Abstract
Camellia sinensis leaves are one of the most economically important crops, and various tea cultivars are available in Japan. In the present study, we compared the constituents of Camellia sinensis var. sinensis cv. Yabukita (“Yabukita”), a standard tea cultivar, and those of C. sinensis var. sinensis cv. Sayamakaori (“Sayamakaori”), grown in Saitama Prefecture, using proton nuclear magnetic resonance (1H-NMR) metabolomics technology. Principal component analysis and hierarchical cluster analysis of 1H-NMR spectroscopic data of the MeOH extracts of C. sinensis leaves revealed differences in the metabolic profiles of each tea cultivar. Chromatographic separation based on 1H-NMR spectral data showed that kaempferol 3-O-β-D-glucopyranosyl-(1 → 3)-α-L-rhamnopyranosyl-(1 → 6)-β-D-glucopyranoside was a distinct component of “Sayamakaori”. This compound can be used as maker for the identification of “Sayamakaori” and the maintenance of its quality.
Keywords
Introduction
The tea plant Camellia sinensis is one of the most economically important crops in Japan and other countries. The three main types of tea can be classified according to fermentation (fermentation here means an oxidation process) as follows: green (unfermented), oolong (semi-fermented), and black (fermented). Flavan-3-ols, phenolic acids, purine alkaloids, condensed tannins, hydrolyzable tannins, saponins, flavonols, and their glycosides are secondary metabolites in fresh tea leaves. Particularly, flavan-3-ol is a basic skeleton of (−)-epicatechin (EC), (−)-epicatechin gallate (ECG), (−)-epigallocatechin (EGC), and (−)-epigallocatechin gallate (EGCG), which are well known green tea ingredients.1,2 These components have anticancer, anti-cardiovascular, neuroprotective, antioxidant, hepatoprotective, anti-obesity, antidiabetic, antibacterial, and antiviral effects.3,4 Studies on volatile components have also been conducted, and more than 600 volatile components have been identified to date. 5 Although various tea cultivars are cultivated in Japan, almost all of these have been bred from C. sinensis var. sinensis cv. Yabukita (“Yabukita”). “Yabukita” accounts for over 70% of the cultivation area in Japan. Some compositional studies regarding the differences between cultivars have been conducted, and it has been concluded that the differences were due to differences in the concentrations of common components.6–8 C. sinensis var. sinensis cv. Sayamakaori (“Sayamakaori”), originating from Saitama Prefecture, Japan, is said to have a strong young shoot aroma that differs from that of “Yabukita” and has a bitter taste and high yield. However, the components that characterize “Sayamakaori” have not yet been clarified. Characteristic compounds can be used to distinguish between species and contribute to quality control. Furthermore, these marker compounds may be useful for counterfeit detection as protecting crop brands such as tea is important.
Metabolite fingerprinting approaches aided by nuclear magnetic resonance (NMR) spectroscopy provide valuable metabolite signatures for complex plant extracts. The analysis per sample is fast, no derivatization is required, and specific chemical entities can be identified using this method.9–11 Some studies have used NMR-based metabolomic analysis to characterize plant cultivars and genera and identify the characteristic components of these cultivars. In addition, NMR metabolomics is known to be suitable for identifying the differentiable compounds of some cultivars.12–14 In this study, we determined the characteristic components of the tea cultivar “Sayamakaori” using NMR metabolome analysis and multivariate statistical analyses, including principal component (PCA) and hierarchical cluster analyses (HCA).
Results and Discussion
A total of 23 samples were used in this study (Table 1). The samples were macerated with methanol to obtain the corresponding extracts. After thoroughly drying in a vacuum, the extracts were dissolved in CD3OD for 1H-NMR analysis. NMR spectra were analyzed using PCA and HCA. PCA was depicted as a score plot and consisted of two composite variables: principal component PC1 (with the largest variance of the data) and PC3 (with the third-largest variance of the data). The samples were divided into two groups based on PCA score plots (Figure 1). These two groups were related to the variety and were distinguished into the “Sayamakaori” and “Yabukita” groups. In addition, the HCA constellation dendrogram of C. sinensis leaves was divided into two groups (Figure 2). These findings indicate that the components of the tea cultivars differ from each other. Next, we identified an 1H-NMR signal specific to the leaves of “Sayamakaori.” As shown in Figure 3, these signals were selected as specific signals for “Sayamkaori” in the loading plot of the PCA. Among these signals, δ 8.02 was focused on because it appeared independently without overlapping with other signals in the 1H-NMR spectrum. The signal was statistically specific for “Sayamakaori” (P < 0.0001), as shown in Figure 4. Furthermore, a signal was observed at δ 8.02 for “Sayamakaori” by direct comparison of 1H-NMR spectra (Figure 5). Therefore, we attempted to isolate the compound whose signal appeared at δ 8.02 using chromatographic methods. The “Sayamakaori” methanol extract was fractionated based on the specific 1H-NMR signals using a Diaion HP-20 polymeric adsorbent (Mitsubishi Chemical Industries Ltd, Chiyoda, Tokyo, Japan) and eluted with water and 30%, 50%, and 70% aqueous MeOH to obtain the corresponding fractions. Thereafter, polymerized tannins were removed from the 70% methanolic elute by passing it through an open silica gel column. The elute was concentrated under reduced pressure, and the residue was purified by medium-pressure liquid chromatography with octadecyl silica gel to obtain Fr.1 to Fr.240. The specific signals for “Sayamakaori” were observed in Fr. 92-95, using 1H-NMR spectroscopy. Furthermore, 1H-NMR spectra indicated that these fractions (Fr. 92-95) contained only one pure component. Therefore, these fractions were combined and designated as compound

PCA score plot based on 1H-NMR spectra of 23 tea samples. Numerals in the score plot represent samples in Table 1.
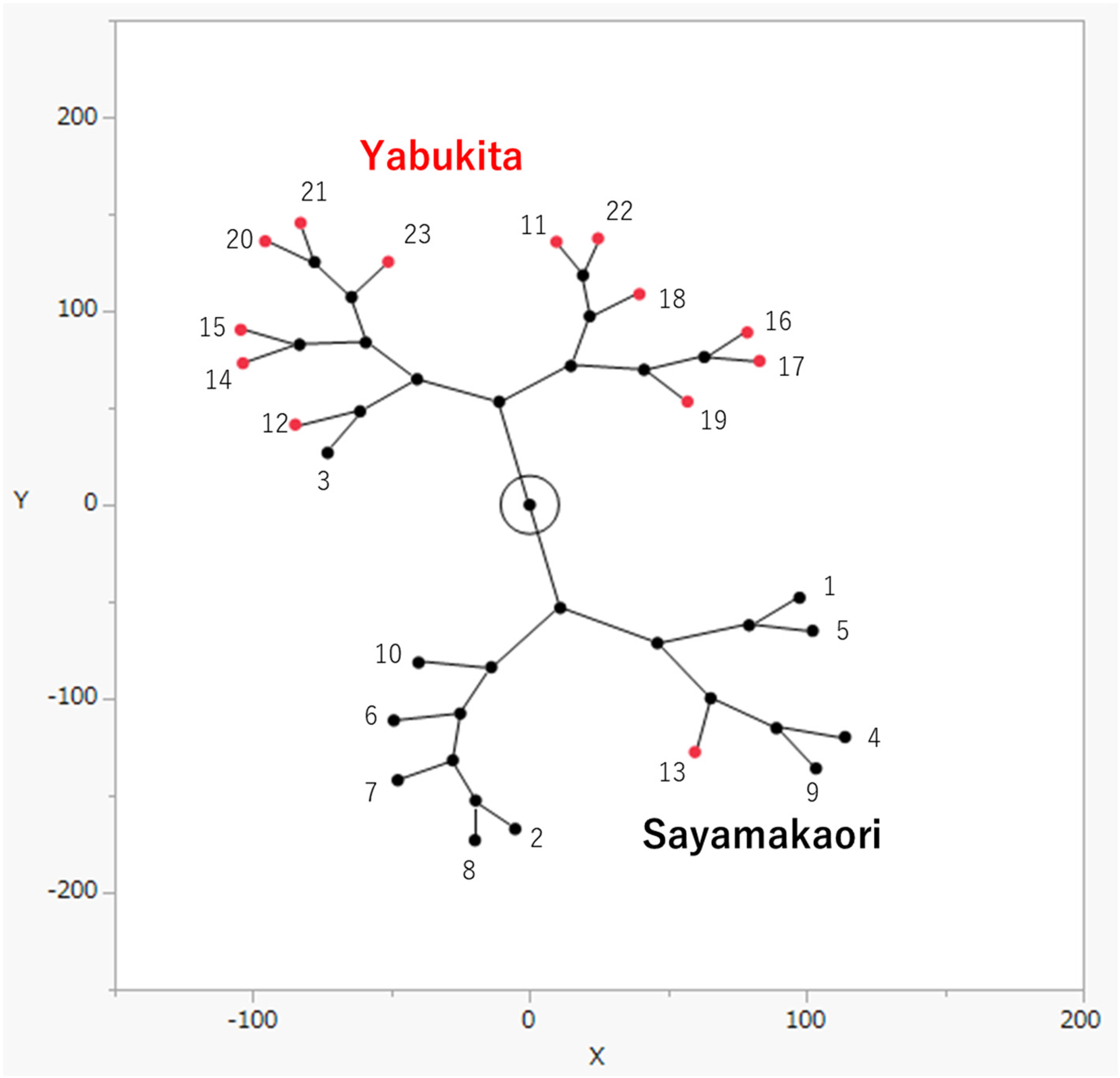
HCA constellation dendrogram derived from 1H-NMR spectral data. Numerals in the score plot represent samples in Table 1.

Loading plot of PCA. Numerals in the loading plot represent 1H-NMR chemical shifts.
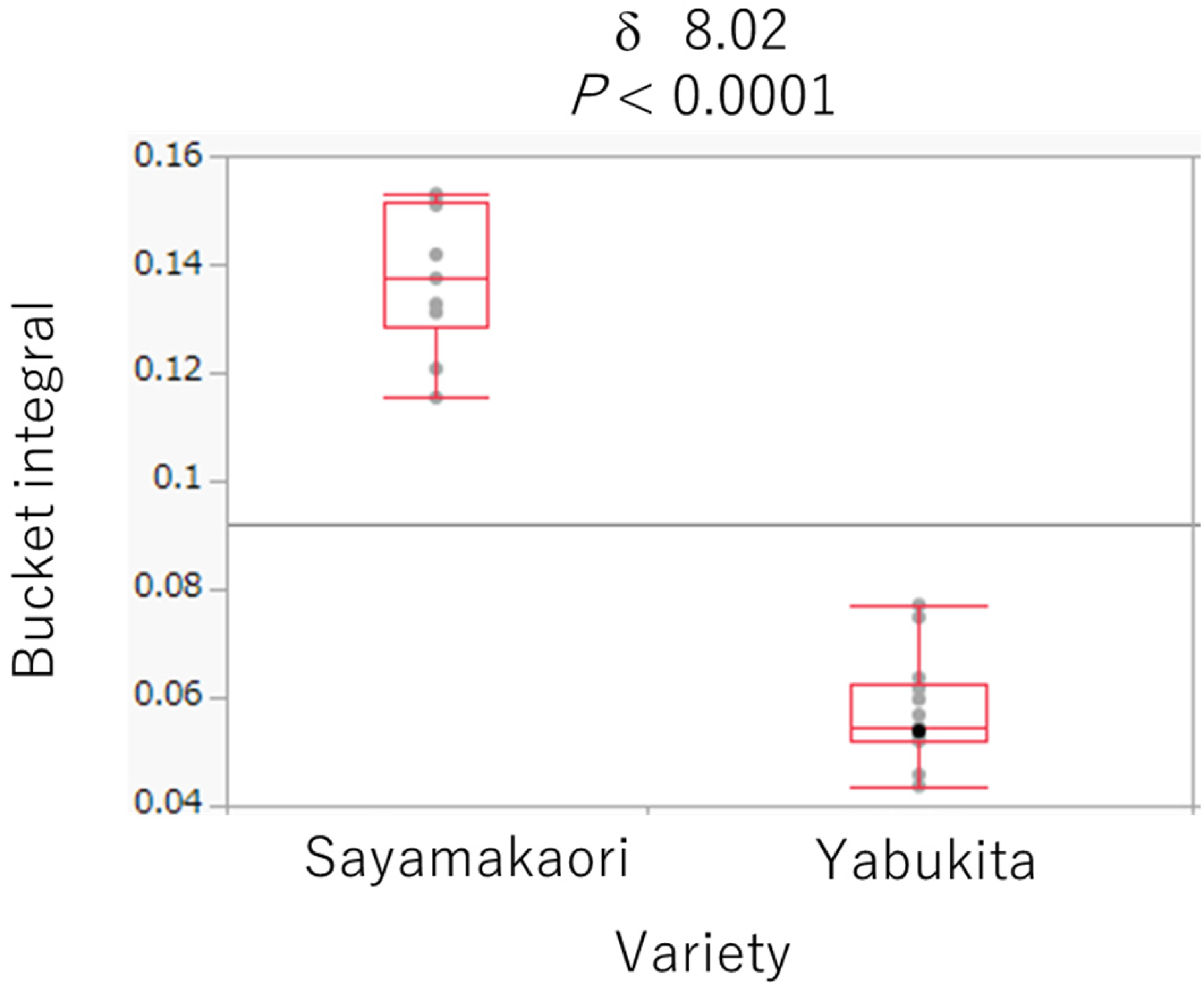
Box plots of the bucket integral at δ 8.02 of “Sayamakaori” and “Yabukita.”

Comparison of 1H-NMR spectra of representative tea cultivars. (a) Overview, (b) Enlarged view.

Chemical structure of compound
Tea Samples Used in this Experiment.

Material and Methods
The Reagents
Methanol-d4 (CD3OD) was purchased from Kanto Chemical Co. (Chuo-ku, Tokyo, Japan).
Tea Samples
Fresh green tea (C. sinensis var. sinensis cv. Sayamakaori and C. sinensis var. sinensis cv. Yabukita) leaves (one bud and two leaves) were obtained from the The Green Tea Laboratory of Saitama Prefectural Agriculture and Forestry Research Center, Iruma, Saitama, on May 7, 2021. All tea leaves were dried at 23 °C for 2 weeks. The specimens were maintained at the Laboratory of Natural Products and Phytochemistry, Department of Pharmaceutical Sciences, Faculty of Pharmacy and Pharmaceutical Sciences, Josai University, Japan.
Instrumentation
1H- and 13C-NMR spectra were recorded at 400 MHz on an AVANCE NEO 400 (Bruker Corporation, Billerica, MA, USA) operating at 400 MHz for 1H and 100 MHz for 13C. MS was performed on a JMS-700 double-focusing magnetic sector mass spectrometer (JEOL, Tachikawa, Tokyo, Japan).
Extraction
The tea samples were extracted with MeOH for 24 h at 23 °C, after which the extract was evaporated to obtain the corresponding residue. The MeOH extracts were dissolved in CD3OD at a concentration of 10 mg/mL for 1H-NMR spectral measurements.
NMR Spectra Measurements of Green Tea Extracts
One-dimensional NMR spectra were recorded at 20 °C with solvent signals as internal references. Each spectrum consisted of 65 536 complex data points with a spectral width of 8196.722 Hz. The spectra were obtained over 16 scans with an acquisition time of 4.0 s and a relaxation delay of 1.00 s per scan. The flip angle of the detection pulse was set to 30°.
NMR Data Reduction Procedures and Pattern Recognition Analysis
Each NMR spectrum was divided into 250 regions between 0.00 and 10.00 ppm (0.04 ppm width). Segments of each spectral region were integrated; the integration regions from 1.12 to 1.24 ppm, 3.20 to 3.36 ppm, and 4.76 to 4.92 ppm containing solvent and water signals were removed from the data leaving 243 regions for all data. The remaining integrals for each spectrum were normalized by summing up 100 integrals to correct for concentration differences among tea extracts. Spectral processing was performed using ALICE2 for Metabolome (version 5.0; JEOL). Multivariate analyses, such as PCA and HCA, were conducted using JMP Pro 16 (SAS Institute Inc., Cary, NC, USA). The variables were standardized with a mean of 0 and a standard deviation of 1. For HCA, Euclidean distances and Ward's method were used to establish the dendrograms. A Mann–Whitney U test was used to compare the differences in the integral value at δ 8.02 between “Sayamakaori” and “Yabukita,” and a P-value < 0.0001 was considered statistically significant.
Isolation and Identification of Kaempferol-3-O-β-D-glucopyranosyl-(1 → 3)-α-L-Rhamnopyranosyl-(1 → 6)-β-D-Glucopyranoside
A large amount of “Sayamakaori” was extracted again to isolate a specific compound for this variate. Methanol extracts prepared by extracting a large amount of “Sayamakaori” were fractionated guided by 1H-NMR signals identified as specific for “Sayamakaori.” The leaves of “Sayamakaori” (400 g) were extracted with MeOH for 24 h at room temperature, and the same procedure was repeated twice for 24 h. The MeOH extracts were combined and concentrated to obtain 108 g of residue. Since a specific signal on δ 8.07 was observed in this residue by 1H-NMR measurement, 100 g of the residue was subjected to a Diaion HP-20 polymeric adsorbent (Mitsubishi Chemical Industries Ltd) and eluted with water and 30%, 50%, and 70% aqueous MeOH to obtain the corresponding fractions. (Water fraction, 26.7 g; 30% aqueous MeOH fraction, 31.7 g; 50% aqueous MeOH fraction, 21.0 g; 70% aqueous MeOH fraction, 3.7 g; MeOH fraction, 10.7 g). The 70% aqueous MeOH fraction (3.4 g) was loaded onto an open silica gel column (ϕ 50 × 420 mm) and separated by elution with CHCl3: H2O = 5:1. The eluate (200 mg) was purified by preparative MPLC using an ULTRA PACK ODS-SM-50C size C (ϕ 37 × 300 mm; Yamazen Corp., Osaka, Japan). Preparative conditions were eluted in an isocratic system (MeOH: H2O = 1:1, 6 mL/min) to obtain Fr.1–Fr.240. Each fraction was collected at 6 mL/tube. Fr. 92-95 contained a specific component of “Sayamakaori.” Each fraction was combined with Compound
Supplemental Material
sj-xlsx-1-npx-10.1177_1934578X231180461 - Supplemental material for Discrimination of Sayamakaori and Yabukita Which Are Original Plant Source of Japanese Green Tea and Identification of Specific Compounds for the Former by Nuclear Magnetic Resonance Metabolomics Techniques Combined with Isolation by Chromatography Methods
Supplemental material, sj-xlsx-1-npx-10.1177_1934578X231180461 for Discrimination of Sayamakaori and Yabukita Which Are Original Plant Source of Japanese Green Tea and Identification of Specific Compounds for the Former by Nuclear Magnetic Resonance Metabolomics Techniques Combined with Isolation by Chromatography Methods by Aiko Sano, Takako Shibata, Atsushi Takahasi, Takami Yokogawa, Masashi Kitamura and Ryuichiro Suzuki in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not include contain any studies on human or animal subjects.
Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
