Abstract
Introduction
Melanin, which is produced by melanocytes in either the epidermis or hair follicle, is the main physiological defense against solar irradiation, such as ultraviolet light. 1 However, its excessive production and deposition can darken the skin and could even lead to hyperpigmentation and uneven skin tone. 2 Melanin synthesis in melanocytes is mainly controlled by melanogenesis-related enzymes, including tyrosinase (TYR), dopachrome tautomerase (DCT), and tyrosinase-related protein-1 (TRP-1). Tyrosinase, a copper-containing oxidase, is the rate-limiting enzyme in the catalysis of melanin synthesis. 3 Although melanin can be synthesized from tyrosine by tyrosinase, TRP-1, and DCT, microphthalmia-associated transcription factor (MITF) is the major factor regulating the expression of these enzymes. 4
Recent studies have suggested that a number of upstream activators and repressors manipulate MITF activity. They include cyclic adenosine monophosphate (cAMP)/cAMP response element-binding protein (CREB), mitogen-activated protein kinases (MAPKs), and protein kinase B (Akt)/glycogen synthase kinase 3β (GSK-3β) signaling pathways. 5 In the cAMP/CREB pathway, the phosphorylation of CREB bound to cAMP increases MITF gene expression. 6 The activation of ERK mediates the phosphorylation of MITF, leading to its degradation by the proteasome. 7 The inhibition of phosphorylated c-Jun N-terminal kinase (JNK) and p38 MAPK may regulate melanogenesis by decreasing MITF expression. 8 The Akt-mediated phosphorylation of GSK-3β decreases the binding of MITF to the tyrosinase promoter, 9 thereby reducing melanin synthesis. In addition, protein kinase C-β PKC-β is a major factor in the phosphorylation and activation of tyrosinase. 10
Most skin lightening agents in cosmetics are classified as tyrosinase inhibitors that minimize melanogenesis. 11 Although some commercial tyrosinase inhibitors (hydroquinone, kojic acid, and arbutin) can treat hyperpigmentation disorders effectively, they may exert serious side effects on the skin.12‐14 Tyrosinase inhibitors regulating melanin synthesis have always been the focus of researchers. Previous studies have also pointed out that tyrosinase inhibitors are usually similar to phenolic derivatives in structure and exhibit competitive inhibition. 15
Orcinol (3,5-dihydroxytoluene) is a phenolic compound that is found naturally in lichens and plants 16 and is commonly used to detect the presence of pentoses in Bial's test. 17 It has been evaluated for its antioxidant activity against 1,1-diphenyl-2-picrylhy-drazyl and antiradical activity. 18 Lopes et al found that 0.5 mM orcinol inhibited mushroom tyrosinase activity by approximately 49%. 19 Chen discovered that 0.2 mM orcinol significantly inhibited the activity of cellular tyrosinase by approximately 93% at 72 h. 20 Although orcinol has antioxidant and tyrosinase inhibitory abilities, the exact mechanism through which it reduces tyrosinase activity and melanin content in melanocytes has yet to be determined. In this study, we applied orcinol to identify the signaling pathways that regulate melanogenesis. Moreover, we discussed the inhibitory effect of orcinol on melanogenesis in melanocytes and determined its mechanism.
Results
Effects of Orcinol on Cell Viability, Intracellular Tyrosinase Activity, and Melanin Contents
For the determination of the cytotoxic effect of orcinol, B16F10 cells were treated with various concentrations of orcinol (Figure 1) for 72 h and assessed with the MTT assay. The results showed that compared with the control, 1 mM orcinol did not affect cell viability (Figure 2a).

Structure of orcinol.

Effect of orcinol on cell viability (a), intracellular tyrosinase activity (b), and melanin content (c). Results are presented as mean ± SD of 3 independent experiments. One-way ANOVA followed by Dunnett's T3 test was performed to test differences among each sample. * P < .05 compared with the control sample.
Therefore, the intracellular tyrosinase activity and melanin content in B16F10 cells treated with different concentrations of orcinol (0, 0.5, and 1.0 mM) were measured. Intracellular tyrosinase activity was inhibited when the orcinol concentration was 1.0 mM (Figure 2b). In addition, melanin content decreased when orcinol concentration was increased (Figure 2c). Our data showed that at concentrations above 0.5 mM, orcinol can significantly decrease melanin content. These results suggested that in B16F10 cells, the decrement in melanin content under orcinol application could be related to the decrease in intracellular tyrosinase activity.
Effects of Orcinol on the Expression of Melanogenesis-Related Genes
Previous studies showed that intracellular tyrosinase activity and melanin content could be influenced by the expression of melanogenesis-related genes. In this study, we mainly examined MITF, TYR, TRP-1, and DCT expression levels by using Western blot and RT-qPCR analyses. The results revealed that the protein levels of TYR, DCT, TRP-1, and MITF were prominently downregulated after orcinol treatment (Figure 3a). The mRNA levels of TYR, DCT, TRP-1, and MITF were also significantly decreased in orcinol-treated cells (Figure 3b). These results suggested that the inhibitory effects of orcinol on melanogenesis may be attributed to the suppression of the expression of melanogenesis-related genes.

Effect of orcinol on the expression levels of melanogenesis-related genes in B16F10 cells. (a) Western blot analysis depicting the reduction in the protein levels of TYR, TRP-1, DCT, and MITF after 24 h of treatment with different concentrations of orcinol (0, 0.5, and 1 mM). Results were normalized against β-actin expression. (b) mRNA levels of MITF, TYR, DCT, and TRP-1 in B16F10 cells analyzed after 24 h of orcinol treatment. GAPDH was used as the reaction calibrator.
Effect of Orcinol on Melanogenesis-Related Signaling Pathways
Many studies have demonstrated that melanogenesis is highly associated with several signaling pathways, including PKA/CREB, AKT/GSK-3β, and MAPKs. For the elucidation of the mechanism underlying the inhibitory effects of orcinol on melanogenesis, Western blot analysis was conducted to examine the core protein levels of the above-mentioned signaling pathways. Figure 4a shows that orcinol incubation effectively increased the phosphorylation level of ERK ½, but did not affect other signaling pathways (Supplemental Document Figure S1). Given that ERK phosphorylation enhances the degradation of MITF, ultimately leading to the inhibition of melanogenesis, 21 we further investigated the role of the ERK signaling pathway in the orcinol mediated-inhibition of melanin synthesis by using the ERK inhibitor PD98059. 22 Figure 4b illustrates that the melanin content and intracellular tyrosinase activities in B16F10 cells treated with orcinol and PD98059 were lower than those in cells treated only with PD98059. PD98059 pretreatment also decreased p-ERK1/2 levels, melanin content, and cellular tyrosinase activity (Figure 4c). Moreover, the cells treated with orcinol and PD98059 displayed increased protein levels of TYR, MITF, and p-ERK relative to those treated with PD98059. Taken together, these results indicated that in B16F10 cells, orcinol induced the phosphorylation of ERK to downregulate the protein levels of MITF, TYR, TRP-1, and DCT, thereby reducing melanin synthesis.

Effect of the ERK pathway on orcinol-induced melanogenesis. (a) Effect of orcinol on the expression levels of ERK1/2 and p-ERK1/2 in B16F10 cells treated with different concentrations of orcinol (0, 0.5, and 1 mM). (b) B16F10 cells with or without orcinol (1 mM) and/or PD98059 (10 μM) treatment for 24 h. Melanin contents and tyrosinase activity were tested. (c) Expression levels of TYR, TRP-1 DCT, MITF, ERK1/2, and p-ERK1/2 measured through Western blot analysis.
Discussion
In this study, we examined the expression of melanogenesis-related proteins and melanogenesis-associated signaling pathways to elucidate the underlying mechanism by which orcinol inhibits melanogenesis in B16F10 melanoma cells. First, we found that 0 to 1.0 mM orcinol had no significant cytotoxic effect on B16F10 cells, but decreased melanin content and cellular tyrosinase activity. These findings suggested that in B16F10 cells, orcinol could affect intracellular signal proteins to regulate melanogenesis-related proteins.
Previous studies demonstrated that MITF acts as a central transcription factor to regulate melanogenesis by activating TYR, TRP1, and DCT transcription. 23 Therefore, the inhibition of MITF expression reduced melanin production. 24 In this study, we found that orcinol decreased the levels of MITF protein, melanogenetic proteins, and melanin. These results indicated that orcinol decreased melanogenesis by downregulating MITF expression.
MITF expression levels can be regulated by several signaling pathways. 25 In this study, although we measured several signaling pathways (PKA/CREB, AKT/GSK-3β, JNK, ERK, p38 MAPK, and PKC-β), we found that only the ERK pathway was affected under orcinol application. Several findings suggested that the ERK pathway can regulate MITF expression to control melanin production.11,26 The activation of ERK can induce the degradation of MITF, leading to tyrosinase inhibition and reduction in melanin production.7,27 Similar to prior studies,28,29 our study found that orcinol induced ERK phosphorylation to downregulate MITF expression. Through the application of an ERK-specific inhibitor (PD98059), we verified that orcinol activated ERK pathways, leading to the reduction in MITF protein and melanin contents in B16F10 cells.
Conclusions
The results of comprehensive experiments indicated that orcinol reduced melanogenesis by decreasing the expression of the MITF protein via ERK activation (Figure 5). The results of this work provide evidence supporting the beneficial effects of orcinol on the inhibition of melanin synthesis. Therefore, orcinol is a potential effective inhibitor of pigment production that can be used in whitening cosmetics.
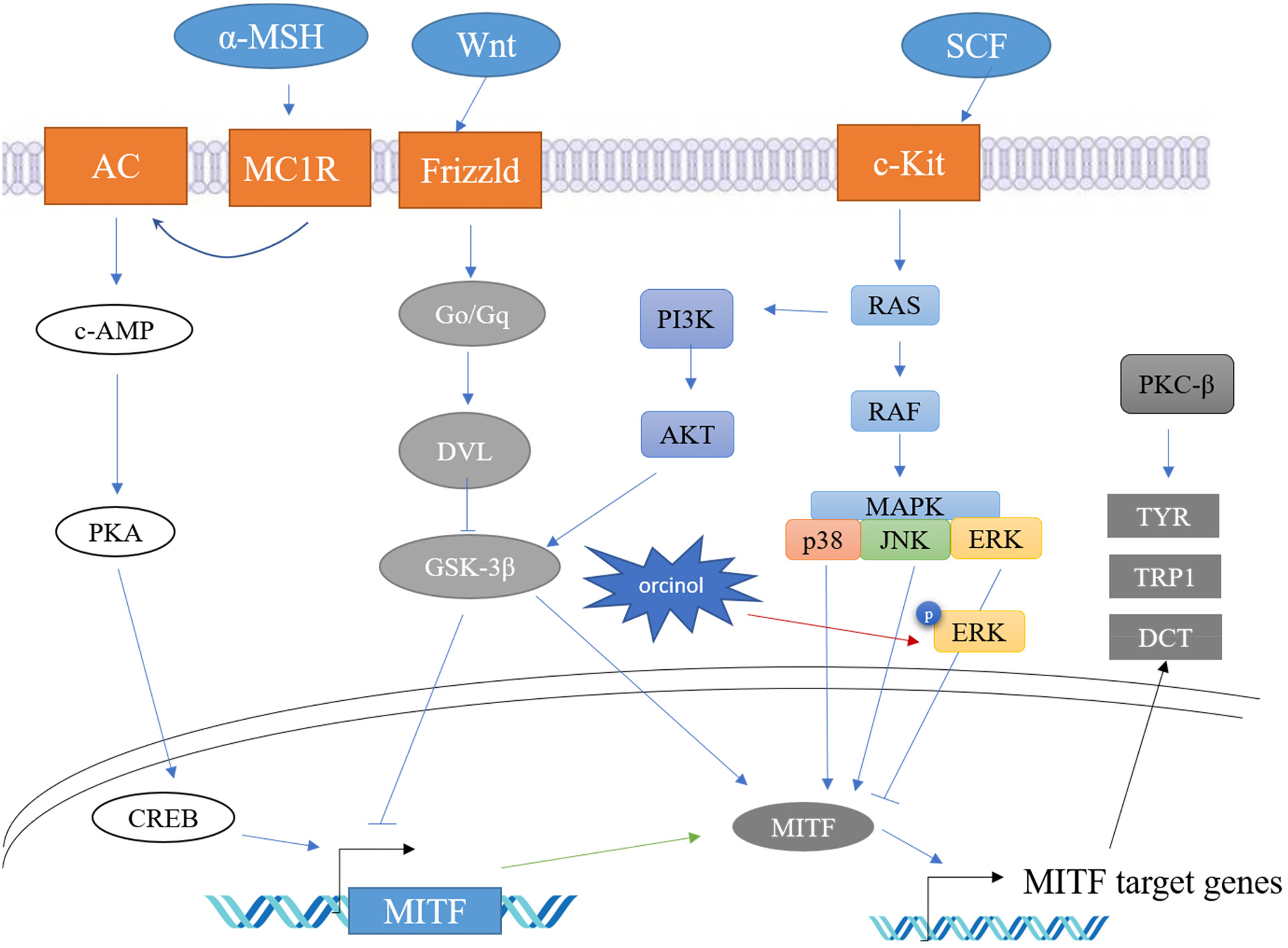
Proposed mechanism of the effect of orcinol on melanogenesis in B16F10 melanoma cells. Orcinol upregulates ERK1/2 phosphorylation to reduce MITF protein expression, resulting in the inhibition of TYR, TRP1, and DCT expression.
Materials and Methods
Reagents
Orcinol (3,5-dihydroxytoluene), 3,4-dihydroxyl-L-phenylalanine (L-DOPA), and RIPA lysis buffer I were purchased from Sangon Biotech Co., Ltd, mushroom tyrosinase from Sigma-Aldrich Co. (St. Louis, MO, USA), antibodies specific for TYR, DCT, ERK1/2, p-ERK1/2 (T202/T185), β-Actin, JNK, p-JNK, p38, p-p38, AKT, p-AKT (S473), CREB, p-CREB (S133), GSK-3β, p-GSK-3β, PKC-β, and p-PKC-β from Company ABclonal, Inc., an antibody for TRP1 was from Abcam Co., MITF from Proteintech Co., and BeyoECL Plus from New Cell & Molecular Biotech Co., Ltd.
Cell Culture
Murine melanoma B16F10 cells were purchased from the Cell Resource Center of the Shanghai Institute for Biological Sciences (Shanghai, China). B16F10 cells were incubated in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin in a humidified atmosphere with 5% CO2 at 37 °C.
Cell Viability Assay
Cell viability was estimated by using the MTT assay. Briefly, B16F10 cells were seeded at a density of 5 × 104 cells/well in 96-well plates and incubated overnight in DMEM. The cells were incubated with various concentrations of orcinol (0.5-2.0 mM) for 72 h. The cells without orcinol treatment were set as the control. After incubation, each well was mixed with MTT (dissolved in DMEM to the concentration of 5 mg/mL) and incubated at 37 °C for 4 h. After the medium was removed, 150 μL of dimethyl sulfoxide (DMSO) was added to each well. The plate was gently shaken for 15 min. Absorbance was read at 570 nm. The absorbance of the control was considered as 100%. The results were reported as the percent change relative to the control.
Measurement of Intracellular Tyrosinase Activity and Melanin Content
B16F10 cells were incubated with different orcinol concentrations (0, 0.5, and 1.0 mM) for 24 h. The cells without orcinol were set as the control. After being washed twice with phosphate-buffered saline (PBS), the cells were lysed with RIPA lysis buffer I (Sango Biotech Inc.) on ice for 4 min. After centrifugation at 12 000 × g for 10 min, the supernatant was used to measure intracellular tyrosinase activity, and the precipitate was used to measure melanin content. The BCA assay was used to measure the protein concentration of the supernatant. A total of 50 μL of supernatant and 50 μL of L-DOPA (150 mM) were transferred to a 96-well microplate. The microplate was incubated at 37 °C for 20 min, and the absorbance was read at 475 nm. The precipitate was solubilized with 200 μL of 1 M NaOH and 10% DMSO at 100 °C for 10 min. Its absorbance was read at 475 nm. Melanin content and tyrosinase activity were calculated and corrected on the basis of the protein concentration of the supernatant. The absorbance of the control was considered as 100%. The results are reported as the percent change relative to the control.
Western Blot Analysis
B16F10 cells were incubated with different concentrations of orcinol (0, 0.5, and 1.0 mM). After 24 h, the cells were lysed with RIPA lysis buffer I (Sango Biotech Inc.) on ice for 4 min. The BCA assay was used to measure the protein concentration of the supernatant that was obtained after centrifugation at 12 000 × g for 10 min. An equal amount of each sample (10 µg) was loaded into each well of a 10% sodium dodecyl sulfate polyacrylamide gel. After running the gels, the protein was transferred from the gel to a polyvinylidene difluoride (PVDF) membrane. The PVDF membrane was blocked with 5% (w/v) bovine serum albumin solution for 1 h at room temperature. The membrane was incubated with the primary antibody overnight at 4 °C. After being washed 3 times with TBST (TBS buffer containing 0.05% Tween 20), the membrane was incubated with horseradish peroxidase-conjugated antimouse and antirabbit secondary antibodies for 1 h at room temperature. Protein bands on the PVDF membrane were detected by using NcmECL Ultra reagent and a BIO-RAD CheniDocTM XRS + Imaging System. The data of Western blot analysis were quantified with Image J 1.53 software.
Quantitative Real Time RT-RCR
B16F10 cells were treated with orcinol for 24 h then washed with PBS twice and harvested. Total RNA was isolated by using a HiFi-MMLV cDNA kit. Extracted total RNA (500 ng) was converted into cDNA by using UltraSYBR Mixture. Gene expression was analyzed by using a LightCycler® 96 System (Roche Diagnostics USA). All reactions were performed in triplicate for 45 cycles. The relative fold expression levels of melanogenetic proteins and MITF were calculated. The threshold cycle (Ct) of proteins was determined and normalized to the average of the housekeeping gene's (GAPDH) level (ΔCt). The ΔCt of orcinol-treated cells was subtracted from that of nontreated cells (ΔΔCt), and the relative quantification of gene expression was performed by using the 2−ΔΔCt method. The results were expressed as % of control. 30 The sequences of oligonucleotide primers were as follows: TYR upstream 5′-CATTTTTGATTTGAGTGTCT-3′ and downstream 5′-TGTGGTAGTCGTCTTTGTCC-3′.TRP-1 upstream 5′-GCTGCAG GAGCCTTCTTTCTC-3′ and downstream 5′-AAGACGCTGCACTGCTGGTCT-3′. DCT upstream 5′-GGATGACCGTGAGCAATGGCC-3′ and downstream 5′-CGGT TGTGACCAATGGGTGCC-3′. MITF upstream 5′-GTATGAACACGCACTCTCTCG-3′ and downstream 5′-CTTCTGCG CTCATACTGCTC-3′ . GADPH upstream 5′-ATGTACGTAGCCATCCAG GC-3′ and downstream 5′-AGGAAGGAAGGCTGGAAGAG-3′.
Data Analysis
The results are shown as mean ± standard deviation for the 3 independent experiments. One-way ANOVA followed by Dunnett's T3 was used to determine statistical significance. Statistical comparisons were analyzed by using Sigmaplot software 12.0; * means significantly different at P < .05.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X231156704 - Supplemental material for Orcinol Inhibits Melanogenesis in B16F10 Cells via the Upregulation of the MAPK/ERK Signaling Pathway
Supplemental material, sj-docx-1-npx-10.1177_1934578X231156704 for Orcinol Inhibits Melanogenesis in B16F10 Cells via the Upregulation of the MAPK/ERK Signaling Pathway by Chih-Li Yu, Hongtan Wu, Yu-Pei Chen, Fangfang Chen and Guey-Horng Wang in Natural Product Communications
Footnotes
Data Availability Statement
Data from this investigation are available in the manuscript and Supplemental material.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable to this work.
Funding
The author(s) disclose the receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by Fujian Provincial Health Commission (grant number 2019-CXB-27).
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
