Abstract
Aspidiata E (
Introduction
The genus Aspidistra Ker Gawler comprises herbaceous rhizomatous plants, 1 which are widely distributed in subtropical and tropical monsoon forests of Southeast Asia, ranging from lowlands up to 1500 m in altitude. 2 Research into the species diversity of this evergreen genus has long been neglected, since flowers of Aspidistra species are usually located on the ground. 2 However, in recent decades, many new species have been discovered in southern China and northern Vietnam. As a result, this area is regarded as the center of species diversity of this genus. From 2000 to 2018, the number of Aspidistra species rapidly increased from 55 to more than 170, 3 and at least 12 new species were described and reported in 2021. Consequently, the genus Aspidistra currently includes about 200 species, approximately half of which are known to exist in Vietnam. 1
Previous phytochemical studies indicated that saponins are the major constituents of this genus with over 90 compounds being reported.
4
Fractional extracts as well as isolates from the genus Aspidistra revealed remarkable biological activity including antiviral, antitumor,5,6 cytotoxic,4,7 antioxidant,8,9 and anti-inflammatory effects.
10
In some Asian countries, certain Aspidistra species have been used in traditional medicine for the treatment of fractures, back pain, and hypercholesterolemia.8,11 Our previous study isolated 5 compounds from A. triradiata, including 1 new spirostanol steroid (aspidiata A) and 1 new spirostanol saponin (aspidiata B), and evaluated the cytotoxicity of these isolates against 4 cancer cell lines.
12
In the ongoing exploration for natural compounds with potential cytotoxic properties from A. triradiata, we have isolated a new spirostanol saponin (
Results and Discussion
Compound
The 1H NMR spectrum of
1H and 13C NMR Data of Compound

Assignments were accomplished by 1H NMR, 13C NMR, HSQC, HMBC, COSY, and ROESY experiments.
Xyl, β-D-xylopyranosyl.
aOverlapping signals.
b150 MHz.
c600 MHz.
Additionally, the attendance of 1 sugar unit was deduced by a signal for 1 anomeric proton at δH 5.14 (1H, d, J = 7.8 Hz) correlating with an anomeric carbon at δC 103.1. The 13C NMR spectrum of
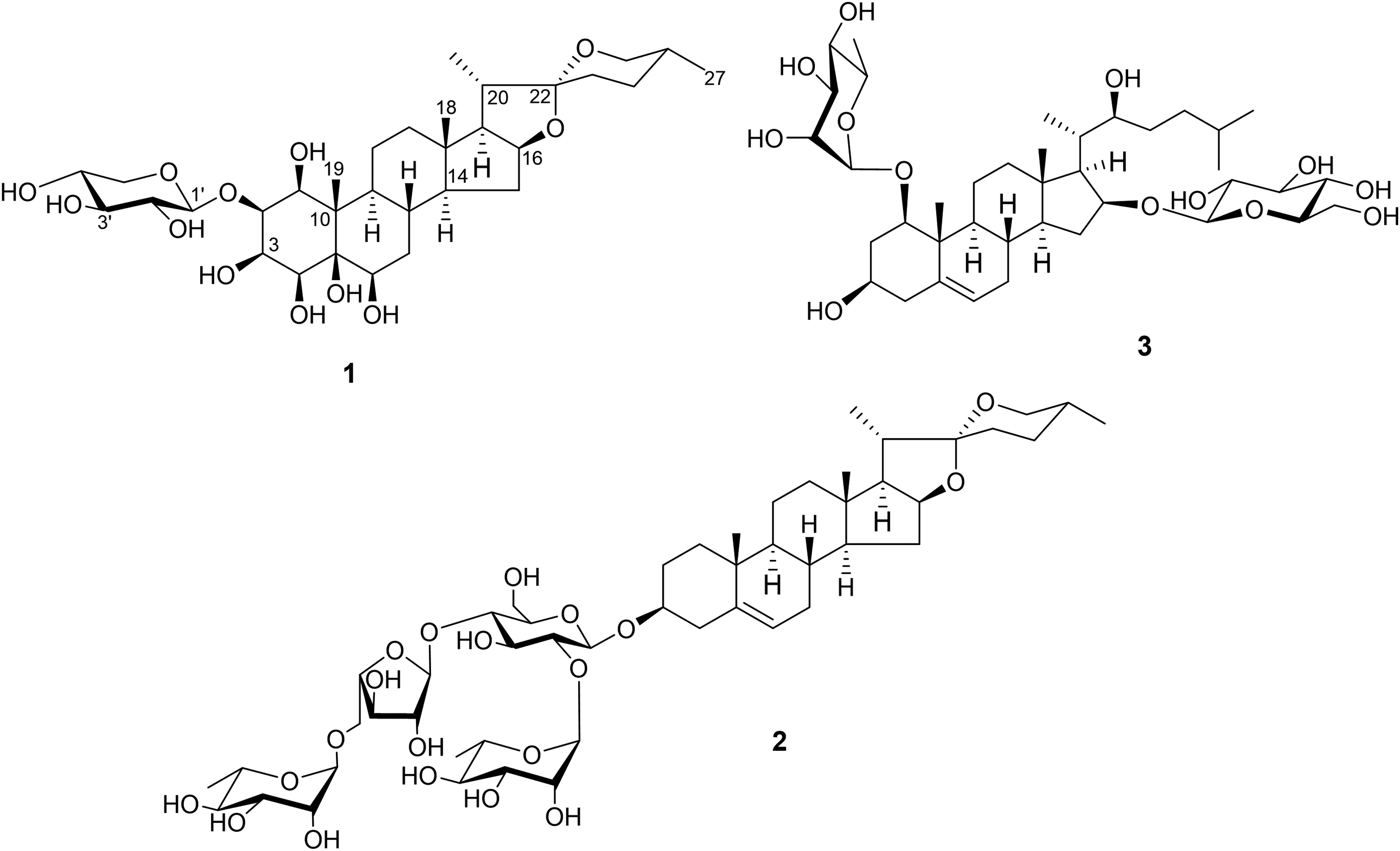
Chemical structures of compounds
The 1H and 13C NMR spectroscopic data of

Key HMBC (1H→13C, arrows) and COSY (bold lines, different colors) correlations of
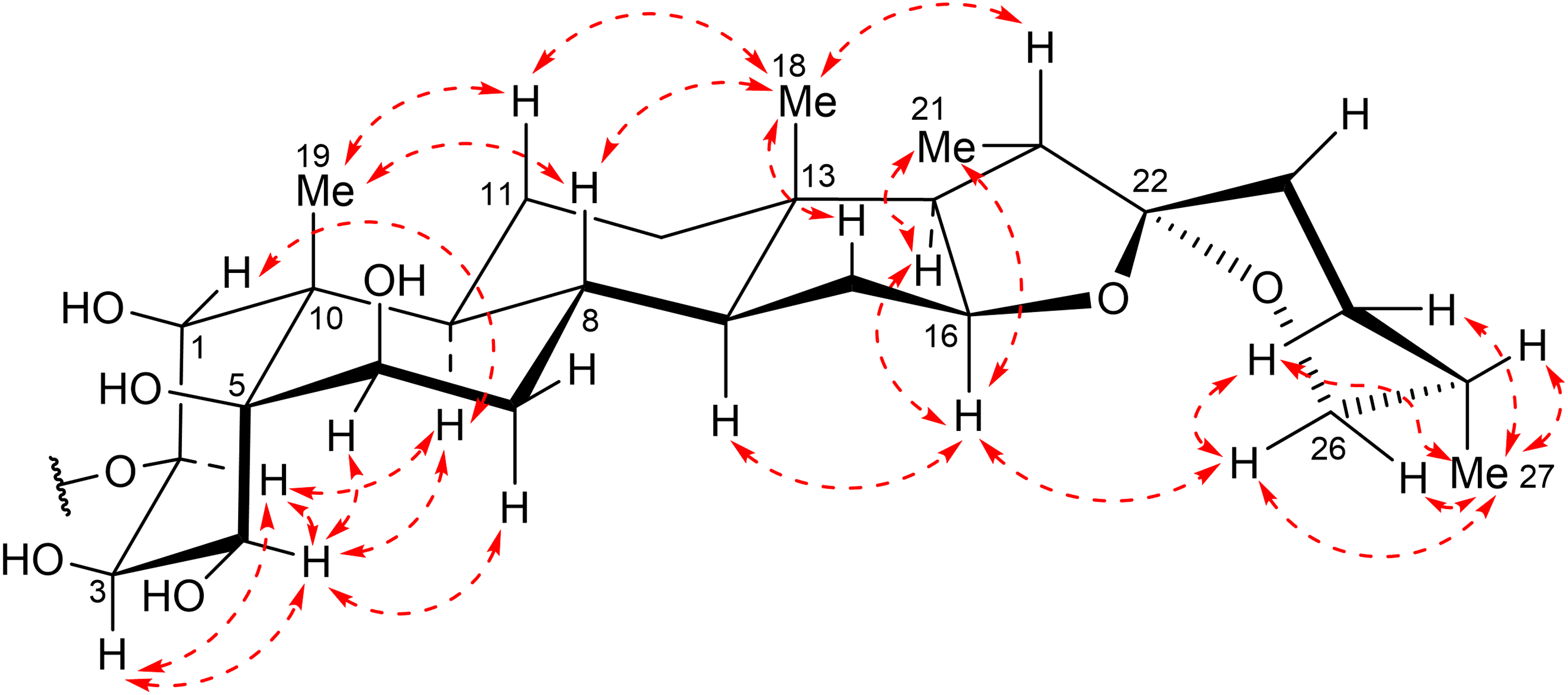
Key ROESY correlations (dashed arrows) of
The ROESY correlations of H-20 (δH 1.92)/H3-18, H-16 (δH 4.55)/H3-21 implied that the CH3-21 and H-20 groups were oriented in α and β, respectively. Because H-20 resonated at higher field than δH 2.2, the orientation relationships between this proton and the oxygen atom on F ring was trans.
14
Consequently, the S* configuration was assigned for C-22.
15
The 13C NMR data of the F ring and C-27 [δC 109.2 (C-22), 31.8 (C-23), 29.2 (C-24), 30.5 (C-25), 66.8 (C-26), 17.2 (C-27)] of
A series of reactions, including hydrolysis of
The remaining compounds were confirmed as (22S*,25R*)-spirost-5-ene-3β-yl O-α-L-rhamnopyranosyl-(1→2)-O-[O-α-L-rhamnopyranosyl-(1→5)-α-L-arabinofuranosyl-(1→4)]-β-D-glucopyranoside (
The sulforhodamine B (SRB) assay was used to access the cytotoxic potential of the isolated compounds against MCF-7, SK-LU-1, HepG2, and HT-29 human cancer cell lines (
Cytotoxicities of

aIC50 (concentration that inhibits 50% of cell growth).
bPositive control.
Based on our results, the following cytotoxic structure-activity relationships can be revealed. Regarding the spirostanol saponins without a hydroxyl group at C-17, such as compounds
Conclusion
Through an investigation of the chemical composition of A. triradiata, a new spirostanol saponin, aspidiata E (
Materials and Methods
General Experimental Procedures
Optical rotations were recorded on a JASCO-2000 Polarimeter (JASCO, Tokyo, Japan), IR spectra on an IR Prestige-21 spectrometer (Shimadzu, Kyoto, Japan), NMR spectra on Bruker Avance Neo 600 (Bruker, Billerica, MA, USA) and Bruker Avance 500 spectrometers (Bruker, Billerica, MA, USA), and high resolution electrospray ionisation mass spectrometry (HRESIMS) on an Agilent 6530 Accurate-Mass spectrometer (Agilent, Santa Monica, CA, USA). Normal-phase silica gel (40-50 µm, Kanto Chemical, Tokyo, Japan), C18 reverse-phase silica gel (Fuji Silysia Chemical, Aichi, Japan), and Sephadex LH-20 (Sigma-Aldrich, St. Louis, MO, USA) were employed for column chromatography. Analytical thin-layer chromatography was conducted using pre-coated silica gel 60 F254 and silica gel 60 RP-18 F254 plates (Merck, Darmstadt, Germany). The cancer cell lines were acquired from Long Island University (Brooklyn and Brookville, NY, USA). The 96-well plates were procured from Corning (Corning, NY, USA). The cytotoxic activity assay concluded with the measurement of cell absorbance using an ELISA Plate Reader (Agilent BioTek, Santa Monica, CA, USA).
Plant Material
In March 2020, whole A. triradiata plants were collected from Quang Tri province, Vietnam (16° 29′ 20.6"N 107° 00′ 33.1"E) and authenticated by Dr Nguyen The Cuong of the Institute of Ecology and Biological Resources, VAST, Vietnam. To serve as a reference, a specimen labeled AT-01 is preserved at the Faculty of Pharmacy, Hue University of Medicine and Pharmacy, Hue University, Vietnam.
Extraction and Isolation
The entire dried plant of A. triradiata (3.31 kg) was ground into powder and subjected to maceration with MeOH at room temperature. After the solvent was eliminated under vacuum, a MeOH extract weighing 550 g was acquired. This extract was then dispersed in water and fractionated continuously using hexane, ethyl acetate (EtOAc), and 1-butanol (BuOH) (3 times, each 4.0 L) to give the respective fractions.
The BuOH fraction (201.2 g) was separated on a normal phase open column by elution with CH2Cl2-MeOH-H2O (6:1:0.1, v/v) to gain 13 fractions, B1-B13. Fraction B9 (4.6 g) was subjected to partitioning on a YMC RP-18 column, using Me2CO-MeOH-H2O (1.5:1:1, v/v) as an eluent, resulting in 5 fractions, B9.1-B9.5. Fraction B9.4 (783.0 mg) was chromatographed on a silica gel column and eluted with CH2Cl2-MeOH-TFA 0.1%/H2O (5.5:1:0.1, v/v) to give 4 fractions, B9.4.1-B9.4.4. Fraction B9.4.2 (278.4 mg) was further purified by RP-18 column chromatography and elution with Me2CO-MeOH-H2O (2.7:1:1, v/v) to afford
The EtOAc fraction (92.0 g) was passed through a silica gel column using a stepwise gradient mixture of hexane-EtOAc (100:0-0:100, v/v) to give 7 fractions, E1-E7. Further chromatography of fraction E6 (16.4 g) on a silica gel column with the eluent CH2Cl2-EtOAc-MeOH (10:2:1, v/v) resulted in 12 fractions, E6.1-E6.12. By subjecting fraction E6.12 (119.5 mg) to chromatography on a RP-18 column with the eluent MeOH-H2O (3.5:1, v/v), 4 fractions, E6.12.1-E6.12.4 were yielded. Lastly, fraction E6.12.1 (42.0 mg) was fractionated by Sephadex LH-20 column chromatography and elution with MeOH-H2O (4:1, v/v) to furnish
Aspidiata E (
Determination of the Monosaccharide Composition of 1
The method of Pettolino et al.
24
with minor alterations was used to clarify the monosaccharide constituent of compound
SRB Assay for Determining Cytotoxic Activity
The SRB assay
25
was carried out to evaluate the cytotoxicity of the isolated compounds on 4 cancer cell lines. The cells were grown in Dulbecco's modified Eagle medium consisting of 1.5 g/L sodium bicarbonate, 2 mM L-glutamine, and 10% fetal bovine serum, with a media change every 48 h. The cells were separated by 0.05% (w/v) trypsin-EDTA, further cultured at a scale of 1:3 every 3 to 5 days, and maintained under a 5% CO2/atmosphere at 37 °C. After 48 h of incubation, the viable cells were seeded into 96-well plates at a density of 4 × 104 cells/well in growth medium (180 µL) and allowed to attach for 12 h. The cells were then treated with varying concentrations of test samples and incubated in the same state for 72 h. After eliminating the medium, cold 20% (w/v) trichloroacetic acid was supplemented to stabilize the remaining cells for 1 h at 4 °C. The cells were stained by adding 1X SRB solution and maintained for 30 min. The disconnected dye was eliminated by continual washing with 1% (v/v) acetic acid, while the dye bound to the cellular proteins was dissolved in 10 mM Tris base. The optical density was measured using an ELISA Plate Reader at a wavelength of 515 nm. The positive reference control was ellipticine, while 10% dimethyl sulfoxide served as the blank sample. The TableCurve 2Dv4 program was used to calculate the concentration giving 50% inhibition (IC50) to illustrate cytotoxic activity. Data were the average of 3 independent experiments. The cell inhibition rates (IR) were calculated by the following formula, in which OD0 and ODt represent the mean optical density values at time zero and day 3, respectively, while ODc represents the mean optical density value of the blank: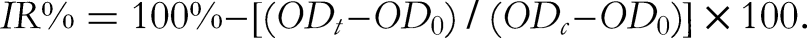
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231179412 - Supplemental material for Aspidiata E, a New Spirostanol Saponin From Aspidistra triradiata and its Cytotoxic Activity
Supplemental material, sj-docx-1-npx-10.1177_1934578X231179412 for Aspidiata E, a New Spirostanol Saponin From Aspidistra triradiata and its Cytotoxic Activity by Hien Bich Thi Le, Van Anh Thi Tran, Hien Minh Nguyen and Linh Thuy Khanh Nguyen, Duc Viet Ho, Hoai Thi Nguyen in Natural Product Communications
Footnotes
Acknowledgments
We are grateful to Mr Luong Vu Dang (Institute of Chemistry, VAST, Hanoi, Vietnam) for recording the NMR spectra.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by Hue University under Grant (ID No.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
