Abstract
In recent years, essential oils (EOs) have gained popularity for their potential as natural antimicrobial agents and mosquito larvae control. This study aimed to explore the chemical composition of EOs extracted from the leaves and rhizomes of Distichochlamys benenica Q.B. Nguyen & Škorničk, and to evaluate their mosquito larvicidal and antimicrobial activities. The EOs were obtained using hydrodistillation and subjected to gas chromatography (GC) and GC-mass spectrometry to determine their chemical constituents. The main components identified in the leaf EO were β-pinene (47.7%), cis-β-elemene (8.5%), and α-pinene (7.8%), while the rhizome EO mainly consisted of geranyl acetate (25.7%), geranial (23.3%), neral (16.6%), and geraniol (8.4%). The mosquito larvicidal activity of the EOs was evaluated using larvae of Aedes aegypti and Culex quinquefasciatus during 24 and 48 h of treatment. The results demonstrated that these EOs effectively inhibited the mosquito larvae, with LC50 values ranging from 31.15 to 71.53 μg/mL and LC90 values ranging from 46.28 to 148.66 μg/mL. Additionally, the minimum inhibitory concentration (MIC) and half-maximal inhibitory concentration (IC50) values were used to evaluate the antimicrobial efficacy of EOs. Both EOs exhibited potent antimicrobial effects against Enterococcus faecalis ATCC 299212, Staphylococcus aureus ATCC 25923, Bacillus cereus ATCC 14579, Pseudomonas aeruginosa ATCC 27853, and Candida albicans ATCC 10231, with MIC values ranging from 16 to 64 μg/mL and IC50 values ranging from 4.67 to 45.67 μg/mL. Overall, the mosquito larvicidal and antimicrobial activities of D. benenica EOs highlight their potential as natural alternatives for combating mosquito-borne diseases and microbial infections.
Introduction
The family Zingiberaceae, commonly known as the ginger family, comprises approximately 50 genera and over 1600 known species. 1 Zingiberaceae species are predominantly found in tropical and subtropical regions around the world, with a significant presence in Southeast Asia, India, China, and Africa. 2 From an ethnobotanical perspective, Zingiberaceae plants have been valued for their culinary, medicinal, and cultural uses. Many members of this family are renowned for their aromatic rhizomes, which are extensively used as spices and flavorings in various cuisines.3–5 Apart from their culinary applications, Zingiberaceae species have also been extensively used in traditional medicine systems across different cultures. In traditional Vietnamese medicine, Zingiberaceae species have been employed for treating digestive disorders, relieving pain and inflammation, and boosting immune function. 5 In recent years, scientific research has focused on exploring the phytochemical composition and pharmacological potential of Zingiberaceae species. Studies have identified several bioactive compounds, including gingerols, curcuminoids, and zingiberene, which contribute to the medicinal properties of these species.6–8
Essential oils (EOs) have gained significant attention in recent years due to their diverse therapeutic properties, including mosquito larvicidal and antimicrobial activities.9–15 Mosquitoes are known vectors for diseases such as malaria, dengue fever, Zika virus, and West Nile virus, causing significant morbidity and mortality worldwide. 16 EOs have shown promise as effective mosquito larvicides, offering an eco-friendly approach to controlling mosquito populations and preventing the spread of mosquito-borne diseases. 9 Furthermore, they have the potential to address the issue of microbial resistance, which has become a significant concern associated with conventional antimicrobial agents. 17 Studies suggest that the complex chemical composition of EOs makes it difficult for microorganisms to develop mechanisms of resistance, thereby reducing the likelihood of resistance formation.18,19 Among these EOs, those extracted from the Zingiberaceae family have gained significant attention due to their potential applications in the fields of vector control and medicine.20–23
Distichochlamys is an endemic Vietnamese genus belonging to the family Zingiberaceae. 24 Four species have been identified since the genus was established in 1995 by M.F. Newman: D. citrea M.F. Newman, 24 D. benenica Q.B. Nguyen & Škorničk, 25 D. orlowii K. Larsen & M.F. Newman, 26 and D. rubrostriata W.J. Kress & Rehse. 27 D. benenica was the last of these species to be described following its discovery in 2012 in Ben En National Park, Vietnam. 25 Several recent studies have focused on the phytochemical composition and bioactive properties of Distichochlamys species.28–31 Additionally, investigations have been conducted into the chemical composition and biological activity of EOs derived from the leaves and rhizomes of D. citrea, D. orlowii, and D. benenica.32–37 Notably, a study by Hoang et al 32 demonstrated the acetylcholinesterase activity of D. benenica rhizome EO collected from Quang Nam, Vietnam. Furthermore, variations in chemical composition were observed in the EOs of D. citrea leaves and rhizomes collected from different regions of Vietnam, including Quang Binh, Quang Tri, Thua Thien Hue, and Quang Nam.35–37 However, there is currently no information available regarding the chemical composition of the leaf EO of D. benenica. Moreover, the mosquito larvicidal and antimicrobial properties of the EOs derived from the leaves and rhizomes of D. benenica have not been investigated. Given this knowledge gap, we conducted the present study to report on the chemical constituents, mosquito larvicidal activity, and antimicrobial properties of the EOs obtained from the leaves and rhizomes of D. benenica collected from Ben En National Park, Vietnam.
Results and Discussion
Chemical Constituents of EOs
Hydrodistillation was performed on the leaves and rhizomes of D. benenica using a Clevenger-type apparatus, resulting in the production of light-yellow oils. The yields of EOs obtained from the leaves and rhizomes of D. benenica were 0.22% and 0.35% w/w, respectively, based on the dried weight of the materials. Table 1 presents the compositions of the EOs, with the constituents listed according to their elution from a HP-5MS column.
Chemical Constituents of EOs From the Leaves and Rhizomes of Distichochlamys benenica.

aElution order on HP-5MS column.
bRetention index on HP-5MS column.
dIdentification: RI, coherence of the temperature-programmed retention index with respect to those reported by Adams 38 ; MS, matching with mass libraries.38,39
eNot identified.
In the present study, we identified 47 compounds in the leaf EO, accounting for 88.5% of the total compounds detected, and 28 compounds in the rhizome EO, accounting for 97.8% of the total compounds detected (Table 1). The predominant chemical class of the EOs from the leaves and rhizomes of D. benenica was that of monoterpenes. However, the leaf EO was primarily composed of monoterpene hydrocarbons, comprising 60.5% of the composition, whereas the rhizome EO was dominated by oxygenated monoterpenes, accounting for 85.4% of the composition. The most abundant component in the leaf EO was β-pinene, with a concentration of 47.7%. Additionally, cis-β-elemene and α-pinene were also present in significant quantities in the leaf EO, representing 8.5% and 7.8% of the total chemical composition, respectively. In the rhizome EO, geranyl acetate was the main compound, constituting 25.7% of the chemical composition, followed by geranial (23.3%), neral (16.6%), and geraniol (8.4%). However, these compounds were either absent or found in lower amounts in the leaf EO. Our findings demonstrated that not only the quantity but also the qualitative composition of the EOs obtained from the leaves and rhizomes of D. benenica differed.
It is important to mention that there has only been one previous study examining the composition of an EO derived from D. benenica rhizomes. Hoang et al 32 reported the major components of the EO collected from D. benenica rhizomes in Quang Nam, Vietnam, which included 1,8-cineole (54.39% of the total chemical composition), β-pinene (7.50%), geranial (7.26%), and neral (6.79%). Interestingly, the present study yielded different results compared to those reported by Hoang et al. 32 Specifically, we found geranyl acetate to be the most abundant component in the rhizome EO, whereas it was not detected in the rhizome EO studied by Hoang et al. 30 Moreover, the concentration of 1,8-cineole was higher in Hoang et al's study of the rhizome EO than in our study. 32 These differences may be attributed to variations in the plant organs used, as well as geographical and environmental factors.40,41 Furthermore, our study represents the first report on the components of D. benenica leaf EO.
Although there is limited specific information available about D. benenica, numerous studies have been conducted on the EO compositions of other Distichochlamys species (Table 2). Previous studies have shown that terpene compounds make up the majority of EOs in Distichochlamys species, which aligns with the composition of the EOs obtained from the leaves and rhizomes of D. benenica in this study. However, the main compounds vary between species. For instance, a previous investigation revealed that the primary constituents of the rhizome EO of D. orlowii were geranyl acetate (16.5%), β-elemene (9.2%), β-pinene (9.0%), and β-caryophyllene (7.9%), whereas 1,8-cineole (23.0%) was the most abundant compound in the rhizome EO of D. citrea from Nghe An. 34 Furthermore, EOs derived from the leaves and rhizomes of D. citrea collected from different geographical locations have shown notable variations in their chemical composition.35–37 These findings indicate that the chemical composition of EOs in Distichochlamys is primarily influenced by the specific species under investigation, as well as factors such as the plant organ used, environmental conditions, geographic variations, and EO extraction methods.33,41,42
Major Components (> 5%) Identified From Distichochlamys EOs.

Abbreviations: HD, hydrodistillation; SD, steam distillation; MAHD, microwave-assisted hydrodistillation.
Mosquito Larvicidal Potential of EOs
The mosquito larvicidal potential of the EOs extracted from D. benenica leaves and rhizomes was assessed. The results, presented in Tables 3 and 4, clearly indicate distinct larvicidal activity of the leaf and rhizome EOs against Aedes aegypti and Culex quinquefasciatus larvae. For A. aegypti, the rhizome EO showed higher activity against the larvae than the leaf EO, with LC50 and LC90 values of 32.78 and 49.96 µg/mL after a 24-h exposure, respectively. The corresponding values after a 48-h exposure were 31.46 and 46.28 µg/mL, respectively. On the other hand, the leaf EO was more effective against C. quinquefasciatus than the rhizome EO, with LC50 values after a 24 and 48-h exposure of 39.01 and 31.15 µg/mL, respectively. The corresponding LC90 values were 67.36 and 69.01 µg/mL, respectively. It is worth noting that previous studies have indicated that EOs exhibit promising larvicidal activity when their LC50 values are below 100 µg/mL.43,44 Therefore, based on this criterion, we can conclude that the EOs derived from the leaves and rhizomes of D. benenica in our study demonstrated significant larvicidal potential against A. aegypti and C. quinquefasciatus larvae.
Mosquito Larvicidal Potential of EOs From the Leaves and Rhizomes of Distichochlamys benenica Against Aedes aegypti Larvae.

aConfidence interval at 95%.
Mosquito Larvicidal Potential of EOs From the Leaves and Rhizomes of Distichochlamys benenica Against Culex quinquefasciatus Larvae.

aConfidence interval at 95%.
The mosquito larvicidal potential of EOs from leaves and rhizomes of D. benenica may be due to the presence of major components such as β-pinene, α-pinene, geranyl acetate, geranial, neral, and geraniol. EOs containing these components have been previously studied, and showed good larvicidal activity.44–48 β-Pinene, for instance, exhibited larvicidal activity against A. aegypti, with LC50 values ranging from 12.1 to 42.5 μg/mL, and C. quinquefasciatus, with an LC50 value of 19.6 μg/mL.44,45 Similarly, α-pinene has been shown to possess larvicidal properties against both A. aegypti and C. quinquefasciatus.44,45 Geraniol and geranyl acetate displayed larvicidal activity against A. aegypti,46,47 while geranial and geraniol exhibited larvicidal effects against C. quinquefasciatus. 45 Cymbopogon flexuosus EO, containing geranial, neral, geranyl acetate, and geraniol as its primary components, has demonstrated significant activity against A. aegypti. 48 In addition to the main components, minor compounds in EOs may also contribute to their larvicidal properties. 49 The synergistic effect between the major and minor components in EOs provides a multifaceted approach to combating mosquito larvae. Despite this, some specific larvicidal mechanisms have been proposed; for example, it is assumed that the volatile compounds in EOs can penetrate the respiratory system of the larvae and interfere with their oxygen uptake.44,50 Furthermore, they can also interfere with neurotransmitters, ion channels, and enzymes that are vital for the proper functioning of mosquito larvae.44,50 As a result, EOs can disrupt the larvae's physiological processes, including feeding, locomotion, and development, resulting in larval mortality and preventing the emergence of adult mosquitoes.
Antimicrobial Potential of EOs
The antimicrobial effects of EOs derived from D. benenica leaves and rhizomes were assessed against 7 test microorganisms, comprising 3 Gram-positive bacteria, 3 Gram-negative bacteria, and 1 yeast. The potency of these EOs was quantitatively evaluated using minimum inhibitory concentration (MIC) and half-maximal inhibitory concentration (IC50) values, as presented in Table 5. The results indicated that the EOs from the leaves and rhizomes demonstrated activity against all 3 Gram-positive bacteria (Enterococcus faecalis, Staphylococcus aureus, and Bacillus cereus) and 1 yeast (Candida albicans), with MIC values ranging from 16 to 64 μg/mL, while the IC50 values ranged from 4.67 to 23.45 μg/mL. Additionally, both EOs showed activity against Pseudomonas aeruginosa, with a MIC value of 64 μg/mL. The leaf EO had an IC50 of 45.67 μg/mL, while the rhizome EO had an IC50 of 26.56 μg/mL. However, these EOs did not exhibit activity against the Gram-negative bacteria Escherichia coli and Salmonella enterica. It is worth noting that the antimicrobial activity of both EOs was comparable to that of the positive controls. These findings are consistent with a study conducted by Thinh et al, 33 which reported the antimicrobial activity of EOs from D. citrea against various microorganisms. Notably, the MIC values of D. benenica EOs were lower than those of D. citrea EOs, indicating the more potent antimicrobial activity of D. benenica EOs. Previous research has demonstrated that EOs exhibit strong antimicrobial effects when their MIC values are below 100 μg/mL.51,52 According to this criterion, the EOs derived from the leaves and rhizomes of D. benenica in our study exhibited significant antimicrobial activity.
Antimicrobial Potential of EOs From the Leaves and Rhizomes of Distichochlamys benenica.
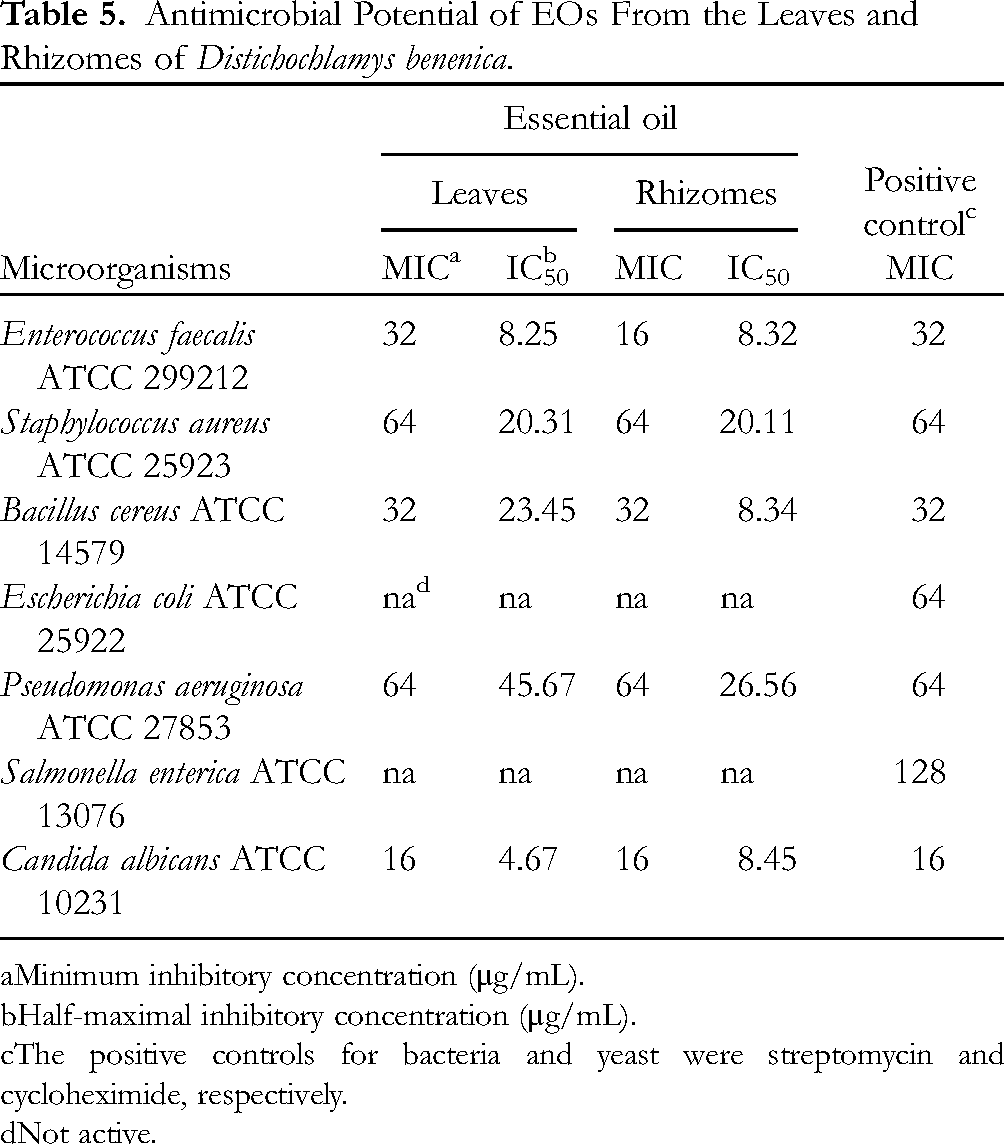
aMinimum inhibitory concentration (μg/mL).
bHalf-maximal inhibitory concentration (μg/mL).
cThe positive controls for bacteria and yeast were streptomycin and cycloheximide, respectively.
dNot active.
The primary reason behind the antimicrobial properties of D. benenica EOs seems to be the presence of active components, particularly monoterpenes and their derivatives.53,54 Notable compounds like β-pinene, cis-β-elemene, α-pinene, geranyl acetate, geranial, neral, and geraniol were identified as major constituents of the D. benenica EOs, and are well-known for their antimicrobial properties.54–56 Indeed, β-pinene and α-pinene, 2 monoterpenes, have exhibited antimicrobial activity against bacterial and fungal strains, including drug-resistant ones. 57 EO from Syzygium szemaoense, primarily consisting of cis-β-elemene, has demonstrated noteworthy antimicrobial properties against bacteria and fungi. 58 Many EOs contain geranial, neral, geraniol, and geranyl acetate as their primary components, which have shown antimicrobial activity against a broad range of bacteria, including both Gram-positive and Gram-negative strains.59,60 These compounds commonly exert their antimicrobial properties through mechanisms including disruption of cell membranes, inhibition of enzymatic processes, interference with protein synthesis, and disruption of essential microbial metabolic pathways. 61 Furthermore, the synergistic interaction of both main and minor components is frequently cited as the cause of the antimicrobial activity of EOs. 62 As a result, minor components such as 1,8-cineole, linalool, and β-caryophyllene may also contribute to the antimicrobial properties of EOs. Overall, the antimicrobial activity of EOs is a complex phenomenon that involves a variety of chemical and biological interactions with microbes. Additionally, compared to Gram-negative bacteria, the EOs from D. benenica were more effective against Gram-positive bacteria. This disparity in efficacy is thought to be due to the different cell wall structures of Gram-positive and Gram-negative bacteria. 61 Gram-positive bacteria have a thick coating of peptidoglycan on their cell walls, which makes it easier for EOs to enter the cell and rupture the membrane. 61 On the other hand, Gram-negative bacteria have an outer membrane made of lipopolysaccharides and a thinner peptidoglycan layer, acting as a barrier that prevents EOs from entering and interfering with the cell membrane. 63
Conclusions
In summary, we conducted an investigation into the chemical composition of EOs derived from the leaves and rhizomes of D. benenica collected from Ben En National Park, Vietnam, as well as their mosquito larvicidal and antimicrobial activities. The main components of the leaf EO, as determined by gas chromatography (GC) and gas chromatography-mass spectrometry (GC-MS) analysis, were β-pinene (47.7%), cis-β-elemene (8.5%), and α-pinene (7.8%). Meanwhile, the rhizome EO contained geranyl acetate (25.7%), geranial (23.3%), neral (16.6%), and geraniol (8.4%). Furthermore, our study yielded promising results with regard to the mosquito larvicidal and antimicrobial activities of D. benenica EOs, which was significant. This activity highlights the EOs’ potential as natural alternatives for the management of mosquito-borne diseases and microbial infections. However, further research is needed to explore the mechanisms of action, optimize formulations, and assess the safety and long-term effects of these EOs for potential vector control and therapeutic applications.
Materials and Methods
Plant Material and EO Extraction
Fresh leaves and rhizomes of D. benenica were collected from Ben En National Park, Thanh Hoa, Vietnam, in December 2020. The collection site was located at 19°36'24.66"N and 105°23'42.80"E, with a relative humidity of 62.1% and a temperature of 27.4 °C. The plant sample was authenticated by Assoc. Prof. Dr Le Thi Huong from Vinh University (Nghe An, Vietnam) and subsequently assigned a registration number in the herbarium with the voucher code DND 880. The leaves and rhizomes were chopped into small pieces and air-dried at room temperature (25 °C) for a week before the EO was extracted. Each sample was then separately hydrodistilled for 4 h at normal pressure using a Clevenger-type apparatus, as previously reported.64,65 The EOs obtained were dried using anhydrous Na2SO4 and carefully stored in tightly closed dark vials at a temperature of 4 °C until it was ready for analysis.
Analysis of EOs
According to the previously stated procedure, 66 GC and GC-MS were used to examine the EOs extracted from the leaves and rhizomes of D. benenica. A gas chromatograph of Agilent Technologies, model number HP 7890A Plus, was utilized for the GC analyses. It was connected to a flame ionization detector and an HP-5MS capillary column measuring 30 m in length, with an internal diameter of 0.25 mm and a film thickness of 0.25 µm. The GC oven temperature program was designed to start at 60 °C for 2 min before rising to 220 °C at a rate of 4 °C/min. The injector and detector were kept at temperatures of 250 °C and 260 °C, respectively. Helium was employed as the carrier gas, flowing at a rate of 1.0 mL/min.
The GC-MS analyses were conducted using the same equipment and experimental conditions as the GC analyses connected to the HP 5973 MSD mass spectrometer. The split ratio was set at 10:1, the ionization energy at 70 eV, and the emission current at 40 mA. The sampling rate was 1.0 scan/s and the mass range was 35 to 350 amu. To identify the components in the EO, their GC retention times were compared to those of known compounds, and their mass spectra were compared to those stored in the computer database and published spectra.38,39 The normalization of peak areas without the use of corrective factors served as the foundation for calculating the percentage composition.
Mosquito Larvicidal Screening
A. aegypti mosquitoes were maintained continuously at Duy Tan University, Vietnam. C. quinquefasciatus wild larvae were collected from the Hoa Khanh Nam district (16°03'14.9"N and 108°09'31.2"E) and were maintained in a laboratory. Dog biscuits and yeast powder were combined in a 3:1 weight ratio to feed the larvae. The mosquito larvicidal activity of the EOs extracted from the leaves and rhizomes of D. benenica was tested based on the method previously described by Huong et al. 67 Briefly, EOs dissolved in 1% ethanol (EtOH) were placed in a 500-mL container and added to water containing 20 larvae at the third and early fourth instar stage. The experiments took place at a temperature of 25 ± 2 °C. Each test included 4 repetitions and 5 concentrations (100, 75, 50, 25, and 12.5 μg/mL). Permethrin and EtOH were used as positive control and negative control, respectively. The mortality rate was observed after 24 and 48 h of exposure, without the addition of any nutritional supplements. LC50 values, LC90 values, and 95% confidence intervals were calculated using log-probit analysis in XLSTAT v. 2018.5 software (Addinsoft, Paris, France). 68
Antimicrobial Screening
The antimicrobial effects of the EOs extracted from the leaves and rhizomes of D. benenica were evaluated against 7 microorganisms: 3 Gram-positive bacteria (E. faecalis ATCC 299212, S. aureus ATCC 25923, and B. cereus ATCC 14579), 3 Gram-negative bacteria (E. coli ATCC 25922, P. aeruginosa ATCC 27853, and S. enterica ATCC 13076), and 1 yeast (C. albicans ATCC 10231). These microorganisms were provided by the Institute of Marine Biochemistry, VAST, Vietnam. The determination of MIC and IC50 values was conducted using the previously described broth microdilution susceptibility method. 67 The bacteria were cultured in Mueller-Hinton broth, whereas the yeast was grown in Sabouraud broth.
To determine the MIC values, the EOs were dissolved in 1% dimethylsulfoxide, then diluted to the greatest concentration. In a 96-well microtiter plate, serial doubling dilutions of the EOs were made. The final concentration in each well was set to 5 × 105 CFU/mL for bacteria and 1 × 103 CFU/mL for yeast after overnight broth cultures of each strain were made. After that, the bacteria and yeast were cultured for 24 h at 37 °C and 30 °C, respectively. Positive controls included streptomycin for bacteria and cycloheximide for yeast. The lowest concentration at which no visible growth of the microorganism occurs was defined as the MIC value.
To determine the IC50 values, the percentage of microorganisms exhibiting growth inhibition was determined by measuring turbidity using an Epoch 2 Microplate Spectrophotometer (Agilent Technologies Inc., USA). The obtained data were analyzed using the RawData computer software (Brussels, Belgium). The IC50 values were defined as the concentration at which the EO inhibits 50% of microbial growth, indicated by a 50% reduction in turbidity.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231193541 - Supplemental material for Essential Oils of Distichochlamys benenica: Chemical Constituents, Mosquito Larvicidal and Antimicrobial Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X231193541 for Essential Oils of Distichochlamys benenica: Chemical Constituents, Mosquito Larvicidal and Antimicrobial Activities by Le Thi Huong, Do Ngoc Dai, Dau Ba Thin and Nguyen Huy Hung, Bui Bao Thinh in Natural Product Communications
Footnotes
Acknowledgments
The authors would like to thank the editor and anonymous reviewers for their thoughtful comments and efforts toward improving our manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
