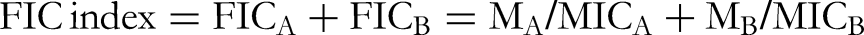Abstract
To screen and identify the antimicrobial ingredients of Forsythia suspensa leaves (FL). A screening method based on a constituent knock-out/knock-in strategy was conducted. The antimicrobial activities of FL components against seven commonly infective strains were determined using the Oxford cup method. Their antimicrobial effects when used in combination with common antibiotics were investigated by a checkerboard titration method. Two of the seven strains, Staphylococcus aureus and methicillin-resistant S. aureus (MRSA), were considered to be the most sensitive to the inhibitory effect of FL. The main antimicrobial components in FL were forsythoside I and forsythoside A. The co-administration of vancomycin with forsythoside A was found to have a significant synergistic inhibiting effect on MRSA. Some phenylethanol glycoside components play a major contribution to the antimicrobial effect of FL. This provides a reference for the research and development of FL for both medicinal and food purposes.
Forsythia suspensa, a traditional Chinese medicine (TCM) with the effect of clearing heat and detoxifying, 1 is often used for the treatment of upper respiratory tract infection and acute nephritis. 2 Research on F. suspensa has focused on its fruit, while other parts have been relatively little studied, which is a waste of resources. In recent years, F. suspensa leaves (FL) have attracted research interest, and various effects have been discovered, such as antibacterial, antioxidant, neuroprotective, anti-obesity, and improving insulin resistance.3–7 A number of compounds were also isolated from FL.8–10 However, the medicinal use and its value in food and animal feed have not been fully explored. In China, FL is considered for both medicinal and food purposes. Some teas and beverages with health benefits also use FL as their raw material. Therefore, it may have a better safety profile compared to the fruits of the plant. 11 Research in FL has good prospects.
In this study, a screening method of the TCM based on a constituent knock-out/knock-in strategy12,13 was employed to search for antimicrobial constituents in FL. Three compounds were obtained from the screening. Compounds 2 and 3 were found to contribute the most to the antimicrobial activity of FL. Afterward, compound 3 was selected and its combined effects with common antibiotics were evaluated, using the checkerboard method.14,15 The results demonstrated that the combination of compound 3 and vancomycin exhibited significant synergistic inhibition against methicillin-resistant Staphylococcus aureus (MRSA). This provides a reference for in-depth research and resource utilization of FL. The design of the study is shown in Figure 1.

Research scheme. The “p-LC” to the right of the arrow below M1 indicates “preparative liquid chromatography”.
Materials and Methods
Forsythia suspensa Leaves
FL were collected from Anze, Shanxi, China in July and were identified as the leaves of Forsythia suspensa (Thunb.) Vahl, family Oleaceae, by Prof. Liwei Zhang. All voucher specimens were deposited at the Institute of Molecular Science, Shanxi University, Taiyuan, China.
Bacterial Strains
S. aureus (ATCC 25923), MRSA (ATCC 29213), Escherichia coli (ATCC 25922), drug-resistant E. coli (ATCC 53832), Pseudomonas aeruginosa (ATCC 27853), multidrug-resistant P. aeruginosa (ATCC 18189), and Streptococcus porcinus (ATCC 9619) were provided by the clinical laboratory of Shanxi Bethune Hospital. Cell culture plates (96-well) were purchased from Shanxi cell Biotech Co. Ltd (Taiyuan, China), and iodonitrotetrazolium chloride (INT), Luria–Bertani (LB) agar medium, LB broth, and blood plates from Aobox Biotech Co. Ltd (Beijing, China). Redistilled water was used in separation and antibacterial experiments.
Antimicrobial Activity Assay
The Oxford cup method 16 was used to determine the inhibition zones of the samples against bacteria. Sample solutions (4 mg/mL) dissolved in a mixture of methanol and water (5%: 95%) were used for the inhibition activity assay against S. aureus and MRSA. Similarly handled solutions (8 mg/mL) were used for the inhibition activity assay against E. coli and other bacteria. Then, the sample solutions were sterilized by passing through a 0.22 μm filter. An appropriate amount of LB nutrient agar was completely dissolved by heating, sterilized, and placed in 90 mm dishes (20 mL/dish) after slight cooling. After the LB nutrient agar had solidified, 200 μL of bacterial suspension prepared with sterile saline (106 CFU/mL) was added and covered evenly by using an applicator stick. The Oxford cup was held perpendicular to the surface of the medium and pressed gently to bring it into close contact with the medium. Subsequently, 200 μL of sample stock solution was added and placed in a biochemical incubator (Shuli Instruments, Shanghai, China) at 37 °C for 16–18 h. The diameter of the antimicrobial zone was calculated as the average of three parallel measurements for each sample.
Minimal Inhibitory Concentration Determination
The micro-dilution method14,15 was used to determine the minimal inhibitory concentration (MIC) of drugs against S. aureus and MRSA. Forsythiaside A stock solution (2 mg/mL) and antibiotics stock solution (1 mg/mL), prepared by dissolving in LB liquid medium, were sterilized by passing through a 0.22 μm filter. Serial doubling dilutions of the antimicrobial agent in LB liquid medium to a final volume of 100 μL were placed in each well of a 96-well round-bottom microtiter plate. 10 μL of bacterial suspension (106 CFU/mL) was added to each well, shaken for 5 min, incubated at 37 °C for 24 h, and stained by INT. Three independent experiments were conducted. The growth of bacteria in each well was observed and the MIC was determined.
Assessment of Combination Effect
The antimicrobial efficacy of a selected compound in combination with either vancomycin or cefazolin was determined in a 96-well round-bottom microtiter plate by a chequerboard titration method. The selected compounds, vancomycin and cefazolin, were diluted by serial 2-fold steps vertically. The range of selected compound dilutions was from 0.03125 to 2.0 mg/mL. Concentrations of cefazolin and vancomycin were from 0.03 to 1.92 µg/mL and 0.06 to 3.84 µg/mL, respectively. The fractional inhibitory concentration (FIC) index for the combination of the two antimicrobial agents was calculated by the following equation
14
:
The results of FIC indexes (FICI) were interpreted as follows: FICI ≤ 0.5, synergy; 0.5 < FICI ≤ 1, additivity; 1 < FICI < 4, indifference; FIC ≥ 4.0, antagonism. 17 Each FICI was obtained from three independent experiments.
Extraction and Activity-Directed Separation
FL (300 g) was extracted sequentially with 3000 and 2400 mL of 40% ethanol. The two extracts were combined, filtered, and concentrated under reduced pressure to a volume of 200 mL. The concentrated solution was fractionated by medium-pressure liquid chromatography (Bonna-Agela Technologies, Tianjin, China) using D101 macroreticular absorption resin and eluted by water, and 20%, 40%, 60%, and 80% ethanol, successively. Eluents were collected, vacuum-concentrated, and freeze–dried. Five fractions of eluents were obtained and their antimicrobial activities were measured. The fraction with the most inhibiting potential was further separated by C18 column chromatography, by eluting with 30%, 40%, and 60% methanol. Three eluates of this fraction were obtained and, similarly, their antimicrobial activities were determined. The eluate with the best activity was separated by preparative liquid chromatography and five ingredients were obtained, which were evaluated for their antimicrobial activities. The components from these ingredients with good activity were further treated by Sephadex LH-20 column chromatography (40–70 μm, Amersham Pharmacia Biotech AB, Uppsala, Sweden) and compounds 1–3 were obtained.
Analytical Conditions of HPLC
Following the screening results of the antimicrobial activities, an appropriate amount of eluate obtained from the previous step was dissolved in methanol and filtered through a 0.45 µm Millipore filter. The separation was carried out using a high-performance liquid chromatograph (1260, Agilent, Santa Clara, CA, USA) equipped with an Agilent Zorbax Extend-C18 column (250 × 4.6 mm, 5 µm, Bonna-Agela Technologies). The mobile phase was methanol (A) and water (B) (38%: 62%). A flow rate of 0.8 mL/min, a detection wavelength of 235 nm, a column temperature of 25°C, and injection volume of 10 μL was applied.
Preparation of Knock-out Samples
Fifty mg of eluate was separated by semi-preparative liquid chromatography (LC-6AD, Shimadzu, Kyoto, Japan) with an Agilent Zorbax Extend-C18 column (250 × 21.2 mm, 5 µm, Bonna-Agela Technologies), eluting with a solution of methanol (A) and water (B) (38%: 62%). According to the HPLC results, different effluent fractions were collected, vacuum-concentrated, and freeze–dried, and several parts of the eluate were obtained. A knock-out sample was prepared by mixing all these parts, except the target part. The proportion in which they were mixed was determined by their weight ratio in the eluate. Then, the antimicrobial activities of the obtained knock-out samples were assayed.
Preparation of Knock-in Samples
A knock-in sample was prepared by adding a different amount of the previously knocked-out part back into the knock-out sample. Hereafter, the antimicrobial activities of the knock-in samples were measured.
Structure Identification of the Obtained Compounds
Molecular weights of three compounds were determined by LC-MS (Waters HPLC-Thermo Finnigan LCQ Advantage ion trap mass spectrometer, Milford, MA, USA). NMR spectra were obtained with either a Bruker 600 MHz or 400 MHz spectrometer, Bruker, Karlsruhe, Germany). The chemical shifts are expressed in δ values (ppm) with the coupling constants (J) in Hz.
Statistical Analysis
All data were analyzed using GraphPad Prism 9.0.0 software (GraphPad, USA). Results were presented as means ± standard deviations (SD). Statistical differences between groups were analyzed by t-test. P < 0.05 indicated that the difference has a statistical significance.
Results and Discussion
Screening of Antimicrobial fractions
The antimicrobial activities of the different eluted fractions E1–E5 of FL separated by D101 macroporous adsorbent resin were determined by the Oxford cup method. From the diameters of the inhibition zones of the eluted fractions (Table 1), E2 and E3 showed strong inhibitory effects on S. aureus and MRSA. In terms of the diameters of the inhibition zones of the eluted fractions (Table 1), E2 and E3 showed strong inhibition against S. aureus and MRSA. All fractions showed weak inhibition of E. coli, P. aeruginosa, and multidrug-resistant P. aeruginosa. No inhibitory effect was observed against S. pneumoniae and drug-resistant E. coli. Taken together, E3 was the most effective fraction, and so it was selected for the next step of C18 column separation, which yielded M1–M3. The results (Table 1) revealed that M1 had the highest antimicrobial activity. Therefore, further activity-directed separation started from M1, and subsequent activity evaluation was based on the strength of inhibition of S. aureus and MRSA.
Inhibitory Effects of Different Fractions (Inhibition Zones, mm, n = 3).

A, S. aureus; B, MRSA; C, E. coli; D, drug-resistant E. coli; E, P. aeruginosa; F, multidrug-resistant P. aeruginosa; G, S. pneumonia. E1–E5, five fractions of FL extract; M1–M3, three fractions of E3; “—” means no inhibition effect. *P < 0.05, **P < 0.01. E1, E2, E4, and E5 compared with E3, M2 compared with M1. Those in bold indicate their relative best results in that step of activity screening.
Screening of Antimicrobial Components
Antimicrobial activities of five components, I1–I5, isolated by preparative liquid chromatography from M1, were measured by the Oxford cup method. From the diameters of the inhibition zones of the different components (Table 2), I2 and I4 displayed stronger inhibition against S. aureus and MRSA. Therefore, I2 and I4 were considered to be the main antimicrobial components. Compounds 1 and 2 were obtained from I2. Compound 3 was obtained from I4. Compound 1 did not show any inhibitory effect, but compounds 2 and 3 presented strong inhibition against S. aureus and MRSA. Meanwhile, the activity of compound 3 was stronger than that of compound 2.
Inhibitory Effects of Different Components (Inhibition Zones, mm, n = 3).

A, S. aureus; B, MRSA; I1–I5, five components isolated from M1; “—” means no inhibition effect. * P < 0.05, ** P < 0.01 compared with I4. The group indicated in bold is the one with the relatively best results in this step of activity screening.
Validation Using Knock-out and Knock-in Samples
According to the aforementioned results, M1 was in the middle of the activity-directed separation process, carrying the top and bottom. Therefore, in addition to more refined isolation, knock-out and knock-in evaluation was performed on M1. I2 and I4 were then selected as the knocked-out and knocked-in components.
As shown in Table 3, the antimicrobial capacity of M1 decreased after the removal of I2, and significantly dropped after either the removal of I4 or the removal of both I2 and I4 (P < 0.05). The results illustrated that I2 and I4 may contribute significantly to the antimicrobial activity of M1. As shown in Table 2, compound 1 coming from I2 did not show any inhibitory effect on S. aureus and MRSA. Therefore, compounds 2 and 3 may contribute the most to the antimicrobial activity of M1, which is consistent with the results in Table 2.
Inhibitory Effects of Knock-out Samples (Inhibition Zones, mm, n = 3).

A, S. aureus; B, MRSA; M1a, I2 removed from M1; M1b, I4 removed from M1; M1c, Both I2 and I4 removed from M1. *P < 0.05, **P < 0.01 compared with M1. The group indicated in bold is the one with the relatively best results in this step of activity screening.
The antimicrobial effects of knock-in samples on S. aureus and MRSA, and rough dose-dependent trends of I2 and I4 are shown in Table 4. With the increase in the amount of either I2 or I4, the antimicrobial activity was enhanced gradually. On the basis of the above results, we concluded that compounds 2 and 3 were the key antimicrobial constituents of FL. Compound 3, with the best activity performance, was then followed by a further study of drug combinations.
Inhibitory Effects of Knock-in Samples (Inhibition Zones, mm, n = 3).

A, S. aureus; B, MRSA. The numbers in the leftmost column of the table indicates the ratio of the amount of the knocked-in components to the amount of this components originally present in M1. *P < 0.05, **P < 0.01, compared with Sample 1. Sample 1 actually corresponds to the original M1 extract, that is, the amount of the knocked-in component (I2 or I4) in this sample is the same as that in the original M1 extract. The group indicated in bold is the one with the relatively best results in this step of activity screening.
Evaluation of Efficacy of Compound 3 Combined with two Common Antibiotics Against S. aureus and MRSA
The combination effects of compound 3 with either vancomycin or cefazolin against S. aureus and MRSA are shown in Table 5. According to the results, the combination of compound 3 with vancomycin produced a significant synergistic inhibitory effect against MRSA, with a FICI of 0.5, and a 4-fold decrease in the MIC of both agents. When compound 3 was combined with cefazolin, an additive effect was shown in the inhibition against S. aureus, showing a FICI of 0.75, and a 4-fold decrease in the MIC of cefazolin. The other two combinations also exhibited additive effects.
Combination Effects of Compound 3 (Forsythoside A) with two Common Antibiotics (n = 3).

A, S. aureus; B, MRSA. The group indicated in bold is the one that works relatively best in combination with these two antibiotics.
Structure Identification of Obtained Compounds from Active Components I2 and I4
Three compounds (1–3) from the active components I2 and I4 were isolated by Sephadex LH-20 column chromatography. Compound 1 was identified as forsythenside F, compound 2 as forsythoside I, and compound 3 as forsythoside A by comparison of their spectroscopic and mass spectrometric data with those in the literature.18–20 Their NMR spectra are displayed in Figures S1–S6. The structures of compounds 1–3 are displayed in Figure 2.

Chemical structures of forsythenside F (1), forsythoside I (2), and forsythoside A (3).
Conclusion
FL is commonly consumed as a medicinal tea in China because of its health benefits. In recent years, studies such as the activity and safety of FL have been gradually carried out. 21 Similar to the fruit of Forsythia suspensa, FL is also rich in forsythosides. 22 In this study, alternate and mutual validation of activity evaluation and component separation were implemented. The results demonstrated that forsythoside I and forsythoside A were the main antimicrobial compounds of FL. In addition, synergistic and additive effects of two common antibiotics combined with forsythoside A were found. The combination of vancomycin and cefazolin with forsythoside A reduced their doses. As a result, fewer side effects and improved safety can be expected. The combination of forsythoside A with vancomycin and cefazolin could be a potential alternative for the treatment of infectious diseases associated with MRSA and S. aureus.
Antibiotic resistance is a serious medical and social problem. This study shows that the combination of herbal ingredients with antibiotics is promising. As one of the representatives with both medicinal and edible value, FL has its own local food safety standards (DBS 14/001-2017) in Shanxi province, China. The present research provides a reference for the use of FL for medicinal and dietary purposes as well as animal feed. More in-depth exploration is expected.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231163034 - Supplemental material for Screening of Antimicrobial Ingredients in Forsythia Leaves Using Target Constituent Knock-out/Knock-in Strategy and Evaluation of the Combination Effects with Antibiotics
Supplemental material, sj-docx-1-npx-10.1177_1934578X231163034 for Screening of Antimicrobial Ingredients in Forsythia Leaves Using Target Constituent Knock-out/Knock-in Strategy and Evaluation of the Combination Effects with Antibiotics by Tao Wang, YuanBo Zhang, BaiFang Wang, JianKai Tang, XueJian Guo, ShiFei Li, LingHao Qin and Liwei Zhang in Natural Product Communications
Footnotes
Acknowledgments
We would like to give special thanks to Ruixue Wang and Jianrong Rong from the Clinical Laboratory of Shanxi Bethune Hospital for generously providing the bacterial strains.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Shanxi Provincial Key Research and Development Project, Shanxi Provincial Postgraduate Education Innovation Project (grant number 201603D3114015, 2018BY036).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.