraceae plants B tonkinensis and L gracilipes were identified by the GC-FID/MS analytical procedure. For both 2 oils, sesquiterpene hydrocarbons (64.2%-66.0%) were the main chemical class, as well as bicyclogermacrene (23.3%-32.2%), can be seen as the main components. Both 2 oil samples exhibited cytotoxicity toward A-549 cancer cell lines. In particular, B tonkinensis leaf oil strongly controlled the proliferation of Hep-G2 and MCF-7 with the IC50 of 9.36 to 20.6 µg/mL. Two oil samples exhibited antimicrobial activity at different levels, in which B tonkinensis leaf oil showed strong activity against the Gram (–) bacterial strain P aeruginosa and fungal strain A niger with MIC values of 16 to 32 µg/mL. Molecular docking studies showed a good binding affinity, in which bicyclogermacrene was the main contributor to MCF-7 cytotoxicity activity, sharing many important residues with the p38α MAPK active site, whereas (E)-caryophyllene remarkably bound 2 bacterial proteins P aeruginosa QS regulator and glucosamine-6-phosphate synthase (GlmS). As a result, they may act as strong inhibitors when synergizing with other minor components.
Experimental
Materials
Beilschmiedia tonkinensis fresh leaves were gathered from Pu Huong-Nghean, Vietnam, whereas L gracilipes fresh leaves were collected from Thanh Chuong-Nghean, Vietnam, in March 2023. Botanical taxonomy was confirmed by the coauthor Dr Do Ngoc Dai. Two voucher specimens, BTL-2023 (B tonkinensis) and LGL-2023 (L gracilipes), have been deposited in the faculty of chemistry, HUP2 University.
Distillation
The fresh leaves of B tonkinensis (1.0 kg) were cut into small pieces, and then submitted to water distillation using a Clevenger-type apparatus for 3.2 h, to afford a yellowish oil (0.6% w/w). The obtained oil was dried over Na2SO4 and maintained in a sealed vial at −10 °C for further analysis. The same manner was assigned to the fresh leaves of L gracilipes (1.0 kg), to yield a yellowish oil with 0.5% w/w.
The GC-FID/MS Analyses
Chemical components in the studied essential oils were identified by the GC-FID/MS (gas chromatography-flame ionization detection/mass spectrophotometry) analyses as previously reported.27–29 The GC analysis was performed using an HP 7890A Plus Agilent Technologies gas aided by an FID and coupled with an HP-5MS column (60 m × 0.25 mm i.d., s 0.25 μm film thickness). The temperature of the oven was kept at 55 °C for 3 min and then operated at 240 °C and a rate of 3 °C/min. The corresponding temperatures of the injector and detector were 260 °C and 270 °C. Helium (1.0 mL/min) was used as a carrier gas. The injection volume of the oil sample was 1.0 µL.
The GC-MS analysis has been carried out using the same HP-5MS column and coupled with a mass HP 5973 MSD spectrometer. The temperature of the oven was 55 °C to 240 °C at a rate of 3 °C/min. He (1 mL/min), split ratio 9:1, ionization energy of 70 eV, emission current of 40 mA, sampling rate of 1.0 scan/s, and mass scanning of 50 to 450 amu were established. An EI source at 240 °C was used, while the interface temperature was 270 °C. The retention index (RI) of each component was calculated by a comparison between the experimental result and a homologous series of n-alkanes C7-C40. Each chemical was identified by comparing its RI value to that in literature.12,13 Based on the NIST 20 and WILEY 12 Libraries, the MS fragmentations were compared to those of other essential oils with known compositions, whereas the peak area in the GC-FID was used to calculate the relative percentage (%) without using any correction factor.
Cytotoxic and Antimicrobial Assays
Cytotoxic and antimicrobial experiments were deduced from our previous reports.27–31
Docking Calculation
Docking simulations have been conducted to consider the interactions between the main components of B tonkinensis leaf oil and the targeted proteins. Auto Dock Vina v1.2.332,33 was employed for the simulations, utilizing a united atom scoring function and generating a maximum of 20 binding modes. The global search exhaustiveness was set to 400. The structures of the target proteins have been collected from the RCSB PDB database 34 and underwent preprocessing steps, including the deletion of water molecules, the addition of hydrogen atoms and missed residues, and the Kollman charge, before initiating the molecular docking process. Gasteiger charges were incorporated into the ligand molecules, which were then converted to the PDBQT format. Auto Dock Tools v.1.5.6 was utilized to confirm a grid box of size 22 × 22 × 22 Å with a grid spacing of 1 Å. During docking, the receptor molecule was held rigid while the ligand molecules remained flexible. The best docking poses were examined to identify conformers with the lowest binding energy and to analyze 2-dimensional interactions. The docked conformations and 3-dimensional target-ligand interactions were visualized using BIOVIA Discovery Studio v2021.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231224995 - Supplemental material for The Leaf Oils of Beilschmiedia tonkinensis (Lecomte) Ridl. and Lindera gracilipes H. W. Li: Chemical Composition, Cytotoxicity, Antimicrobial Activity, and Docking Study
Supplemental material, sj-docx-1-npx-10.1177_1934578X231224995 for The Leaf Oils of Beilschmiedia tonkinensis (Lecomte) Ridl. and Lindera gracilipes H. W. Li: Chemical Composition, Cytotoxicity, Antimicrobial Activity, and Docking Study by Duong Quang Huan, Nguyen Dinh Luyen, Nguyen Xuan Ha and Do Ngoc Dai, Nguyen Quang Hop, Do Thi Lan Huong, Ninh The Son in Natural Product Communications
Footnotes
Authors's note
Ninh The Son is also affiliated with Graduate University of Science and Technology, VAST, Caugiay, Hanoi, Vietnam.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by Hanoi Pedagogical University 2 foundation for Sciences and Technology Development via grant number: HPU2.2023-UT-05.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
