Abstract
Introduction
Atractylodis Rhizoma (AR) is the dried rhizome of Atractylodes lancea (A. lancea) De Candolle and Atractylodes chinensis Koidzumi, which belongs to the Compositae family. AR is a perennial herb; A. chinensis is found primarily in Hebei, Shaanxi, and Jilin Provinces; A. lancea is distributed mainly in Hubei, Zhejiang, and Jiangsu Provinces. 1 AR has been valued as a traditional herbal medicine (THM) in East Asia for centuries.2–4 It is believed to have various bioactivities and is commonly used to treat gastrointestinal and digestive disorders. 5 Recent studies have also demonstrated that the extract from A. lancea exhibits anticancer, hepatoprotective, anti-diabetic, and antiinflammatory properties.3,6–8
THMs are typically formulated using a decoction method in which each herb is boiled in water to extract the various bioactive compounds.9,10 However, unlike most traditional extraction methods, previous studies have mainly focused on studies using various organic solvent extraction methods, and pharmacopeia also requires solvent extraction to be verified. 11 A. lancea is subject to quality control as a sesquiterpenoids-based component when extracted with an organic solvent, but these components are not detected at all when extracted with water. 12 Therefore, there may be problems in prescribing THM because existing studies do not fit THM's efficacy and quality control. So, to establish scientific quality control for THM, studying the active compounds and their bioactivity in water extracts is essential.
There have been reports of the anti-diabetic effects of water extracts of A. lancea, but the specific compounds of the water extract that are responsible for this activity have not been identified, and no studies have analyzed the activity of these components. 13 Therefore, this study aims to establish quality control standards through simultaneous analyses of multicomponents after isolating the active compounds from A. lancea water extract, identifying their structures, and verifying their anti-diabetic effects. This study demonstrates the anti-diabetic effect of water extracts of A. lancea and active compounds using mechanisms related to glycation such as Bovine Serum albumin (BSA)-glucose and BSA-methylglyoxal (MGO) as well as α-glucosidase inhibition. In addition, it contributes to establishing standards for quality control of THM by developing and validating a simultaneous analysis method using an HPLC-photodiode array detector (PDA) for active compounds separated from A. lancea water extract and presenting content standards. This study is the first to research the active components and bioactivity in water extracts of A. lancea, indicating the importance of THM water extract research and its potential as a medicine for diabetes. It is hoped that this study will serve as a basis for evaluating the efficacy of water extract from THM and establishing standards for safe quality control methods.
Results and Discussion
Identification of Isolated Compounds From A. lancea Water Extract
The
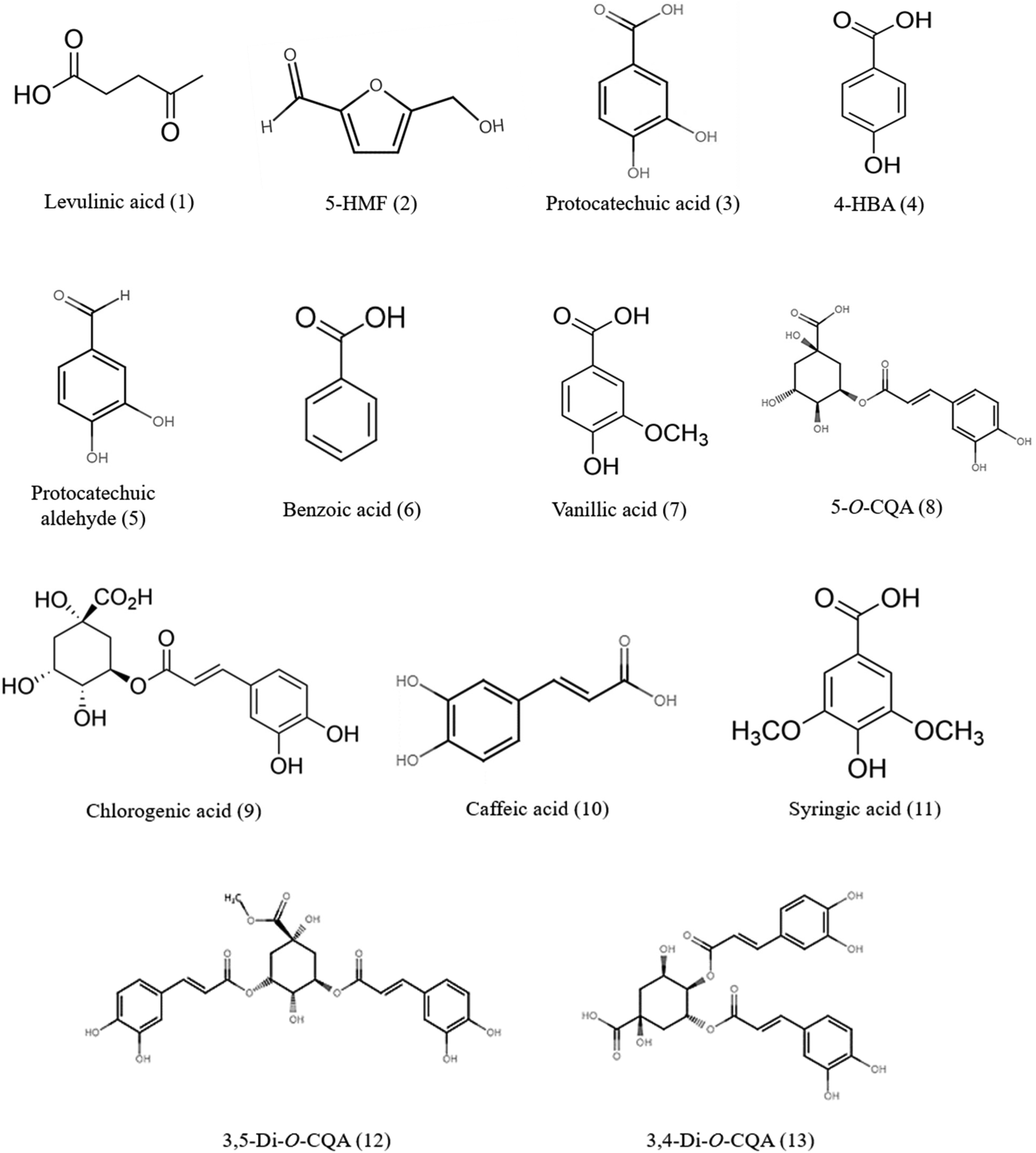
Structures of the 13 identified compounds.
Identification of Compounds

Inhibitory Activity of the Anti-diabetic
AGEs formation inhibitory activity of extracts and fractions of A. lancea
The formation of advanced glycation end-products (AGEs) and the excessive activity of α-glucosidase enzyme are two key factors implicated in diabetic complications. 27 AGEs are harmful compounds formed through nonenzymatic reactions between reducing sugars and proteins. 28 Elevated levels of AGEs are associated with diabetic complications. 29 To demonstrate the inhibitory effects of the extract and fractions of A. lancea, the formation of AGEs was assessed using BSA-glucose and BSA-MGO systems. The n-BuOH fraction exhibited significant inhibition of AGE formation (IC50 of 299.23 ± 1.02 µg/mL, 168.01 ± 1.47 µg/mL). The water extracts and water fractions also demonstrated inhibitory effects in BSA-glucose (IC50 of 502.11 ± 3.10 µg/mL, 414.47 ± 41.51 µg/mL) and BSA-MGO (IC50 of 389.11 ± 2.62, 625.23 ± 8.31 µg/mL), respectively. The EtOAc fraction was only minimally effective (IC50 of 741.13 ± 2.41 µg/mL, 604.44 ± 32.42 µg/mL) while the DCM fraction also showed little to no inhibition. Upon comparing the activities of the polar (water) and nonpolar (n-Hx) extracts in the AGE systems, we observed that the water extract exhibited significantly higher inhibitory values compared with the n-Hx extract. In the BSA-glucose system, the water extract had an IC50 value of 502.11 ± 3.10 µg/mL. Similarly, in the BSA-MGO system, the water had an IC50 value of 389.11 ± 2.62 µg/mL, whereas the n-Hx extract did not inhibit either AGE assay (Table 2).
IC50 of the Atractylodes lancea Extracts and Fractions Against AGEs Formation and α-Glucosidase.

Abbreviation: AGEs: advanced glycation end-products.
Data are presented as mean ± standard deviation (SD) (n = 3).
IC50 values were determined by fitting a least-squares regression line to the logarithmic concentrations plotted against the residual activity.
ND was not detectable.
Aminoguanidine (AG) was used as a positive control of advanced glycation end-products formation inhibitory activity.
Acarbose was used as a positive control of α-glucosidase inhibitory activity.
*Indicates a significant difference from control; *P < 0.05, **P < 0.01, ***P < 0.001; -: not measured.
α-glucosidase inhibitory activity of extracts and fractions of A. lancea
α-glucosidase is an enzyme that breaks down complex carbohydrates into absorbable simple sugars. 30 Inhibition of α-glucosidase activity can delay the absorption of glucose, thus managing postprandial hyperglycemia. 31 When the inhibitory activity was examined, the n-BuOH fraction demonstrated the most potent α-glucosidase inhibitory activity (IC50 of 187.11 ± 1.01 µg/mL). The water fraction and water extract also showed effective inhibitory activity (IC50 of 322.10 ± 5.01, 379.04 ± 2.14 µg/mL, respectively), while the EtOAc fraction showed slight inhibitory activity (IC50 of 599.02 ± 4.33 µg/mL). The inhibitory values of the water extract were much higher than those of the n-Hx extract (IC50 of 379.04 ± 2.14 and >1000 µg/mL) (Table 2), demonstrating that the water extracts and fractions of A. lancea exhibited promising anti-diabetic potential compared with the nonpolar extracts.
AGEs formation inhibition of isolated compounds
The inhibitory activity of the 13 compounds against the formation of AGEs was assessed in both the BSA-glucose and BSA-MGO systems. The generally high inhibitory activity group in AGEs was the chlorogenic acid-derived group of phenolic acid. Compounds
IC50 of Compounds Against AGEs Formation and α-Glucosidase.

Abbreviation: AGEs: advanced glycation end-products.
Data are presented as mean ± SD. (n = 3).
IC50 values were determined by fitting a least-squares regression line to the logarithmic concentrations plotted against the residual activity.
ND was not detectable.
Aminoguanidine was used as a positive control of advanced glycation end-products formation inhibitory activity.
Acarbose was used as a positive control of α-glucosidase inhibitory activity.
*Indicates a significant difference from control, *P < 0.05, **P < 0.01, ***P < 0.001; –: not measured.
α-glucosidase inhibitory activity of isolated compounds
Among the tested compounds, Compound 13 demonstrated the most potent inhibitory activity against α-glucosidase (31.66 ± 9.33 µg/mL) while Compounds
Among the 13 compounds isolated from A. lancea, nine (
Simultaneous Quantification HPLC Analysis
Validation of HPLC analysis
A simultaneous analysis method was developed for the specificity quantitative of nine (

HPLC chromatograms of standards mixture (1) and Atractylodes lancea water extract. (A) compound
Five different concentrations were employed to evaluate the linearity of the nine compounds. All calibration curves exhibited excellent linearity, with correlation coefficients (r2) of 0.999 or higher for all compounds. The limit of detection of the nine compounds ranged from 0.32 to 2.51 μg/mL and the limit of quantitation was 0.97-7.61 μg/mL (Table 4). The precision and accuracy of the analysis method were measured intraday and interday by testing nine standard mixture solutions. The intraday precision was measured within a day and repeated on three different days. Compounds
Calibration Curves and Linear Range of Compounds.

Each measurement value was calculated as the mean of three replicates.
Y = peak area, X = concentration of standard (μg/mL).
r2 = correlation coefficient for five final concentrations in the calibration curve. Compound
Intraday and Interday Precision and Accuracy of Compounds.

Each measurement value was calculated as the mean of three replicates. Compound
Quantitative HPLC analysis
The contents of nine compounds were calculated using the calibration equation derived from the calibration curve. The content percentage of each compound in the extract was determined based on the extraction solvent volume (A) in mL and the sample powder weight (B) in μg. The analysis was performed in triplicate, and the mean contents of Compounds
Contents (%) of Compounds From Atractylodes lancea (n = 3).
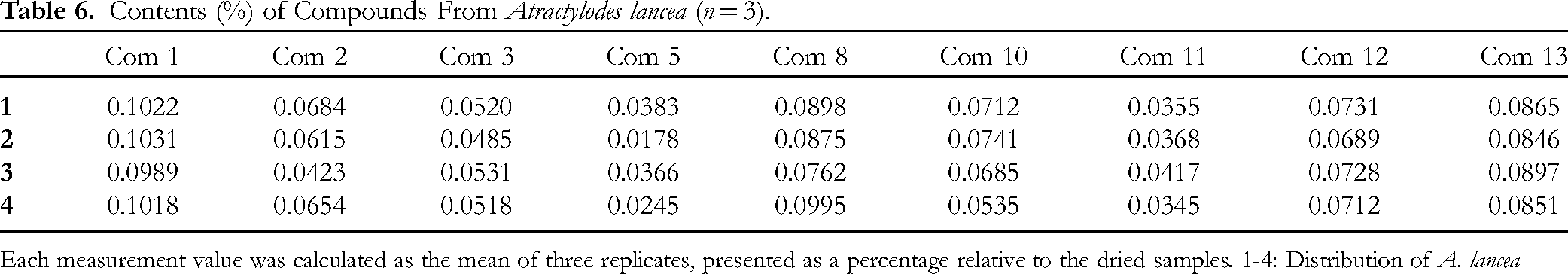
Each measurement value was calculated as the mean of three replicates, presented as a percentage relative to the dried samples. 1-4: Distribution of A. lancea
Materials and Methods
Plant Materials
A. lancea samples (Figure 3) were purchased from the Kyung-dong Market, a medicinal herbal market. The samples were authenticated by Professor Wan Kyunn Whang of the College of Pharmacy, Chung-Ang University, Seoul, South Korea.

Rhizome of Atractylodes lancea De Candolle.
Instruments and Reagents
All samples were weighed by an ML 204 (Mettler Toledo, Switzerland), extracted by a Sonicator Power Sonic 420 (Hwashin Tech CO., Ltd), and evaporated using an EYELA SB-1200 (Shanghai Eyela CO., Ltd). The fractions and compounds were isolated by open column consisting of Diaion® HP-20 (Supelco Inc.), Sephadex LH-20 (25-100 μm; Pharmacia), MCI CHP 20P (Supelco), and Octadecyl-Silica (ODS) gel (400-500 mesh; Waters). The NMR spectra were recorded at 600 and 150 MHz by JEOL ECZ600R spectrometer (JEOL), respectively. The NMR spectra were measured in MeOH-d4 (CD3OD), dimethyl sulfoxide-d6 (DMSO-d6), and chloroform-d (CDCl3; Sigma-Aldrich Co.). Mass spectrometry (MS) was performed using UHPLC-ESI/LTQ-orbitrap-HRMS with an Ultimate 3000 rapid separation LC system (Thermo). The Hypersil GOLD™ SAX LC Column (4.6 × 150 mm, 5 µm) was used for analytical MS (Thermo Fisher Scientific). Thin layer chromatography (TLC) was performed utilizing silica gel, and Kiesel gel 60 F254 (Merck Co.), and spots were observed under ultraviolet (UV) lighting (Vilber Lourmat). HPLC-PDA was analyzed using Empower Pro 2.0 software, a Waters 2695 system pump with a Waters 2489 PDA (Waters), and the Capcell Pak C18 column (4.6 × 250 mm, 5 µm, OSACA SODA CO., LTD). All HPLC-grade solvents, such as acetonitrile, distilled water, formic acid, and acetic acid were supplied by Thermo Fisher Scientific (Waltham) and were filtered through a 0.45 µm polytetrafluoroethylene (PTFE) or a polyvinylidene fluoride (PVDF) filter.
The antidiabetes activity assays, including the BSA-glucose model inhibitory activity assay and BSA-MGO model inhibitory activity assay, were performed using the Infinite® F200 PRO spectrophotometer (TECAN), while the α-glucosidase inhibitory activity assay was performed using Biotek EPOCH2 (Biotek). The solvents used in the activity assays, including sodium phosphate monobasic, sodium phosphate dibasic, sodium azide, Aminoguanidine hydrochloride (AG), Bovine Serum Albumin (BSA), methylglyoxal (MGO), Dimethyl sulfoxide (DMSO), D-(+)-glucose, D-(−)-fructose, acarbose, p-nitrophenyl-α-D-glucopyranoside (p-NPG), and α-glucosidase from Saccharomyces cerevisiae (Saccharomycetaceae) were all purchased from Sigma-Aldrich, Co. (Sigma-Aldrich Co.). The program used for data statistics was SPSS Statistics 20.0 (SPSS Statistics 20.0, IBM).
Extract, Fraction, and Isolation From A. lancea
A. lancea (3.0 kg) was extracted with water (2 L for 60 min, three times) at room temperature and the resulting solution was then filtered. The extract was condensed under lowered pressure using an evaporator to yield 414.8 g of crude extract. The extract was suspended in water and sequentially partitioned with DCM, EtOAc, and n-BuOH (2 L, three times, each) and water fractions. Each fraction yield was 11.24 g, 35.97 g, and 61.2 g, 87.81 g. The n-BuOH fraction (61.2 g) was applied to the Diaion® HP-20 eluting with the MeOH and the water gradient system (Water, 50%, 100% MeOH) to give three fractions (F.1–F.3). Fraction 1 (19.3 g) was subjected to column chromatography on Sephadex LH-20 gel eluted with methanol-water (20:80 to 90:10) to obtain two fractions (F.1-1 and F.1-2). F.1-2 (7.3 g) was subjected to column chromatography on MCI with MeOH-water (10:90 to 80:20) to obtain three fractions (F.1-2-1–F.1-2-3). F.1-2-3 (1.1 g) was subjected to column chromatography on ODS gel with 20% and 50% MeOH to obtain Compound
F.2-1-1 (0.8 g) was subjected to column chromatography on ODS gel with 20% and 60% MeOH giving Compound
Identification of Compounds Isolated From A. lancea Water Extract
1H and 13C-NMR spectra
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
Compound
UHPLC-ESI/LTQ-orbitrap-HRMS conditions
The molecular weights of the 13 isolated compounds were determined using UHPLC-ESI/LTQ-Orbitrap-HRMS. To do this, 1.0 mg of each isolated compound was dissolved in 100 mL MeOH and filtered through a 0.45 µm PVDF membrane filter. The analysis was performed using a Hypersil GOLD column (C18, 2.1 × 50 mm, 1.9 μm). The mobile phase was carried out as a gradient with solvent A (water, 0.1% formic acid, v/v) and solvent B (acetonitrile, 0.1% formic acid, v/v). Solvent B concentrations were increased from 5% to 50% for 18 min and then from 50% to 100% for 2 min. After the 100% concentration was maintained for 3 min, the concentration was reduced from 100% to 5% and maintained for 7 min. The mobile phase was delivered at a flow rate of 0.3 mL/min, and an injection volume was 20 µL. The column temperature was kept constant at 30 °C. The optimal analysis conditions were as follows: the heater and capillary temperature, 300 °C and 360 °C, respectively; S-lens RF level, 50.0 V; auxiliary and sheath gas flow rates, 10 and 45 L/h, respectively; spray capillary voltage, 3.0 kV, overall MS AGC target and MS resolution, 3e6 and 35 000.
HPLC Analysis
Samples preparation
The dried powders (2 g) were extracted with water (50 mL) by ultrasonic bath for 60 min. Next, the samples were filtered through filter paper, and the extracts were evaporated in vacuo and dissolved in MeOH (2 mL) and were then filtered through a 0.45 µm PVDF membrane filter before injection.
Analytical conditions
A simultaneous quantitative analysis method for the nine compounds separated from the water extract of A. lancea was developed using HPLC-PDA with UV detection at 210-400 nm. The gradient mobile phase consisted of 0.1% formic acid in water (Solvent A) and 0.1% formic acid in acetonitrile (Solvent B). The linear gradient mobile phase was performed with the following elution program: 3-8% solvent B at 0-9 min; 8-20% solvent B at 9-30 min; 20-29% solvent B at 30-34 min; 29-34% solvent B at 34-45 min. The mobile phase was delivered at a flow rate of 1.0 mL/min, and an injection volume was 20 µL. The column temperature was kept constant at 25 °C. The maximum UV absorption of these compounds ranged from 215.5 nm to 327.7 nm, and the chromatograms were recorded at an optimized wavelength of 310 nm.
Bioactivity Assay
Anti-glycation activity
The anti-glycation activity was determined by the BSA-glucose and BSA-MGO model systems. The AGEs formation inhibitory assay was conducted using a spectrophotometric method developed in a previous study.32,33
BSA-glucose model inhibitory activity assay
The extracts, fractions, 13 compounds, and positive control were dissolved in 10% DMSO at varying concentrations, with the extracts and fractions ranging from 1000 to 10 000 μg/mL, and the compounds ranging from 100 to 10 000 μM. AG was employed as a positive control in the study. A mixture was prepared to consist of a 50 mM phosphate buffer (pH 7.4) containing 0.02% sodium azide, BSA at a concentration of 10 mg/mL, and a solution of 0.4 M fructose and glucose. This mixture, along with the sample or buffer solution, was then incubated at a temperature of 60 °C for a duration of 2 days. Following the incubation period, the fluorescence was measured using a 96-well black plate, with excitation wavelengths set at 350 nm and emission wavelengths set at 450 nm. The inhibitory activity against AGEs formation in the BSA-glucose system was determined using the formula {(Ac − As)/Ac} × 100, where Ac represents the fluorescence of the control and As represents the fluorescence of the sample. The half maximum inhibitory concentration (IC50) value, which is the concentration that inhibits 50% of the AGEs formation, was calculated based on three repeated measurements and expressed as the mean ± SD.
BSA-MGO model inhibitory activity assay
All the samples were added after being dissolved in 10% DMSO solution. The assay mixture consisted of a 50 mM phosphate buffer (pH 7.4) containing 0.02% sodium azide and BSA (10 mg/mL). To this mixture, 7 mM MGO and the samples were added. After incubating the mixture at a temperature of 60 °C for a duration of 2 days, the fluorescence was measured using a spectrofluorometer with an excitation wavelength of 340 nm and an emission wavelength of 420 nm in a 96-black well plate. Each sample was measured in triplicate and the AGE formation in BSA-MGO system inhibition of the extracts and compounds was calculated using AG as a positive control. The control samples were prepared using the identical mixture, with the exception that the sample solution was substituted with phosphate buffer. The inhibitory activity was calculated by comparing the fluorescence of the samples to that of the positive control, using the same approach as in the BSA-glucose assay.
Inhibitory activity against α-glucosidase
The α-glucosidase inhibitory activity of the samples was evaluated using a modified method based on the one described in a previous study. 34 In brief, the sample dissolved in 10% DMSO was mixed with 100 mM phosphate buffer (pH 6.8) and 3 mM p-NPG solution. The reaction mixture was taken and added to a 96-well plate. In each well, a 10 mM phosphate buffer solution (pH 6.8) containing 0.3 U/mL of α-glucosidase was added, and the reaction proceeded at 37 °C for 15 min. After incubating, the reaction absorbance was measured at 405 nm immediately. The acarbose was used as the positive control. The control samples were prepared using the identical mixture, with the exception that the sample solution was substituted with phosphate buffer. The inhibitory activity was measured at five different concentrations and was determined using the formula {(Ac − As)/Ac} × 100, where As and Ac represent the absorbance values of the sample and control, respectively. The IC50 value was calculated from three repeated measurements and expressed as mean ± standard deviation (SD).
Statistical Analysis
Statistical analysis was conducted using one-way analysis of variance and t-tests to determine the significance of differences in the results. The significance level was set at P < 0.05, P < 0.01, and P < 0.001. The experiments were repeated three times, and the data were expressed as the mean ± SD.
Conclusions
To our knowledge, this study was the first to conduct bioassay-guided isolation from water extract of A. lancea, and to develop simultaneous quantitative HPLC analysis for quality control standards for THM. The A. lancea water extract and fractions were evaluated for anti-diabetic activity, with the n-BuOH fraction demonstrating a greater inhibitory effect than other fractions. Thirteen compounds, including phenolic acids and carboxylic acids, were isolated from the n-BuOH fraction by bioassay-guided isolation. The compounds were identified by 1H, 13C-NMR, and ESI/LTQ-Orbitrap-HRMS. AGEs formation and α-glucosidase inhibitory activity were confirmed for Compounds
In summary, this study was conducted using A. lancea water extract, which is widely used as herbal medicine, as a quality control for THM. The anti-diabetic inhibitory active compounds were isolated from the water extract and compared with the activity of the nonpolar solvent extract, with the results showing that the water extract had a higher activity. Therefore, this study suggested that water extract can be important for the quality control of THM, and also proved that water extract of A. lancea can be developed as a treatment for diabetes and its associated complications. In addition, this study makes a meaningful contribution to the literature by preparing the basis for quality control of THMs by developing a simultaneous analysis method and suggesting the content of each bioactivity compound of A. lancea.
Footnotes
Acknowledgments
This research was supported by a grant (21173MFDS561) from the Ministry of Food and Drug Safety in 2021, and the Chung-Ang University Research Scholarship Grants in 2021.
Author's Contributions
Y.J.J. and W.K.W. conceived and designed the experiments. Y.J.J. and S.J.L contributed equally to performing the extraction, isolation, bioactivity assay, and quantitative analysis. In addition, they analyzed the data and wrote the paper. All authors have read and agreed to the published version of the manuscript.
Availability of Data and Material
The data sets presented in this study are included in the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
