Abstract
Background
Only a few studies have investigated the bioactivity of the essential oils (EOs) of Chamaecyparis formosensis and C. obtusa var. formosana, which are endemic to Taiwan. The wood and leaf EOs extracted from C. obtusa var. formosana had anti-inflammatory and antitermitic effects, respectively, and the twig EO extracted from C. formosensis inhibited the growth of Fusarium oxysporum, Pestalotiopsis funerea, and Ganoderma australe.
Methods
A poisoned food technique was used to evaluate the antifungal effects of wood and leaf EOs extracted from C. formosensis and C. obtusa var. formosana on brown rot fungus, white rot fungus, and mold. A phytatray chamber assay was conducted to investigate the antifungal activities of the EOs in the vapor phase. Gas chromatography-mass spectrometry was used to examine the chemical constituents of both EOs.
Results
The leaf EO extracted from C. formosensis primarily contained monoterpene hydrocarbons, whereas the leaf EO extracted from C. obtusa var. formosana primarily contained monoterpene and sesquiterpene hydrocarbons. The wood EOs extracted from C. formosensis and C. obtusa var. formosana primarily contained sesquiterpene hydrocarbons and oxygenated sesquiterpenes. These results indicated that the wood EOs had higher antifungal activity in the liquid phase, whereas the leaf EOs had higher antifungal activity in the vapor phase.
Conclusion
Wood EOs extracted from C. formosensis and C. obtusa var. formosana have strong antifungal effects against Gloeophyllum trabeum, Lenzites betulina, Trametes versicolor, and Trichoderma sp. Compared with wood EOs, leaf EOs have a greater inhibitory effect on mycelial growth in the vapor phase. In both the liquid and vapor phases, the EOs extracted from C. formosensis and C. obtusa var. formosana serve as effective antifungal agents against pathogenic fungi. Further research is required to elucidate the mechanisms underlying the antifungal activities of EOs in different phases and explore their potential applications.
Keywords
Introduction
The genus Chamaecyparis (Cupressaceae) includes 6 species that are found only in Taiwan, Japan, and the United States. 1 Chamaecyparis formosensis and Chamaecyparis obtusa var. formosana, 2 species that are endemic to Taiwan, are among the 5 most valuable conifers in Taiwan, with the other 3 being Taiwania cryptomerioides, Calocedrus macrolepis var. formosana, and Cunninghamia konishii. C. formosensis, better known as Taiwan red cypress, grows in the central mountain range of Taiwan at elevations of 1500 to 2150 m.2,3 Renowned for its excellent quality wood and attractive fragrance, C. formosensis has a thin bark of pale pinkish color. By contrast, C. obtusa var. formosana grows at elevations of 1300 to 2800 m and has a pungent odor and thicker pale-yellowish bark.2,3
Few studies have investigated the bioactivity of essential oils (EOs) from C. formosensis and C. obtusa var. formosana. Wood EO from C. obtusa var. formosana has anti-inflammatory effects, 4 and the leaf EO from C. obtusa var. formosana has antitermite properties. 5 The twig EO from C. formosensis at concentrations of 500 μg/mL inhibited the growth of Fusarium oxysporum, Pestalotiopsis funereal, and Ganoderma austral, with antifungal indices of 92.7%, 71.1%, and 87.7%, respectively. 6 Kuo et al 7 discovered that the wood EO from C. formosensis has insecticidal properties, giving it potential as an insecticide against mosquito larvae and silverfish. Wang et al 8 demonstrated that the wood EO from C. formosensis exhibits antifungal effects against Laetiporus sulphureus and Trametes versicolor, with significant effects exhibited at concentrations of 50 and 100 μg/mL, respectively. However, the aforementioned studies were performed only with agar plate assays, and one can only understand the bioactivity in the liquid phase. The EO in the vapor phase, even at relatively low concentrations, may be more effective at inhibiting the growth of pathogens compared with the EO in the liquid phase. 9 Evaluating the antifungal properties of the EO in the vapor phase may reveal novel potential applications for Chamaecyparis species.
This study examined the antifungal activities of wood and leaf EOs from both C. formosensis and C. obtusa var. formosana against brown rot fungus, white rot fungus, and mold by using the poisoned food technique. Additionally, a phytatray chamber assay was performed to investigate the antifungal activities of wood and leaf EOs from these 2 species in the vapor phase. The chemical constituents of the wood and leaf EOs from both C. formosensis and C. obtusa var. formosana were analyzed using gas chromatography–mass spectrometry.
Results and Discussion
Yields and Chemical Constituents of EOs
The yields of wood and leaf EOs obtained from C. formosensis were 1.7% ± 0.1% and 0.4% ± 0.1% (w/w) on a dry weight basis, respectively, and the yields of wood and leaf EOs obtained from C. obtusa var. formosana were 1.6% ± 0.1% and 1.0% ± 0.1%, respectively (Table 1). Evidently, the wood yielded more EO than did leaves. According to Lin et al, 10 the yields of wood EO from C. formosensis and from C. obtusa var. formosana ranged from 1.3% to 2.4% and from 1.7% to 2.6% (w/w), respectively. According to Wu and Wang, 11 the yields of heartwood EO from C. formosensis and C. obtusa var. formosana ranged from 1.1% to 1.8% and from 2.7% to 3.3% (w/w), respectively.
Constituents and Relative Contents (%) of Wood and Leaf Essential Oils from C. formosensis and C. obtusa var. formosana.

AIs relative to n-alkanes (C9-C22) on a DB-5 ms column.
Not detected.
The 34 chemical constituents identified in the wood and leaf EOs obtained from C. formosensis and C. obtusa var. formosana are listed in Table 1 in order of their arithmetic indices (AIs) on a DB-5 ms column. Each constituent was classified as a monoterpene hydrocarbon, oxygenated monoterpene, sesquiterpene hydrocarbon, oxygenated sesquiterpene, or others. In total, 14 constituents were identified in the wood EO from C. formosensis (Table 1), with the predominant constituents being oxygenated sesquiterpenes (54.2%) and sesquiterpene hydrocarbons (38.7%). The oxygenated sesquiterpenes included α-cadinol (18.3%), τ-muurolol (11.3%), 1-epi-cubenol (10.6%), and τ-cadinol (8.8%), and the sesquiterpene hydrocarbons included δ-cadinene (26.1%), α-calacorene (4.2%), and cis-calamenene (2.6%). The predominant oxygenated monoterpene was trans-myrtanol (7.1%).
Seven constituents were identified in the leaf EO from C. formosensis (Table 1), which was characterized by a high concentration of monoterpene hydrocarbons (98.8%), including α-pinene (91.5%), β-myrcene (3.9%), and β-pinene (2.5%). Sesquiterpene hydrocarbons, predominately α-humulene and δ-cadinene, accounted for 1.2% of the EO. Other studies have similarly found that α-pinene is the main constituent of the leaf EO from C. formosensis.3,12
In total, 15 constituents were identified in the wood EO from C. obtusa var. formosana, with the predominant constituents being sesquiterpene hydrocarbons (51.4%) and oxygenated sesquiterpenes (36.3%). The predominant sesquiterpene hydrocarbons were γ-cadinene (19.0%), δ-cadinene (13.6%), and α-muurolene (9.7%), and α-cadinol (15.8%), τ-muurolol (9.7%), and τ-cadinol (8.1%) were the predominant oxygenated sesquiterpenes. The predominant monoterpene hydrocarbon was α-pinene (10.6%).
Fourteen constituents were identified in the leaf EO from C. obtusa var. formosana, with the predominant constituents being monoterpene hydrocarbons (64.5%), including sabinene (45.2%), β-myrcene (6.2%), γ-terpinene (4.9%), and α-terpinene (2.9%). The predominant sesquiterpene hydrocarbons were cis-thujopsene (28.2%) and β-himachalene (0.4%). Two oxygenated monoterpenes, namely terpinen-4-ol (3.4%) and terpinolene (1.1%), and 1 oxygenated sesquiterpene, namely β-elemol (2.6%), were also identified.
Antifungal Activity of Wood and Leaf EOs from 2 Chamaecyparis Species
The antifungal activities of wood and leaf EOs from C. formosensis and C. obtusa var. formosana were evaluated using 4 test strains of fungus, namely G. trabeum (brown rot fungus), L. betulina (white rot fungus), T. versicolor (white rot fungus), and Trichoderma sp. (mold). The antifungal activities of the EO in the liquid and vapor phases were evaluated using the poisoned food technique and a phytatray chamber assay, respectively.
The antifungal effects of EO (400 μg/mL) determined using the poisoned food technique are shown in Figure 1. Wood EO from C. formosensis completely inhibited the growth of G. trabeum, L. betulina, T. versicolor, and Trichoderma sp., with half-maximal inhibitory concentration (IC50) values of 88, 56, 50, and < 25 μg/mL, respectively (Table 2). By contrast, the leaf EO from C. formosensis had antifungal indices against the 4 fungi of < 50.0% and IC50 values of > 400 μg/mL, indicating that the leaf EO from C. formosensis did not inhibit the growth of any of the 4 test strains of fungi. The wood EO from C. obtusa var. formosana had IC50 values of 121, 123, 396, and 35 μg/mL against G. trabeum, L. betulina, T. versicolor, and Trichoderma sp., respectively (Figure 1), indicating growth inhibition rates of 84.7%, 100.0%, 55.3%, and 96.9%, respectively. Similarly, the leaf EO from C. obtusa var. formosana inhibited the growth of G. trabeum, L. betulina, T. versicolor, and Trichoderma sp. by 46.7%, 37.8%, 20.0%, and 70.2%, respectively, with IC50 values of 578, 804, > 1000, and 158 μg/mL, respectively (Table 2).

Antifungal activities of essential oil in the liquid phase obtained from the wood and leaves of C. formosensis and C. obtusa var. formosana against 4 fungi determined using the poisoned food technique. Each experiment was performed 5 times, and the results were averaged (n = 5). Numbers followed by different letters (a-g) indicate significant difference at the level of P < .05 according to Scheffe's test. Abbreviations: CFW, C. formosensis wood; CFL, C. formosensis leaf; COFW, C. obtusa var. formosana wood; COFL, C. obtusa var. formosana leaf.
Half-Maximal Inhibitory Concentration (IC50) Values (μg/mL) of Wood and Leaf Essential Oils from C. formosensis and C. obtusa var. formosana Against 4 Fungal species.
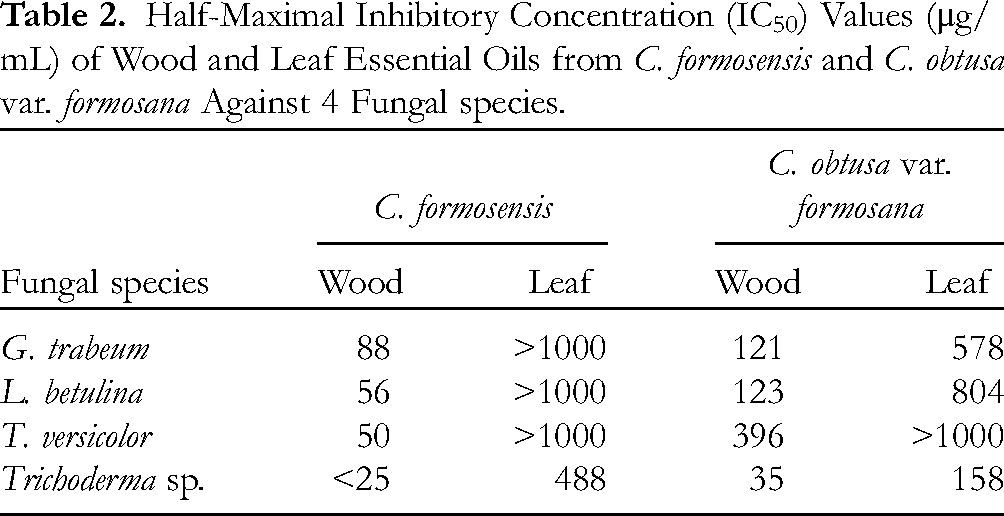
According to the results obtained using the poisoned food technique, the wood EOs from both C. formosensis and C. obtusa var. formosana exhibited high levels of activity against the 4 fungal strains, with antifungal indices exceeding 55.3%. In particular, the wood EO at a concentration of 400 μg/mL completely inhibited the growth of white rot fungus (L. betulina). Regarding IC50 values, antifungal activities under direct EO exposure to G. trabeum, T. versicolor, and Trichoderma sp. were in the order of wood EO from C. formosensis (88, 50, and < 25 μg/mL, respectively) > wood EO from C. obtusa var. formosana (121, 396, and 35 μg/mL, respectively) > leaf EO from C. obtusa var. formosana (578, > 1000, and 158 μg/mL, respectively) > leaf EO from C. formosensis (> 1000, > 1000, and 488 μg/mL, respectively) (Table 2).
The wood EO exhibited stronger antifungal activity than did the leaf EO under direct exposure to the 4 strains of fungi; this finding was consistent with those of other studies. Cheng et al13,14 have investigated the antifungal effects of EOs from the bark, leaves, sapwood, and heartwood of Cryptomeria japonica and from the wood and leaves of Cunninghamia konishii and have found that wood EO but not leaf EO exhibits strong antifungal effects against plant pathogenic fungi. Several studies14–19 have investigated the antifungal effects of the primary constituents of EO. According to Chang et al, 15 sesquiterpenoids were more effective than monoterpenoids in inhibiting mycelial growth of plant-pathogenic fungi. The sesquiterpenoids, β-caryophyllene, caryophyllene oxide, τ-muurolol, and α-cadinol exhibited excellent antifungal activities; in particular, τ-muurolol and α-cadinol exhibited strong inhibitory activities against Rhizoctonia solani, Fusarium oxysporum, Colletotrichum gloeosporioides, Pestalotiopsis funerea, Ganoderma austral, and Fusarium solani and have potential for use as antifungal agents for the control of fungal diseases in plants. 15 Similar observations have been made in other studies.14,16–19 That is, sesquiterpene hydrocarbons including humulene, α-copaene, germacrene-D, β-caryophyllene, δ-cadinene, isoledene, and γ-muurolene and oxygenated sesquiterpenes including τ-cadinol, cedrol, and caryophyllene oxide were found to exhibit favorable antifungal activities. In view of these findings, the antibiotic activities of wood EO can be attributed to these sesquiterpene compounds.
In the present study, the antifungal activities of the EOs in the vapor phase, determined using a phytatray chamber assay, are illustrated in Figure 2. The leaf EO from C. formosensis exhibited the strongest antifungal activity, as indicated by growth inhibition rates of 66.1%, 82.5%, 86.4%, and 86.1% against G. trabeum, L. betulina, T. versicolor, and Trichoderma sp., respectively. The antifungal indices under exposure to EO vapor against G. trabeum, T. versicolor, and Trichoderma sp. were in the order of leaf EO from C. formosensis (82.5%, 86.4%, and 86.1%, respectively) > leaf EO from C. obtusa var. formosana (57.3%, 52.7%, and 60.0%, respectively) > wood EO from C. obtusa var. formosana (36.8%, 37.0%, and 28.2%, respectively) > wood EO from C. formosensis wood (17.3%, 25.9%, and 23.0%, respectively) (Figure 2), and the antifungal indices against G. trabeum were in the order of leaf EO from C. formosensis (66.1%) > wood EO from C. formosensis (65.1%) > leaf EO from C. obtusa var. formosana (49.5%) > wood EO from C. obtusa var. formosana wood (16.4%).
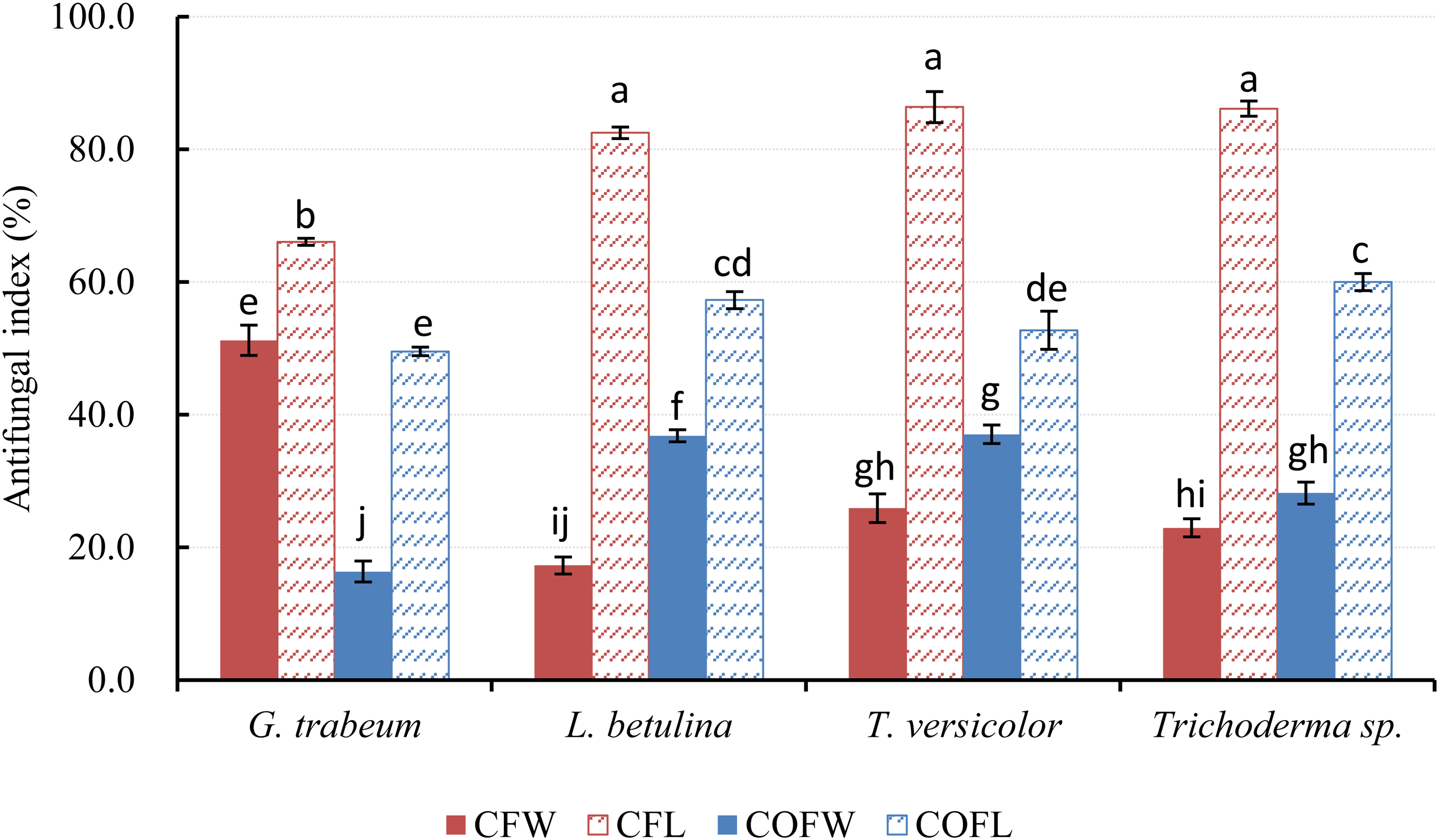
Antifungal activities of essential oil in the vapor phase obtained from the wood and leaves of C. formosensis and C. obtusa var. formosana against 4 fungi determined using phytatray chamber assay. Each experiment was performed 5 times, and the results were averaged (n = 5). Numbers followed by different letters (a-j) indicate significant differences at the level of P < .05 according to Scheffe's test. Abbreviations: CFW, C. formosensis wood; CFL, C. formosensis leaf; COFW, C. obtusa var. formosana wood; COFL, C. obtusa var. formosana leaf.
Notably, for the 4 strains tested, with the exception of G. trabeum, the antifungal activities of the EO vapor determined using the phytatray chamber assay exhibited an opposite trend compared with those of the EO liquid determined using the poisoned food technique. Overall, leaf EO vapors had significantly higher antifungal activity than did wood EO vapors. Comparable antimicrobial activity can be achieved with a smaller amount of EO in the vapor phase than that in the liquid phase.9,20 Volatile EO vapor may have different modes of antifungal activity against fungi. 21 The effects of volatile EO compounds on cell metabolism have been investigated. 22 In terms of their chemical constituents, leaf EOs from both C. formosensis and C. obtusa var. formosana were observed to contain predominantly monoterpene hydrocarbons, whereas wood EOs were comprised primarily of sesquiterpene hydrocarbons and oxygenated sesquiterpenes. Compared with wood EO, leaf EO had a lower proportion of polar compounds and had constituents of lower molecular weights; consequently, the volatility of leaf EOs was higher than that of wood EOs. The relatively strong antifungal activities exhibited by the EO vapors in the present study may be attributed to their higher volatility and more favorable penetration ability of monoterpenes in gaseous form, which facilitate their solubilization in cell membranes.
Conclusion
The present findings revealed strong antifungal potential of wood EO from both C. formosensis and C. obtusa var. formosana against G. trabeum, L. betulina, T. versicolor, and Trichoderma sp. Leaf EO in the vapor phase more effectively inhibited mycelial growth than did wood EO in the vapor phase. The antifungal indices obtained using the poisoned food technique were in the order of wood EO from C. formosensis > wood EO from C. obtusa var. formosana > leaf EO from C. obtusa var. formosana > leaf EO from C. formosensis. The antifungal activities determined using the phytatray chamber assay were in reverse order. This study's findings revealed that in both liquid and vapor phases, EOs obtained from C. formosensis and C. obtusa var. formosana are effective antifungal agents against pathogenic fungi. Further understanding of the mechanisms behind the antifungal activities of EO in different phases would shed additional light on their possible applications.
Experimental
Plant Materials
Mature leaves and wood chips of C. formosensis and C. obtusa var. formosana were obtained from Forestry and Nature Conservation Agency, Pingtung Branch, Luokuei Ranger Station (elevation 1600 m, N22° 59′ 38′, E120° 38′ 52′). Samples were obtained by clipping the foliage from healthy trees of each species, and the leaves were kept frozen at −80 °C until analysis. Each sample was obtained in triplicate. The species were identified by Dr. Lee Chin-Mei from the Taiwan Forest Research Institute (TFRI). A voucher specimen (WYJC-010, 011; LYJC-010, 011) has been deposited at the department of Forest Products Utilization, TFRI, Taiwan.
Fungal Strains
The fungal strains used in this study, namely brown rot fungus G. trabeum (BCRC 31614), white rot fungus T. versicolor (BCRC 35253) and L. betulina (BCRC 35296), and mold Trichoderma sp. (BCRC33561), were obtained from the Bioresource Collection and Research Center of the Food Industry Research and Development Institute. Cultures of each fungus were stored in 20% glycerol at −80 °C.
EO Extraction
The wood chips and fresh leaves of C. formosensis and C. obtusa var. formosana were subjected to hydrodistillation using a modified Clevenger-type apparatus for 6 h. 23 The EO yield (w/w) was calculated according to the dry weight of the plant material, and the means of triplicate experiments were used. The EO extracted was stored in airtight containers until analysis.
Gas Chromatography–Mass Spectrometry Analysis of EOs
Analyses of the volatile constituents were conducted using a Clarus 600 gas chromatograph–mass spectrometer (Turbomass, PerkinElmer, Inc., Waltham, MA, USA) equipped with a DB-5 ms column (J&W Scientific, Folsom, CA, USA). The GC oven temperature program started at 50 °C, increased to 200 °C at 3 °C/min, increased to 250 °C at 5 °C/min, and then was maintained at 250 °C for 5 min. Helium was used as the carrier gas at a flow rate of 1 mL/min. The temperatures of the injection port and transfer line were both 250 °C. The MS parameters are described as follows: Ionization voltage (EI), 70 eV; mass range, 45 to 400 AMU; and ion source temperature, 230 °C. The AIs were calculated for all volatile constituents by using a homologous series of n-alkanes (C9-C22) on the DB-5 ms column. 24 Individual components were identified using the Wiley/NBS Registry of mass spectral database (version 7), the NIST (version 2.0) database, published literature, and several authentic reference compounds.2–4,7,10,12 Compounds were quantified by integrating the peak area of the chromatograms.
Antifungal Assay—Poisoned Food Technique
The fungal toxicity of the EOs was evaluated using the poisoned food technique. Briefly, 800, 400, 200, 100, 50, and 25 μg/mL of leaf and wood EO from C. formosensis and C. obtusa var. formosana were added to sterilized potato dextrose agar in 9-cm Petri dishes containing 15 mL of agar in liquid form. Upon solidification, the agar was inoculated with the mycelia of fungus, and the dishes were then incubated in the dark at 25 ± 1 °C and 70% relative humidity. Dishes containing no EO were used as controls. When the mycelium of fungi reached the edges of the control dishes, the antifungal indices were calculated as follows: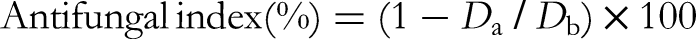
Antifungal Assay—Phytatray Chamber Assay
The effects of volatile EO vapor on fungi were examined using the phytatray chamber assay, as described previously but with slight modifications. 21 Disposable phytatrays (Sigma) with sterilized lids were used as the chamber for the EO and fungi. Potato dextrose agar plates inoculated with the mycelia of test fungi were prepared. Then, 800 µL of leaf and wood EO of the 2 Chamaecyparis species contained in a small vial and a mycelia-inoculated potato dextrose agar plate was placed in a phytatray with a volume of 800 mL. Phytatrays containing no EO were used as controls. All phytatrays with and without EO were incubated in the dark at 25 ± 1 °C and 70% relative humidity for 7 days. After incubation, growth of mycelia was determined through microscopic observation. Fungitoxicity was expressed in terms of percentage of mycelial growth inhibition and calculated according to the aforementioned formula.
Statistical Analyses
All the results were expressed as mean ± standard error (n = 5). The significance of difference among individual means was determined using Scheffe's multiple comparison procedure in SPSS. Results with p < 0.05 were considered to indicate statistically significant differences.
Footnotes
Acknowledgment
The authors gratefully acknowledge Drs HMY, JCS, and Mrs WJK for providing samples and generous help, and all members of the CHEMTREE laboratory for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
