Abstract
The new synthetic anti-inflammatory agent, 4-(5′-dimethylamino)-naphthalenesulfonyl-2(3H)-benzoxazolone (W3D), has demonstrated definite biological activity both in vitro and in vivo. In this study, we investigated the antinociceptive effects of W3D on various animal pain models. Our results showed that W3D, at doses of 12.5 and 25 mg/kg, significantly reduced acetic acid-induced mice writhing by 65.9% and inhibited the pain response in the second phase of formalin-induced pain from 167.75 ± 6.76 s to 97.25 ± 9.63 s. Moreover, W3D prolonged the latent period on the tail-immersion test, which was stronger than the positive drug, aspirin (50 mg/kg). However, W3D did not show analgesic activity on the hot plate test, indicating no central pain response. Furthermore, the antinociceptive effects of W3D were not antagonized by naloxone, EGTA, CaCl2, and reserpine. However, L-Arginine was able to reverse the antinociceptive effect, indicating that W3D's antinociceptive effect does not depend on opioid receptors, Ca2+ concentration, or monoamine content. Instead, it is closely associated with nitric oxide (NO) content. In addition, W3D was found to alleviate nocifensive behavior and improve brain histopathology by inhibiting NO, inducible nitric oxide synthase (iNOS), and calcitonin gene-related peptide in nitroglycerin-induced migraine mice, with more than 50% inhibition ratio at only 12.5 mg/kg orally. However, noradrenaline, dopamine, and 5-hydroxytryptamine levels were not significantly altered after treatment with W3D. In conclusion, the presented data suggest that W3D can be considered a new non-steroidal anti-inflammatory and antinociceptive agent for the treatment of peripheral pain and migraine mediated by NO and iNOS.
Keywords
Introduction
Pain is a pathological manifestation of various diseases and has a significant impact on the quality of life. Pain can be mediated by inflammatory and non-inflammatory mechanisms and can arise from peripheral or central sensitization mechanisms. 1 Although opioids were considered the most effective analgesics, their clinical use was restricted due to side effects such as respiratory depression, gastrointestinal symptoms, tolerance, and dependence. Similarly, non-steroidal anti-inflammatory drugs, the most commonly used medications to alleviate inflammation, have undesirable side effects such as gastric ulcers and myocardial infarction. 2 Therefore, it is imperative to develop new antinociceptives for pain treatment.
Benzoxazolones are present in plants and exhibit a range of biological activities, including antibacterial, anti-inflammatory, antinociceptive, and anti-tumor effects. Inspired by the bioactivity of benzoxazolones, our research team conducted a series of structure modifications and bioactivity evaluations both in vitro and in vivo. Fortunately, we identified a new compound, 4-(5′-dimethylamino)-naphthalenesulfonyl-2(3H)-benzoxazolone (W3D), which demonstrated better anti-inflammatory activity against IL-6 than celecoxib with IC50 values of 8.61 and 36.04 µM, respectively. 3 Surprisingly, W3D significantly inhibited inducible nitric oxide synthase (iNOS) but not COX, suggesting a different mechanism compared to celecoxib. Given the potent anti-inflammatory activity of W3D, we hypothesized that it might also exhibit antinociceptive activity. Therefore, the antinociceptive effects of W3D in various pain models in vivo were evaluated and elucidated the underlying mechanisms associated with nitric oxide (NO) and iNOS.
Materials and Methods
Reagents and Samples
4-(5′-dimethylamino)-naphthalenesulfonyl-2(3H)-benzoxazolone (W3D) was synthesized at the Department of Pharmaceutical Science (Shanxi Medical University, China), and the structure and purity have been confirmed by ESI-MS, 1H NMR, 13C NMR, and high-performance liquid chromatography (HPLC) methods. Aspirin (Bayer Health Care Co. Ltd), Ibuprofen (Southwest Pharmaceutical Co. Ltd), and Rizatriptan benzoate (RBT) (Beijing Haiyan Pharmaceutical Co. Ltd) were used as positive control. Nitroglycerin (NTG) was obtained from Beijing Yimin Pharmaceutical Co. Ltd (Beijing, China). L-Arg and L-NAME were purchased from Dalian Meilun Biological Technology Co. Ltd. NO, iNOS, calcitonin gene-related peptide (CGRP), 5-hydroxytryptamine (5-HT), noradrenaline (NA), and dopamine (DA) assay kits were purchased from Jiangsu Enzyme Industrial Co., Ltd (Jiangsu, China).
Animals
ICR mice (20-22 g, 6-8weeks) purchased from the Experimental Animal Center, Shanxi Medical University (Shanxi, China) were used in this research. The animals were housed under standard conditions with free access to water and food for 1 week prior to the initiation of experiments. All experiments were performed in accordance with the National Institutes of Health Guide for the Care and Use of Laboratory animals.
Assessment of Analgesic Activity
Acetic Acid-Induced Writhing in Mice 4
A total of 48 ICR mice (24 females and 24 males) were randomly divided into 6 groups of 8 each. Animals were orally administrated with 0.5% sodium carboxyl methyl cellulose (CMC-Na), aspirin (12.5 mg/kg, 50 mg/kg), and W3D (3.125, 6.25, and 12.5 mg/kg), then the mice were injected intraperitoneally with acetic acid (0.6%, 10 mL/kg body weight) following the 60 min treatment period. Five minutes later, animals were observed individually to record the number of abdominal writhes for a period of 10 min.
Tail-Immersion test 4
A total of 40 ICR mice (20 females and 20 males) were randomly divided into 5 groups of 8 each. In the tail immersion test, the terminal 3 cm of animal's tail was immersed in the thermostatic water bath with a constant temperature of 55 ± 5 °C, and the time from immersion until the appearance of a jerk of tail was considered as the baseline latency. Baseline latency was tested twice and the mice with the baseline latency between 2 and 5 s was chosen to treat with 0.5% CMC-Na, aspirin (50 mg/kg), and W3D (6.25, 12.5, and 25 mg/kg) in oral, respectively. We recorded the time of tail withdrawal latency at 15, 30, 45, 60, 90, and 120 min after drug administration. 5
Formalin Induced Tonic Pain in Mice 4
A total of 40 ICR mice (20 females and 20 males) were randomly divided into 6 groups of 8 each. 0.5% CMC-Na, aspirin (50 mg/kg), and W3D (6.25, 12.5 and 25 mg/kg) were administered orally in mice; subsequently 20 μL formalin (2%) was injected into the right hind paw of a mouse. The duration of paw licking was recorded for periods of 0 to 10 min (phase 1) and 10 to 35 min (phase 2).
Hot Plate Test
Female mice were placed individually on the preheated hot-plate surface maintained at 55 ± 5 °C, and the mice showed the following withdrawal response as jumping, licking, or shaking the paw in 30 s were selected to oral administration with 0.5% CMC-Na and W3D (3.125, 6.25, and 12.5 mg/kg). The time between placement and reaction was recorded as the thermal pain threshold, and the thermal pain thresholds were measured at 30, 60, 90, and 120 min after drug administration. 6
The Affection Evaluation of Opioid Receptor/Ca2+/NO/ Monamine Transmitter on Acetic Acid-Induced Writhing
To investigate the antinociceptive mechanism of W3D, naloxone (a non-selective antagonist of opioid receptors, 4 mg/kg), L-Arg (a NO precursor, 100 mg/kg), L-NAME (a NOS inhibitor, 37.5 mg/kg) were subcutaneously injected to mice (female and male) 15 min before W3D was administrated. EGTA (a calcium inhibitor, 70 mg/kg)/CaCl2 (a calcium agonist, 30 mg/kg), and reserpine (5 mg/kg) were injected 30 min and 2 h respectively before W3D was treated. One hour later, the acetic acid-induced writhing assay was performed to determine the numbers of abdominal writhing individually. 4
NTG-Induced Migraine Mice Model
The prevalence of migraine is mostly seen in young adults, and the incidence ratio of male to female is about 1:3. 7 Female mice were divided into 7 groups of 6 mice each. All the groups were injected subcutaneously with 10 mg/kg NTG to the neck except the normal group. Thirty minutes later, the mice in positive and W3D groups were orally administrated with ibuprofen (Ibu,100 mg/kg), RBT (a selective serotonin 5-HTID receptor agonist, 1 mg/kg) and W3D (3.125, 6.25, and 12.5 mg/kg), and the normal and model groups were given equivalent volumes of 0.5% CMC-Na solution. Then the numbers of head scratching, head shaking, and face slapping with the hind paws were counted in the further 1.5 h. Two hours after NTG treatment, all the mice were anesthetized with 10% chloral hydrate, and the blood in each group was collected from the orbit. Then the mice were killed to gain the midbrain tissue and fixed with 4% paraformaldehyde for further experiments.
Hematoxylin-Eosin (H&E) Staining
Midbrain tissues fixed with 4% paraformaldehyde were performing the following process dehydration, paraffin embedding, slicing, and staining with hematoxylin and eosin to gain stained sections. After sealed with paramount, the sections were examined and imaged under a photon microscope (Nikon, Japan).
Nitric Oxide Content Assay
The collected blood was centrifuged at 3000 rpm for 15 min at 4 °C to gain the serum. The midbrain tissues were homogenated and centrifuged at 3000 rpm for 10 min to obtain the tissue supernatant. The above samples were measured the NO content using Griess method according to the instructions of the test kits (Beyotime Bio Co. Ltd). The NO levels were determined according to the calculation between NO standard curve and the absorbance of the samples at 540 nm.
ELISA Method to Determine the iNOS, CGRP, NA, DA, and 5-HT Levels
The levels of iNOS, CGRP, NA, DA, and 5-HT in serum and midbrain tissues were detected using enzyme-linked immunosorbent assay (ELISA kits, Shanghai Mibil Co. Ltd) according to the manufacturer's instructions, respectively.
Real-Time Quantitative PCR
Total RNAs in midbrain were extracted with Trizol reagent (Invitrogen, USA), of which 2 µg was inverse-transcribed to obtain cDNA. The obtained cDNA was used for the further real-time qPCR using ABI Quant Studio 3 real-time PCR System and SYBR Green reagent kit (Invitrogen, USA). The data were calculated using the 2−ΔΔCt method normalized to the individual internal control level (GAPDH). The primer sets used in mRNA qRT-PCR were as follows: GAPDH-F: GGCAAGTTCAACGGCACAGT, R- ATGACATACTCAGCACCGGG, iNOS-F: ACTCAGCCAAGCCCTCACCTAC, R- TCCAATCTCTGCCTATCCGTCTCG, CGRP-F: TGCAGGACTATATGCAGATGAA, CTTCTGAGCAGTGACACTAGAG.
Statistical Analysis
Analysis was performed using Graphpad Prism7.0 software. Statistical comparisons were assessed by one-way ANOVA. Data are represented as mean ± SD. Statistical significance was defined as P < .05.
Results
W3D Alleviated Acetic Acid-Induced Writhing
The acetic acid-induced mice writhing model is a widely used screening method for both central and peripheral antinociceptives for the treatment of acute inflammatory pain. As shown in Figure 1A, the mean number of writhes after intraperitoneal injection of acetic acid was 38.13 ± 4.17, while treatment with W3D dose-dependently reduced the number of writhes to 13.00 ± 0.71. Additionally, W3D demonstrated a 65.9% inhibition rate at 12.5 mg/kg, which was comparable to the positive drug aspirin, with a 62.3% suppression rate at 50 mg/kg. These findings indicate that W3D is effective in inhibiting the chemical stimulation that causes acute inflammatory pain through both central and peripheral mechanisms.
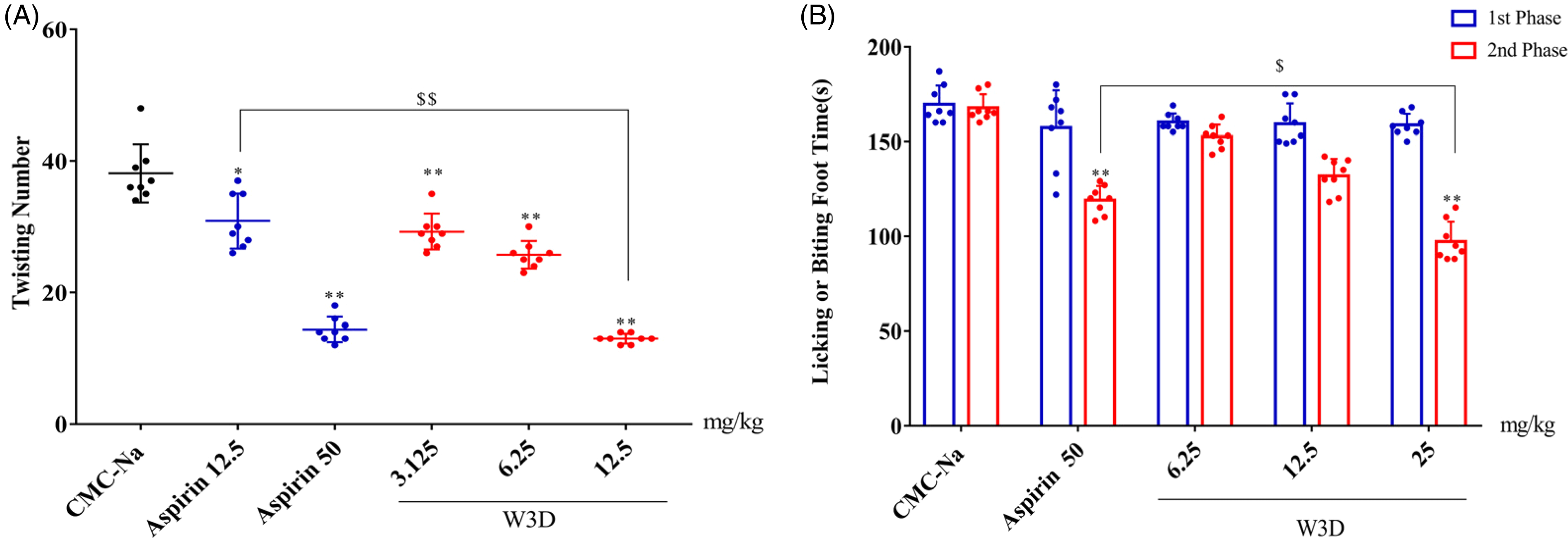
Analgesic effects of W3D on acetic acid-induced mice writhing and 2nd phase pain on formalin test. (A) acetic acid-induced mice writhing; (B) formalin test. Data were expressed as the mean ± SD (n = 8); *P < .05 and **P < .01 versus CMC-Na, $$P < .01 versus Aspirin 12.5, $P < .05 versus Aspirin 50.
W3D Prolonged the Latent Period on the Tail-Immersion Test
In the tail-immersion test, the tail withdrawal latency was significantly increased to 4.5 s after administering aspirin for 30 min. However, this effect gradually weakened over time, with the latent period reduced to 3.75 s after 90 min of aspirin treatment (Table 1). Similarly, W3D also dose-dependently prolonged the latent period. However, the analgesic effect of W3D appeared faster, 15 min after treatment, and this effect was sustained for a longer time at a dose of 25 mg/kg, compared to aspirin at 50 mg/kg.
The Antinociceptive Effect of W3D on Tail-Immersion Test.

Data were expressed as the mean ± SD (n = 8).
*P < .05, **P < .01 versus CMC-Na.
W3D Inhibited the 2nd Phase Pain Response in the Formalin Test
Formalin-induced pain is a tonic pain with a robust antinociceptive response that comprises an early phase (1st phase, 0-10 min) and a late phase (2nd phase, 10-35 min). 8 In our study (Figure 1B), the paw licking time for mice that received 0.5% CMC-Na was 169.63 ± 9.27 s in the 1st phase and 167.75 ± 6.76 s in the 2nd phase, respectively. W3D dose-dependently inhibited the pain response in the 2nd phase, particularly at a dose of 25 mg/kg, which significantly reduced the duration of the pain response to 97.25 ± 9.63 s compared to the control (P < .01). The analgesic effect of W3D (25 mg/kg) in the 2nd phase was more potent than that of aspirin at 50 mg/kg, but W3D did not exhibit any analgesic activity against neurogenic pain in the 1st phase.
W3D Showed no Analgesic Activity on the hot Plate Test
The hot plate test is used to evaluate the central pain response associated with the thermal stimulus. However, W3D did not significantly increase the thermal pain threshold when compared to the model group, indicating that W3D may not have a central analgesic effect (Table 2).
The Antinociceptive Effect of W3D on Hot Plate Test.

Data were expressed as the mean ± SD (n = 8).
NO Played a Significant Role in the Antinociceptive Action of W3D
Studies on the hot plate and formalin tests have suggested that W3D may be a peripheral analgesic that acts independently of central mechanisms. To confirm this hypothesis, we conducted an experiment on acetic acid-induced writhing in mice that were pretreated with naloxone, an opioid receptor antagonist. The results showed that treatment with naloxone did not diminish the analgesic activity of W3D, indicating that W3D is not dependent on the opioid receptor (Figure 2A).

The roles of naloxone, reserpine, Ca2 + and NO in W3D antinociceptive effects on acetic acid-induced mice writhing. Naloxone (4 mg/kg), Asp (Aspirin, 50 mg/kg), Reserpine (5 mg/kg), EGTA (70 mg/kg), CaCl2 (30 mg/kg), L-Arg (L-arginine,100 mg/kg), L-NAME (37.5 mg/kg) were given to mice intraperitoneally (i.p.) before W3D (12.5 mg/kg, i.p.). 1 h after W3D administration, the acetic acid-induced writhing test was performed. Data were expressed as the mean ± SD (n = 8); *P < .05, **P < .01 versus CMC-Na, $P < .05 versus L-NAME, &P < .05 versus W3D.
Calcium channel activation plays a critical role in transmitting pain signals to the central nervous system, and reserpine can consume monoamine transmitters to alleviate the analgesic effect. In this study, we observed that W3D had equivalent activity after 24 h of pretreatment with reserpine in acetic acid-induced writhing mice. Additionally, the mice were pretreated with EGTA/CaCl2 to investigate the involvement of Ca2+ in the analgesic effect of W3D. Our data demonstrated that neither EGTA nor CaCl2 significantly reduced the analgesic activity of W3D (Figure 2C). Therefore, the results indicate that the analgesic effect of W3D is not closely associated with the concentration of Ca2+ or monoamine.
NO is an essential information transmitter that promotes the release of pain mediators. As shown in Figure 2D, pretreatment with L-NAME significantly decreased writhing times, but this effect could be reversed significantly by the addition of L-Arg, which increases the release of NO and promotes pain response. Additionally, we found that the analgesic activity of W3D was significantly attenuated after treatment with L-arginine. This result suggests that W3D may have a similar analgesic mechanism as L-NAME, which is a NOS inhibitor.
W3D Relieved Pain Response in NTG -Induced Migraine Mice
Mice injected with NTG frequently exhibited nocifensive behaviors such as head scratching, head shaking, and face slapping with the hind paws. However, 30 min after administering W3D at a dose of 12.5 mg/kg, the aforementioned symptoms were significantly reduced by 37.04% inhibition rate, which was a significant improvement compared to the positive drug ibuprofen, which had a suppression rate of 12.97% (Figure 3). Nevertheless, the activity of W3D was lower than that of RBT, another positive drug used to alleviate migraines clinically as a 5-HTID receptor agonist (Figure 3).

Behavioral activity in NTG -induced migraine mice for the treatment of W3D. Data were expressed as the mean ± SD (n = 6); ##P < .05 versus Control, *P < .01 versus Model, **P < .05 versus Model, $P < .05 versus Ibu (30-60 min).
W3D Improved Histopathological Changes in NTG-Induced Migraine Mice
H&E staining revealed that the organizational structure of the brain was clear in the control group, with an abundance of neuronal cells and regularly arranged hippocampal pyramidal cells with clearly defined cellular boundaries (Figure 4). However, following NTG treatment, the cells exhibited irregularities, hyperchromatic nuclear pyknosis, and a blurred boundary between the cytoplasm and nucleus (indicated by the red/black/green arrow). Furthermore, W3D dose-dependently alleviated the pathological lesions, resulting in a decrease in karyopyknosis.

Effect of W3D on the histopathological changes of NTG-induced migraine mice ( × 400).
W3D Inhibited NO and iNOS in NTG-Induced Migraine Mice
An increase in NO content was considered an important indicator of NTG-induced migraine. The application of NTG resulted in an increase in NO levels in both the serum and brain tissue, as shown in Figure 5A and C. However, treatment with W3D significantly attenuated the increases in NO levels. Additionally, W3D dose-dependently downregulated the iNOS level by 46.79%, 64.0%, and 74.77% in serum, with a significant difference compared to ibuprofen by 27.27% (Figure 5B). Similar results were observed in terms of brain tissue content and mRNA expression (Figure 5E). These findings further support the notion that the inhibitory effect on iNOS might be the primary mechanism of W3D.

Inhibiting effects of W3D on NO and iNOS levels in serum and brain tissue derived from NTG-induced migraine mice. (A) NO and (B) iNOS contents in serum. (C) NO and (D) iNOS contents in brain tissue. (E) The mRNA expression of iNOS in brain. Data were expressed as the mean ± SD (n = 6). #P < .05, ##P < .01 versus Control, *P < .05, **P < .01 versus Model.
W3D Inhibited CGRP Level in NTG-Induced Migraine Mice
As a vasoactive substance, the content of CGRP in both serum and midbrain was significantly increased, as detected by ELISA. However, following administration of W3D, there was an obvious dose-dependent decrease in CGRP levels, as observed in both serum and midbrain (Figure 6A and B). This was further supported by the decrease in mRNA expression of CGRP in the midbrain (Figure 6C).

Effects of W3D on CGRP level in serum and brain tissue derived from NTG-induced migraine mice. (A) CGRP content in serum. (B) CGRP content in brain tissue. (C) mRNA expression of CGRP in brain tissue. Data were expressed as the mean ± SD (n = 6). #P < .05, ##P < .01 versus Control, *P < .05, **P < .01 versus Model.
W3D Treatment Altered NA, DA, and 5-HT Levels in NTG-Induced Migraine Mice
To evaluate the analgesic mechanism of W3D, the levels of NA, DA, and 5-HT in the midbrain were detected by ELISA. The results showed that the levels of NA, DA, and 5-HT were decreased following NTG injection. While the positive drug RBT was found to increase the levels of NA, DA, and 5-HT. W3D did not have a similar effect on these neurotransmitters. These results suggest that the therapeutic action of W3D is distinct from that of RBT in terms of its effect on NA, DA, and 5-HT levels (Figure 7).

Effects of W3D on the contents of 5-HT(A), NA(B), and DA(C) in brain of NTG-induced migraine mice. Data were expressed as the mean ± SD (n = 6). #P < .05, ##P < .01 versus Control, *P < .05, **P < .01 versus Model.
Discussion
In this study, we utilized the hot plate test to evaluate the central analgesic activity in vivo. The findings demonstrated that W3D did not exhibit antinociceptive effects through central mechanisms. The analgesic effect of W3D was not diminished by opioid receptor blocking, as shown by naloxone tests. Additionally, W3D did not affect analgesic action pretreated with reserpine to consume monoamine transmitters, indicating that the analgesic activity of W3D was not neurotransmitter dependent. The expression of neurotransmitters such as NA, DA, and 5-HT was not affected by W3D treatment in NTG-induced mice migraine experiments. Moreover, W3D was not detected in the midbrain tissue of mice using the HPLC method, indicating that it could not penetrate the blood-brain barrier for central analgesia.
The results of the second phase of the formalin and acetic acid-induced writhing tests demonstrated that W3D significantly attenuated the analgesic reaction of peripheral pain in a dose-dependent manner. Further analysis revealed that NO seemed to contribute to the antinociceptive effect. NO is a highly reactive molecule that plays a central role in various physiological functions, such as increasing vascular permeability, inducing neuroinflammation, and promoting cell apoptosis. 9 Nitric oxide synthases (NOSs), including nNOS, eNOS, and iNOS, catalyze NO biosynthesis. Ca2+-dependent nNOS and eNOS only release small amounts of NO to perform homeostatic functions, while iNOS is Ca2+-independent and produces high levels of NO when stimulated by LPS, IFN-α, IL-1β, and TNF-α. 10
Our research found that L-NAME, a NOS inhibitor, significantly reduced the number of mice writhing induced by acetic acid. However, this effect was reversed by pretreatment with the NO precursor L-arginine. Similarly, W3D exhibited a similar effect to L-NAME, suggesting that its analgesic activity was associated with NO levels. The analgesic activity of W3D was not dependent on Ca2+, as confirmed by CaCl2 and EGTA tests on acetic acid-induced writhing experiments. Therefore, we speculated that the inhibitory effect on iNOS was the primary mechanism of analgesia by W3D.
The hypothesis was tested using NTG, a NO donor, to induce a migraine model in mice and evaluate the pharmacological action of W3D on NO and iNOS. As expected, W3D was able to alleviate the headache symptoms by reducing the levels of NO in the serum and brain tissue. Furthermore, the expression level of iNOS was also reduced by W3D administration. Additionally, CGRP, the strongest vasomotor active substance that is increased in acute exacerbation of migraine, was significantly reduced after W3D treatment. Increased CGRP and iNOS in brain tissue and serum are known to cause vascular dilation and migraine attacks, according to the trigeminal theory of migraine. Therefore, it was concluded that W3D could regulate the CGRP/iNOS signal pathway to inhibit NO release and prevent migraine attacks.
Previous medical experimental and clinical reports have shown that NO and NOS inhibitors can significantly alleviate the symptoms of migraine attacks. 11 In our previous study, we demonstrated that W3D inhibited iNOS by forming 3 hydrogen bonds with the amino acid residues PRO344, VAL346, and MET368. Our findings revealed that oral administration of W3D was able to exert a peripheral analgesic effect by inhibiting iNOS and CGRP expressions, thereby preventing NTG-induced migraine-like symptoms such as head shaking and paw scratching. Therefore, W3D could potentially serve as a new anti-inflammatory drug with anti-inflammatory and analgesic activity for the relief of peripheral pain and migraine attacks through the iNOS inhibition mechanism.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was financially supported by the Shanxi Scholarship Council of China (NO. 2014-2), Shanxi Province Science Foundation for Youths (NO. 201901D211350), Shanxi Key Laboratory of Innovative Drug for the Treatment of Serious Diseases Basing on the Chronic Inflammation (NO. 2021sxcxyw07), Innovation Project of Postgraduate Education in Shanxi Province (NO. 2020SY236).
Ethical Approval
All of the experimental procedures involving animals were conducted in accordance with the Institutional Animal Care guidelines of Shanxi Medical University. The animal production license number is SCXK (Jin) 2019-0004.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
