Abstract
Introduction
The unpaired electron of a free radical renders it unstable and highly reactive.
1
Consequently, it tends to receive and donate electrons to attain structural stability. This results in the beginning of a chain reaction cascade since the attacked molecule loses an electron and becomes an unpaired electron.2,3 The overproduction and accumulation of free radicals damages cells, biomolecules, and lipids.4,5 The excess production of free radicals generates oxidative stress which plays a major role in the development of various diseases including leukemia, cancer, cardiovascular diseases, inflammatory diseases, and diabetes.6–8 Sargassum macrocarpum is a perennial brown alga of the family, Sargassaceae which is distributed along the South Korean and Japanese coasts. It usually grows at depths of 10 m, is up to 3 m in length and contributes to the sea forest.
9
In previous studies, S. macrocarpum have been reported to contain various secondary metabolites such as meroterpenoids, terpenoid lactones, furan derivative, and plastoquinones.10–13 In this paper, the isolation, structural elucidation, and anti-oxidant activities of these compounds (
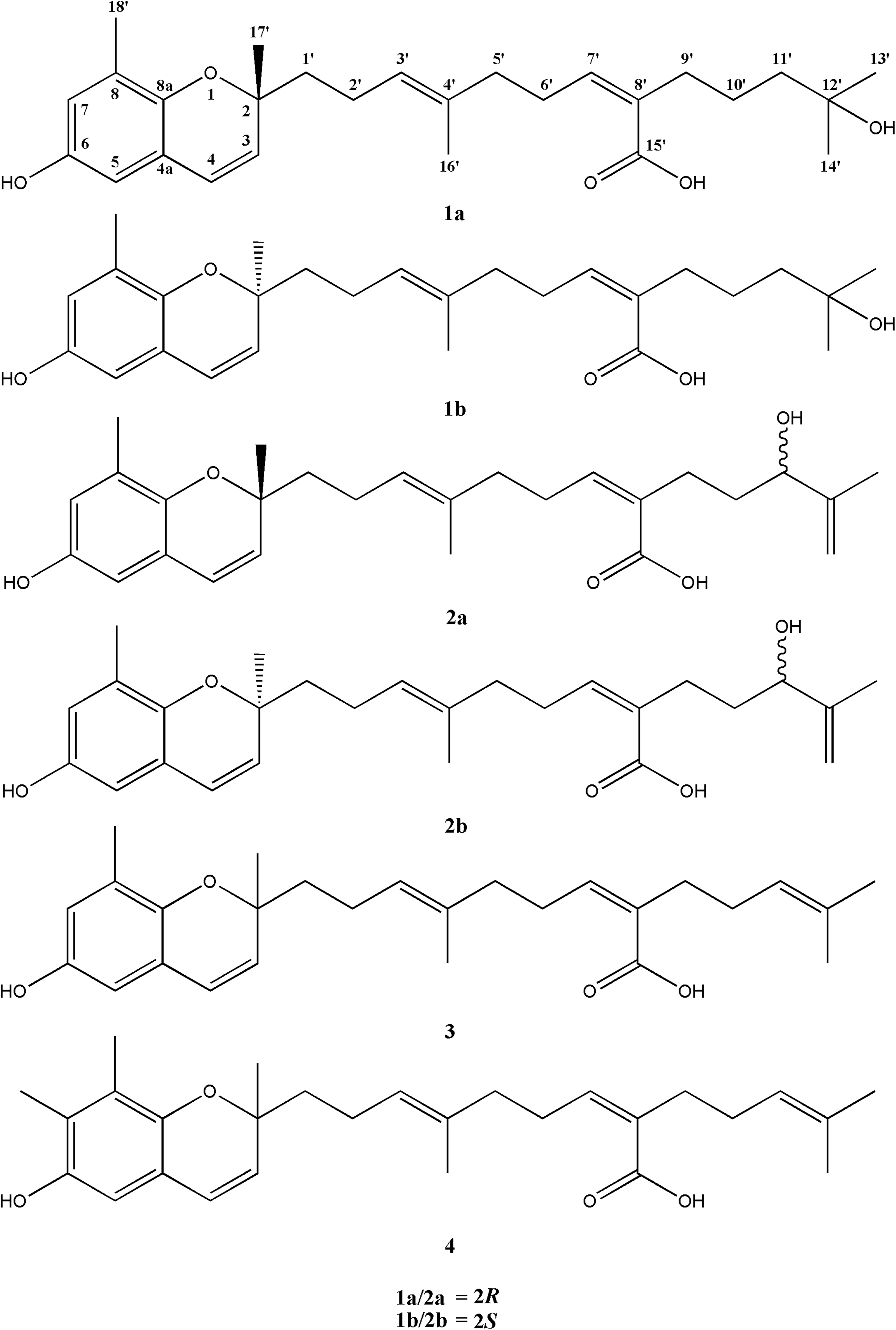
Chemical structures of
Results and Discussion
Compound (

HMBC and 1H-1H COSY correlations of
1H and 13C NMR Spectroscopic Data for Compounds

aNMR spectra were recorded at 500 MHz for 1H and 125 MHz for 13C in CDCl3.
Compound (
Compounds
The 4 isolated meroterpenoid derivatives (
Anti-Oxidant Activities of Compounds

Abbreviations: ABTS, 2,2′-azinobis (3-ethylbenzothiazoline-6-sulfonic acid); DPPH, 1,1-diphenyl-2-picrylhydrazyl.
aResults presented as the mean (n = 3)±SD.
Conclusion
This study reported the isolation, structure elucidation, and anti-oxidant activities of two novel meroterpenoid derivatives (
Experimental Section
General Experimental Procedures
The UV data were obtained using a UV-1650 UV spectrophotometer (Shimadzu, Kyoto, Japan). The NMR spectra were recorded on a Varian VNMRS 500 MHz FT-NMR spectrometer (Varian, Palo Alto, CA, USA). A J-1500 Circular Dichroism Spectrometer (Jasco, Tokyo, Japan) was used to measure the CD spectra. The specific rotation was measured on a JASCO P-2000 Digital Polarimeter (Jasco, Tokyo, Japan). The HPLC Arc system (Waters, Miliford, US) on an ODS column (Cosmosil, C18-MS-II, Kyoto, Japan) and a chiral column (Daicel, Chiralcel OD-R, Osaka, Japan) were used for the isolation and purification. High resolution-electrospray ionization-mass spectroscopy were recorded on a SCIEX X500R Q-TOF LC-MS/MS spectrometer (SCIEX, Massachusetts, US). The preparative HPLC was performed using a Nextra Prep (Shimadzu, Kyoto, Japan). The Biotage Selekt MPLC system (Biotage, Sweden, Uppsala) was used for the fractionation and purification.
Plant Material
The brown alga, S. macrocarpum C. Agardh. was collected from Jeju Island (Jeju-si, Udo-myeon) in June, 2019. A voucher specimen (NP-0764) was deposited at the National Marine Biodiversity Institute of Korea (MABIK).
Extraction and Isolation
Freeze dried and powdered S. macrocarpum (1.0 kg) was extracted with methanol at 20 °C, 3 times (1 day). The extract was evaporated and partitioned using organic solvents (n-hexane, chloroform, ethyl acetate, and butanol). The chloroform-soluble layer (40 g) was subject to silica gel open column chromatography using gradient elution solvents (n-hexane:ethyl acetate:chloroform:methanol = 20:1:0:0 → 0:0:0:1) to provide 9 fractions (S1-S9). S3 (12 g) was fractionated on a MPLC using a solvent mixture of ACN:H2O (3:7 to 10:0) to afford 4 sub-fractions (S3A-S3D). S3B (2.2 g) was continuously separated by preparative HPLC using ACN/H2O (63:37) to obtain
Measurement of Anti-Oxidant Activities
ABTS and DPPH radical scavenging assays were conducted according to a previously reported protocol.21,22 Briefly, ABTS was dissolved in H2O (7 mM) and incubated with potassium persulfate (2.45 mM) in the dark. The absorbances of the samples and mixed solutions were measured at 734 nm. DPPH was dissolved in ethanol. The sample and the DPPH solution were mixed and reacted for 10 min at room temperature. The absorbance of the reaction mixture was recorded at 517 nm.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231191857 - Supplemental material for Two New Meroterpenoid Derivatives From Sargassum macrocarpum With Their Anti-Oxidant Activities
Supplemental material, sj-docx-1-npx-10.1177_1934578X231191857 for Two New Meroterpenoid Derivatives From Sargassum macrocarpum With Their Anti-Oxidant Activities by Ji-Yul Kim, Dae-Cheol Choi, Jeong Min Lee, Hyun-Soo Kim, Dae-Won Ki, Seok-Chun Ko, Mi-Jin Yim, Gun-Woo Oh, Kyung Woo Kim, Chul Hwan Kim, Kyung Lee, Kyunghwa Baek and Dae-Sung Lee in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval in not applicable for this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Marine Biodiversity Institute of Korea (MABIK) Research Program (grant number 2023M00500).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent in not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
