Abstract
In this study, we focused on evaluating the antimicrobial, antioxidant, and tyrosinase-inhibiting properties of leaf and twig essential oils from Calocedrus macrolepis var. formosana. Additionally, we determined the major components associated with these effects. The findings revealed that α-pinene (36.4%) and limonene (23.6%) were the dominant components in the leaf essential oil, while α-pinene (36.2%), ferruginol (22.5%), and limonene (13.1%) were the major constituents in the twig essential oil. However, when comparing the biological activities of the 2 essential oils, the twig essential oil demonstrated excellent inhibitory effects against skin pathogens, as well as strong antioxidant and tyrosinase-inhibiting activities, in contrast to the leaf essential oil. The twig essential oil also exhibited excellent antimicrobial activity against 5 skin pathogenic strains, with the strongest inhibitory effect being that against Staphylococcus aureus, S epidermidis, and Candida albicans, which was accompanied by an inhibition zone of 46 to 48 mm, a minimum inhibitory concentration (MIC) of 125 μg/mL, and a minimum bactericidal concentration (MBC) of 250 μg/mL. The inhibitory activity of the twig essential oil against lipid peroxidation was found to be potent, as evidenced by an IC50 value of 216 ± 1.9 μg/mL. It also exhibited strong scavenging activity against free radicals, including 2,2-diphenyl-1-picrylhydrazyl (DPPH) (IC50 = 121.6 ± 0.5 μg/mL) and 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) (IC50 = 136.8 ± 1.6 μg/mL) cation radicals. In addition, it exhibited a high cupric ion reducing–antioxidant capacity (CUPRAC), with the A0.50 value determined to be 186.28 ± 0.5 μg/mL. It also exhibited adequate activity in terms of inhibiting in vitro tyrosinase activity, as evidenced by an IC50 value of 168.5 ± 0.6 μg/mL. Further analysis revealed that ferruginol was the active component responsible for the antimicrobial, antioxidative, and tyrosinase-inhibiting activities observed in twig oil. These findings suggest that the twig oil is a promising candidate for further research because of its favorable antimicrobial, antioxidative, and tyrosinase-inhibiting activity.
Introduction
Because of its mountainous terrain and subarctic, temperate, subtropical, and tropical regions, Taiwan has a complex climate, which has resulted in a wealth of forest resources. 1 Calocedrus macrolepis var. formosana (Florin) W. C. Cheng & L. K. Fu (Cupressaceae) is an exclusive coniferous species found in Taiwan. It grows at altitudes between approximately 500 and 1900 m and has a fragrant aroma, making it an excellent material for incense manufacturing in Taiwan. 2 Studies on C macrolepis var. formosana have demonstrated that the leaf, bark, heartwood, and seed essential oils exhibit various beneficial properties.3–10 These include antioxidant effects, antifungal properties, antibacterial activity, antitermitic properties, mosquito-repellent properties, anti-inflammatory effects, inhibition of lymphangiogenesis, and potential anticancer activity. Based on the available information, there have been no studies conducted to examine the chemical composition or biological activity of the essential oils derived from the twigs of C macrolepis var. formosana. The objective of this study was to extract leaf and twig essential oils from C macrolepis var. formosana in order to determine their chemical components. Additionally, we assessed the inhibitory effects of these essential oils on the growth of skin pathogens, their antioxidant activity, and their tyrosinase-inhibiting activity. Overall, our results highlight novel application avenues for this tree species endemic to Taiwan.
Results and Discussion
Essential Oils Yields
The extraction of yellow essential oils from the leaves and twigs of C macrolepis var. formosana was performed using hydrodistillation. The yields of the essential oils, calculated based on the dry weight of the leaves and twigs, were determined to be 0.51 ± 0.02 and 0.18 ± 0.01 mL/100 g, respectively. Notably, the leaf oils exhibited a higher yield compared to the twig oils.
Essential Oils Components
In this study, the composition and concentration of the essential oils derived from the leaves and twigs of C macrolepis var. formosana were analyzed using gas chromatography (GC) and gas chromatography-mass spectrometry (GC-MS). The results of this analysis, including the identified compounds, their concentrations, and linear retention index (LRI) values, are presented in Table 1. The compounds in the table are listed in the order of elution from a DB-5 capillary column.
Chemical Constituents of the Leaf and Twig Essential Oils of C macrolepis var. formosana.

LRILit = LRIs from another study. 15
LRIExp = computed calculated LRIs values obtained for a mixture of n-alkane hydrocarbons (ranging from C8 to C30) that were analyzed on a DB-5 capillary column.
Identification through:
MS = comparison of NIST and Wiley mass spectral libraries; LRI = linear retention index, like other studies15–17; CO-ST = co-injection/comparison to linear retention index and mass spectral standards.
Not detected.
A total of 37 compounds were identified in the essential oil extracted from the leaves of C macrolepis var. formosana. The main components included α-pinene (36.4%), limonene (23.6%), myrcene (8.0%), β-caryophyllene (7.2%), and β-pinene (4.5%). These components were categorized into 5 groups. Monoterpene hydrocarbons comprised 78.3% of the total content, while 13.5%, 2.5%, and 2.2% were attributed to sesquiterpene hydrocarbons, oxygenated monoterpenes, and oxygenated sesquiterpenes, respectively. Diterpenes were not detected in the leaf oil.
The results were compared to those of other studies, including Cheng et al, 6 Su et al, 11 and Adams et al. 12 It was found that there are similarities between our findings and their reported results. These studies also identified α-pinene, limonene, myrcene, and β-caryophyllene as the major components of C macrolepis var. formosana leaf oils. However, the composition of our leaf oils differed from those reported by Zhu et al 13 and Lin. 14 Specifically, Zhu et al 13 identified limonene, α-pinene, and α-cadinol as the main compounds of leaf oils, whereas Lin 14 identified β-caryophyllene, limonene, and α-pinene as the main components. These discrepancies in chemical composition may be due to differences in chemotype and geographical location.
In our analysis of the essential oils extracted from C macrolepis var. formosana twigs, a total of 31 compounds were identified. The prominent constituents included α-pinene (36.2%), ferruginol (22.5%), limonene (13.1%), myrcene (4.9%), and δ-2-carene (2.8%). These essential oils contained 64.3% monoterpene hydrocarbons, 23.9% diterpenes, 5.6% oxygenated monoterpenes, 5.2% sesquiterpene hydrocarbons, and 0.9% oxygenated sesquiterpenes, with the most relevant components being α-pinene, ferruginol, and limonene. Based on the information currently available, there have been no prior investigations on the essential oils extracted from the twigs of C macrolepis var. formosana. Therefore, this study represents the first research and publication on the subject.
Antimicrobial Activity
The skin microbiome is a complex ecosystem populated by countless microbes, such as bacteria, and fungi, among others. Similar to the gut microbiota, the skin microbiota performs a crucial effect in safeguarding the human body from pathogen infiltration and breaking down natural products. However, if the integrity of the skin barrier is compromised or there is an imbalance between beneficial and harmful microorganisms, it can lead to the development of skin diseases or even systemic conditions. These concepts have been supported by studies referenced as Scharschmidt and Fischbach, 18 Belkaid and Segre, 19 and Byrd et al. 20 In this study, 5 strains of microorganisms associated with skin lesions were selected: Staphylococcus aureus, S epidermidis, Escherichia coli, Pseudomonas aeruginosa, and Candida albicans. Disk diffusion and serial dilution were used to evaluate their antimicrobial activity. The leaf and twig oils are compared in terms of their inhibition zones (IZs), minimum inhibitory concentrations (MICs), and minimum bactericidal concentrations (MBCs). It can be observed that the twig essential oil displayed greater antimicrobial activity when compared to the leaf essential oil (Table 2). Additionally, the twig essential oil demonstrated a notable antimicrobial effect specifically directed toward all 5 strains. With regard to Gram-positive bacteria and fungi, such as S aureus, S epidermidis, and C albicans, the twig oil displayed the strongest inhibitory effect, as evidenced by inhibition zone diameters ranging from 46 to 48 mm. The minimum inhibitory concentration of the twig essential oil against these strains was determined to be 125 μg/mL, and the minimum bactericidal concentration was found to be 250 μg/mL. Hence, the antibacterial activity of the twig oil was found to be more pronounced against Gram-positive bacteria compared to Gram-negative bacteria. This finding is in line with other research studies,21–24 and the disparity in effectiveness can be attributed to the differing cell wall compositions of Gram-positive and Gram-negative bacteria. In general, the cell wall structure of Gram-positive bacteria consists of a multilayered and thicker peptidoglycan, whereas Gram-negative bacteria have a less dense peptidoglycan layer and an additional asymmetric double layer composed of phospholipids and lipopolysaccharides. This structural disparity often confers greater antibiotic resistance to Gram-negative bacteria compared to Gram-positive bacteria. 25
Antimicrobial Activities of the Leaf and Twig Essential Oils of C macrolepis var. formosana and Twig Oil Main Compounds.
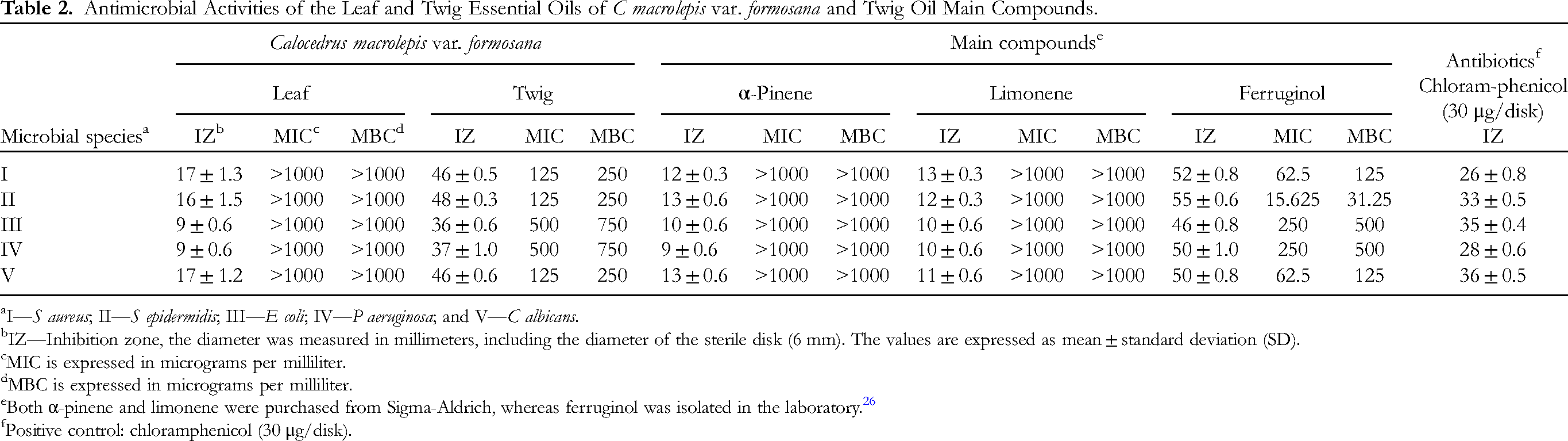
I—S aureus; II—S epidermidis; III—E coli; IV—P aeruginosa; and V—C albicans.
IZ—Inhibition zone, the diameter was measured in millimeters, including the diameter of the sterile disk (6 mm). The values are expressed as mean ± standard deviation (SD).
MIC is expressed in micrograms per milliliter.
MBC is expressed in micrograms per milliliter.
Both α-pinene and limonene were purchased from Sigma-Aldrich, whereas ferruginol was isolated in the laboratory. 26
Positive control: chloramphenicol (30 μg/disk).
Therefore, to examine the resistance to microbe potential of the active compounds in the C macrolepis var. formosana twig essential oil, the main components including α-pinene, ferruginol, and limonene were tested for their antimicrobial activity. We procured α-pinene and limonene from Merck Sigma-Aldrich, whereas ferruginol had been previously isolated in our laboratory and reported in a prior research paper. 26 We discovered that the main active compound was ferruginol (Table 2). Ferruginol has a phenolic structure and exhibits extremely potent antimicrobial properties. Many studies have also indicated that ferruginol has excellent antimicrobial activity.27–29
In this experiment, the major compound of the twig oil was ferruginol, a phenolic compound with excellent antibacterial activity. These observations are in accordance with the results obtained in other references.30–33
Antioxidant Activity
To assess the antioxidative properties of the leaf and twig oils, 4 in vitro assays were utilized. These assays included the β-carotene-linoleic acid assay, DPPH radical scavenging assay, ABTS cation radical scavenging assay, and CUPRAC assay. The positive controls used in these assays were α-tocopherol and butylated hydroxyanisole (BHA).
The β-carotene–linoleic acid test is a commonly utilized technique to measure the antioxidative capacity of natural products. The oils obtained from the twigs exhibited greater lipid peroxidation inhibition compared with the essential oil of the leaves. The IC50 value for the twig oil was determined to be 216.1 ± 1.9 μg/mL, as indicated in Table 3. This finding suggests that the essential oil extracted from the twigs may possess a greater ability to scavenge free radicals and protect against lipid peroxidation, compared to the essential oil derived from the leaves. This mechanism is crucial in preventing oxidative stress-related diseases.
Antioxidant Activities of the Leaf and Twig Essential Oils of C macrolepis var. formosana and Twig Oil Main Compounds.

Abbreviations: DPPH, 2,2-diphenyl-1-picrylhydrazyl; ABTS, 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulfonic acid); CUPRAC, cupric ion reducing–antioxidant capacity.
α-Tocopherol and BHA serving as positive controls.
The free radical-scavenging activities of the oils obtained from the leaves and twigs were evaluated using the DPPH• and ABTS•+ free radical tests, respectively (Table 3). In both tests, the twig oil displayed higher free radical-scavenging activities compared to the leaf oil. The IC50 values of the twig oil were determined to be 121.6 ± 0.5 μg/mL and 136.8 ± 1.6 μg/mL for the DPPH• and ABTS•+ tests, respectively.
The reducing power of the 2 essential oils was assessed using the CUPRAC assay. In Table 3, the twig essential oil (A0.50 = 186.3 ± 0.5 μg/mL) exhibited greater reducing power than did the leaf essential oil (A0.50 > 200 μg/mL).
Using the aforementioned 4 assays, the antioxidative capacities of the major compounds found in the twig oil, including α-pinene, ferruginol, and limonene, were evaluated. Based on the experimental findings, ferruginol demonstrated the most potent antioxidant effects among the major compounds tested. It demonstrated IC50 values of 69.5 ± 1.2, 34.5 ± 0.2, and 5.9 ± 0.3 μg/mL in the β-carotene–linoleic acid, DPPH, and ABTS assays, respectively. Additionally, it displayed an A0.50 value of 53.8 ± 0.4 μg/mL in the CUPRAC assay. Many studies have also reported on the excellent antioxidant activity of ferruginol.34–36 This result is not surprising, as phenolic compounds possess excellent antioxidant activity by effectively scavenging free radicals and exerting antioxidative potentials through proton-coupled electron transfer reactions with peroxide radicals.37,38
Tyrosinase-Inhibiting Activity
Tyrosinase serves as a key regulatory enzyme in the process of melanin synthesis. The activity of tyrosinase can be detected by measuring the rate at which it converts levodopa to dopaquinone, which can be used as a platform for screening melanin synthesis inhibitors. 39 Figure 1 depicts the inhibitory effect of the essential oils derived from the leaves and twigs of C macrolepis var. formosana on tyrosinase. The concentration range used for testing the oils was 31.25 to 1000 μg/mL. The inhibitory effect of the twig essential oil was greater than that of the leaf essential oil, and it exhibited an increasing inhibitory effect with higher tyrosinase concentrations. The IC50 value for the twig essential oil was determined to be 168.5 ± 3.2 μg/mL. Previous research has provided insights into the inhibitory effects of essential oils from different plant species on tyrosinase activity. For example, the Cinnamomum cassia oil demonstrated an IC50 value of approximately 6.25 mg/mL. 40 Similarly, when the concentration of Alpinia aquatica rhizome oil was 1 mg/mL, it exhibited a tyrosinase inhibition rate of 9.5%, 41 while the Eucalyptus camaldulensis flower oil was 26 mg/mL, showed an approximate tyrosinase inhibition rate of 30%. 42 Based on the comparison with the essential oils of other plants, the twig oil of C macrolepis var. formosana exhibits a higher potential for inhibiting melanin synthesis, indicating its potential as a melanin synthesis inhibitor. In vitro, studies were conducted to identify the key compound responsible for tyrosinase inhibition, involving the evaluation of α-pinene, limonene, and ferruginol. Among these compounds, ferruginol demonstrated the most significant inhibitory effect, with a half-maximal inhibitory concentration of 51.2 ± 2.8 μg/mL, confirming its role as the primary compound responsible (Figure 1).

The tyrosinase-inhibiting activity of the leaf and twig oils of C macrolepis var. formosana and Twig Oil Main Compounds.
Conclusion
The aim of this study was to analyze the chemical composition of essential oils extracted from the leaves and twigs of C macrolepis var. formosana. The leaf oil was found to be rich in α-pinene (36.4%) and limonene (23.6%), while the twig oil contained significant amounts of α-pinene (36.2%), ferruginol (22.5%), and limonene (13.1%). Furthermore, the antimicrobial, antioxidant, and tyrosinase-inhibiting properties of these oils were assessed. The findings revealed that the twig oil exhibited the highest activity, attributed to the presence of ferruginol as the active compound. These results emphasize the need for further research and development, highlighting new opportunities for utilizing native tree species in Taiwan.
Materials and Methods
Plant Materials
In November 2022, field collections were conducted to obtain C macrolepis var. formosana fresh leaves and twigs. The collection site was Dalong Mountain, located in Miaoli, Taiwan, with coordinates N 24°55′38″, 121°00′13″ and an altitude of 700 meters. Dr Hsu Chun-Kai, affiliated with the Taiwan Forestry Research Institute (TFRI), performed the taxonomic identification of the plant. A voucher specimen (HCCLH-095) was carefully preserved and deposited at the Department of Wood Cellulose, TFRI, serving as a reference for future studies.
Isolation of Leaf and Twig Essential Oils
In order to obtain leaf and twig oils from C macrolepis var. formosana, the following extraction method was employed. A hydrodistillation technique was used, where 10 kg of each plant material were subjected to hydrodistillation for a duration of 3 hours using a Clevenger apparatus. This process was repeated 3 times. After the extraction process, the obtained essential oils were subjected to a drying step using Na2SO4. The purpose of this step was to remove any remaining moisture from the oils. Once dried, the essential oils were carefully transferred and stored in separate specimen bottles. To ensure their stability and preservation, the bottles were kept at a temperature of 4 °C. This controlled storage condition helps maintain the quality and longevity of the essential oils for further analysis and use. The essential oil yields, along with experimental data, were calculated from triplicate analyses. The experimental data are shown as average ± SD.
Essential oil Analysis
GC-FID and GC-MS were employed to analyze the essential oils extracted from the leaves and twigs of C macrolepis var. formosana, respectively. For GC-FID analysis, a Hewlett-Packard Model 6890 GC equipped with a FID was utilized. The detector was coupled with a DB-5 fused silica capillary column, which had a composition of 5% phenyl and 95% methylpolysiloxane. The column had a length of 30 m, an inner diameter of 0.25 mm, and a film thickness of 0.25 μm. The GC oven was initially set to 50 °C and maintained for 2 minutes. Subsequently, the temperature was ramped up at a rate of 5 °C per minute until it reached 250 °C. The injector temperature was maintained at 270 °C, while the detector temperature was kept constant at 250 °C throughout the analysis. A carrier gas of hydrogen was used for the analysis, flowing at a rate of 1.0 mL/min with a split ratio of 1:60. Prior to injection, the sample volume of 1 μL was diluted in ethyl acetate at a ratio of 1/100 (v/v). Linear Retention Indices (LRIs) of compounds were calculated by comparing their elution times with those of a reference set of n-alkanes spanning from C8 to C30. The content of each compound in the essential oil is expressed as a percentage composition (relative quantity) determined using the normalization method, based on the electronic integration of peak areas obtained through GC-FID, without any correction factors.
For GC- MS analysis, a Hewlett-Packard Model 6890/5973 GG-MS equipped with a DB-5 fused silica capillary column was utilized. The carrier gas used was Helium (99.995% purity) at 1 mL/min, and the GC-FID analysis parameters were utilized for the GC-MS analysis as well. The data acquisition was performed in full-scan mode (m/z 30-500).
Component Identification
The identification of all compounds was based on the analysis and comparison of their LRIs, retention times, and mass spectra. These analyses and comparisons were conducted using data from the NIST and Wiley mass spectral databases, authentic standards, and relevant literature references.15–17
Antimicrobial Activity
In this study, microorganism strains associated with skin lesions were selected, including S aureus (ATCC 6538P), S epidermidis (ATCC 12228), E coli (IFO 3301), P aeruginosa (IFO 3080), and C albicans (ATCC 10231). The 5 microbial strains were sourced from the Bioresource Collection and Research Center (BCRC) of the Food Industry Research & Development Institute (FIRDI) in Hsinchu, Taiwan.
To evaluate the antimicrobial properties, the essential oils extracted from the leaves and twigs of C macrolepis var. formosana were subjected to the disk-diffusion assay, MICs, and MBCs assays.
The method of Bauer et al 43 uses the paper disc diffusion method. To promote uniform growth on the plate, the bacterial strains were cultivated on Mueller-Hinton agar (MHA) (Merck), while the yeast strain was cultured on Sabouraud dextrose agar (SDA) in separate Petri dishes. Once the agar plates were prepared, 10 μL of undiluted oil was added onto a 6 mm diameter filter paper disk. Afterward, the filter paper disk was meticulously positioned in contact with each plate and inoculated with the targeted microorganisms. Following incubation at a temperature of 37 °C for a period of 24 h, the inhibitory zone surrounding the 6 mm disc was measured. Chloramphenicol served as the positive control. The data is reported as mean ± standard deviation of 3 independent tests.
To determine the MIC, a microdilution susceptibility test was performed following the guidelines established by the National Committee for Clinical Laboratory Standards (NCCLS), 44 as previously described. 45 The essential oils under investigation were diluted using a series of twofold dilutions with DMSO, resulting in concentrations ranging from 15.625 to 1000 μg/mL. Each test was conducted in triplicate to ensure the accuracy and consistency of the results.
To determine the MBC, the method proposed by Chikezie 46 was used. Mueller-Hinton agar (MHA) was used for bacterial cultures, and Sabouraud dextrose agar (SDA) was used for yeast cultures. One hundred μL aliquots of each dilution, which did not exhibit visible growth, were aseptically spread onto agar plates. The plates were then incubated at 37 °C for 24 h. Subsequently, the quantity of colony-forming units (CFUs) was calculated and compared to the count observed on the control plates. The MBC was determined as the minimum concentration of the essential oil that achieved a killing efficacy of over 99.9% against the initial inoculum. To ensure precision and consistency, each experiment was repeated 3 times.
Antioxidant Activities
β-Carotene-Linoleic Acid Assay
The β-carotene-linoleic acid assay was employed with minor adjustments to evaluate the antioxidant activity. 47 The sample's ability to inhibit lipid peroxidation by 50% (IC50, μg/mL) was determined by plotting the level of antioxidant efficacy (percentage of inhibition) against sample concentration (μg/mL).
DPPH Free Radical Assay
A DPPH assay was carried out following the procedure outlined by Ho and Su
48
To summarize, the experimental procedure involved mixing 50 μL of essential oil dilutions with different concentrations and 5 mL of a methanol solution containing 0.004% DPPH. After a 30-minute incubation period. The samples were subjected to spectrophotometric analysis using a Jasco 7800 spectrophotometer, and their absorbance was measured at a wavelength of 517 nm. The experiments were conducted 3 times, with α-tocopherol and BHA serving as positive controls.
The IC50 values were determined by plotting the percentage of scavenging activity against the sample concentration.
ABTS Cation Radical Scavenging Assay
This assay was determined using spectrophotometry, following a method described by Re et al. 49 The IC50 value, indicating the concentration of the sample needed to inhibit 50% of ABTS•+ radicals, was determined by plotting the level of inhibitory effect (inhibition %) versus the concentration (μg/mL).
Cupric Reducing Antioxidant Capacity (CUPRAC) Assay
This assay was conducted using the method described by Apak et al. 50 The A0.50 value, which represents the sample concentration that provides an absorbance value of 0.500, was determined by plotting the CUPRAC absorbance signal against sample concentration (μg/mL). This value was used to report the results of the CUPRAC assay.
Tyrosinase Inhibition Assay
The spectrophotometric method described by Masuda et al 39 was employed to evaluate the tyrosinase-inhibiting activity. Kojic acid is used as the positive compound. The inhibitory effect of essential oils and the main compounds of twig oil on the enzyme is expressed as a percentage of inhibition at concentrations ranging from 31.25 to 1000 μg/mL.
The inhibition for each enzyme assay was calculated using the formula:
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
