Abstract
Objectives
The aim of the project was the isolation, structural elucidation, and antimicrobial and antifungal activities of compounds from the culture broth of the marine fungus Penicillium oxalicum M893 (isolated from the marine brown alga Spatoglossum sp.).
Methods
Combined chromatographic techniques were used to isolate antibacterial and antifungal compounds from the MeOH extract Penicillium oxalicum M893. The structures were elucidated by analyses of high-resolution electrospray ionization mass spectral and nuclear magnetic resonance data. The antimicrobial and antifungal effects of compounds were evaluated using the dilution turbidimetric broth method.
Results
One new sesterterpenoid, oxaliterpenoid (
This is a visual representation of the abstract.
Introduction
Marine fungi represent potential sources of structurally novel and diverse natural products, with approximately 38% (22,000) bioactive microbial-derived compounds from only 5% of discovered fungal species.1,2 Those compounds have shown a broad spectrum of biological properties, including antimicrobial, antiviral, anti-neuritis, antioxidant, and antitumor activities.3,4 Among these fungi, the Penicillium genus is one of the most widespread genera in the marine environment. The genus includes over 354 species, which have been intensively exploited due to their chemical diversity and associated biological activities.5-7 They are well known to produce various classes of bioactive compounds, such as alkaloids, polyketides, terpenoids, and lipopeptides.8,9 As a part of our ongoing search for secondary metabolites from marine-derived fungus, the methanol (MeOH) extract of Penicillium oxalicum M893 fermentation exhibited antimicrobial activity against three Gram-(+) bacteria (Enterococcus faecalis, Staphylococcus aureus, and Bacillus cereus) and one yeast strain (Candida albicans) with MIC values of 32, 128, 64, 16 µg/mL, respectively. Thus, the MeOH extract of M. oxalicum M893 has been further studied to identify the active compounds. Herein, we report the isolation, structural elucidation, and antibacterial and antifungal effects of one new sesterterpenoid and six known compounds from the fermentation broth of P. oxalicum M893 isolated from the marine brown alga Spatoglossum sp., collected at Phu Quy, Binh Thuan, Vietnam.
Results and Discussion
Compound

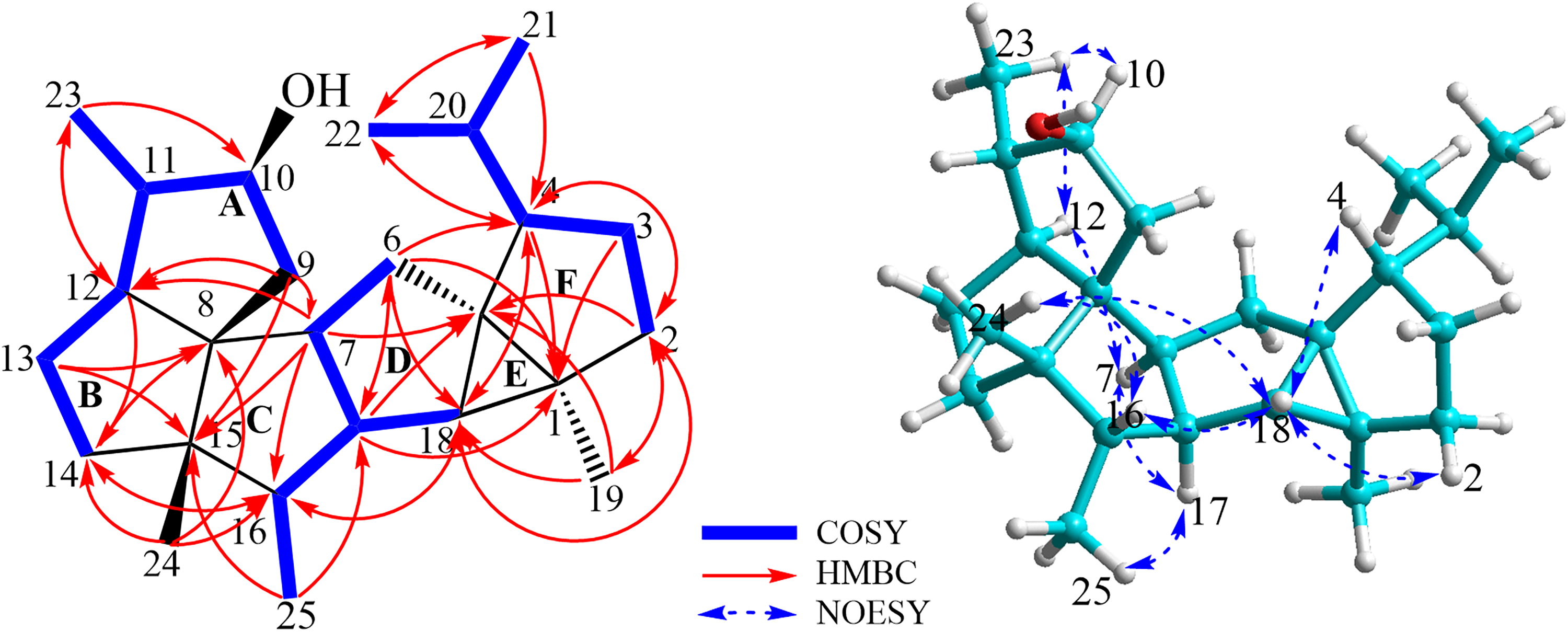
The key HMBC, COSY, and NOESY correlations of compound 1.
1H- and 13C-NMR Spectroscopic Data for Compound

The relative configuration of
The known compounds were identified to be aspergillusidone C (
Chemical structures of compounds 1–7.
All compounds were evaluated for their antibacterial effects against Gram-(+) bacteria, Enterococcus faecalis (ATCC299212), Staphylococcus aureus (ATCC25923), and Bacillus cereus (ATCC14579), Gram-(–) bacteria, (Escherichia coli (ATCC25922), Pseudomonas aeruginosa (ATCC27853), and Salmonella enterica (ATCC13076), and antifungal effect against Candida albicans (ATCC10231) (Table 2). Streptomycin and cycloheximide were used as positive controls. All compounds showed potent antibacterial activities against the Gram-(+) bacteria E. faecalis (ATCC299212), S. aureus (ATCC25923), and B. cereus (ATCC14579), and the yeast Candida albicans (ATCC10231), with MIC values ranging from 2 to 128 µg/mL. In previous studies, compounds from Penicillium sp. have been reported to exhibit an antibacterial effect.
14
The results suggested that compounds
Antibacterial and Antifungal Activities of Compounds

(-) MIC >256 µg/mL.
Material and Methods
General
Isolation and Taxonomic Identification
The fungal strain P. oxalicum M893 was isolated from the marine brown algalSpatoglossum sp., collected at a depth of 10 m, on the coast of Phu Quy, Binh Thuan, Vietnam in April 2021. (Spatoglossum sp. was identified by Prof. Do Cong Thung, Institute of Marine Environment and Resources, VAST); a voucher specimen (SS2104) was deposited at the Institute of Marine Environment and Resources, VAST.
The taxonomy of P. oxalicum M893 was identified by using 18S rRNA gene sequence analysis and compared with fungal 18S rRNA sequences in the GenBank database by NBCI Blast program. Its 18S rRNA gene sequence was registered on the GenBank database with the accession number OP522350.
Fermentation
P. oxalicum M893 was activated and inoculated into 1 L of PDA broth medium (comprising: 12 g/L potato extract, 8 g/L dextrose, 2 g/L soluble starch, 12 g/L instant ocean salt in 1.0 L of distilled water) to prepare the seed cultures. After 7 days of incubation at 30 oC with shaking of 150 rpm, the culture broth was used to inoculate into 20 flasks containing 2.5 L of PDA broth medium, pH 7.0 (comprising: 30 g/L potato extract, 20 g/L dextrose, 5 g/L soluble starch, 30 g/L instant ocean salt). The fermentation was conducted at 30 °C with a shaking speed of 100 rpm and harvested on the 14th day.
Extraction and Purification of Compounds
The fermentation broth of P. oxalicum M893 (50 L) was passed through a XAD-16 column (XAD-16, 10 kg). The column was washed with distilled water (70 L) and then eluted with MeOH (80 L). The MeOH solution was concentrated under reduced pressure to obtain the MeOH extract (20.6 g). This was chromatographed by silica gel column chromatography (CC) eluting with a gradient solvent of CH2Cl2/acetone (100:1, 50:1, 20:1, 10:1, v/v) to give four fractions (F1-F4). Fraction F1 (600 mg) was separated on a silica gel column eluting with n-hexane/EtOAc 5:1, providing four fractions, F1.1-F1.4. Fraction F1.1 (100 mg) was subjected to Sephedex LH-20 CC (100% MeOH) to yield compound
Oxaliterpenoid (1 )
White amorphous powder;
Antimicrobial Assays
See Supporting Information. 15
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231191636 - Supplemental material for New Sesterterpenoid from the Marine Fungus Penicillium oxalicum M893
Supplemental material, sj-docx-1-npx-10.1177_1934578X231191636 for New Sesterterpenoid from the Marine Fungus Penicillium oxalicum M893 by Thi Hoang Anh Nguyen, Thi Quynh Do, Thuy Linh Nguyen and Hong Minh Le Thi, Mai Anh Nguyen, Brian T Murphy, Thanh Xuan Dam, Doan Thi Mai Huong, Pham Van Cuong in Natural Product Communications
Footnotes
Acknowledgements
This research is funded by Vietnam Academy of Science and Technology (Grant No: VAST.TĐ.DLB.03/20-22) and University of Illinois Campus Research Board grant (5D43 TW010530-05).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
