Abstract
Multiple myeloma is an incurable hematological cancer with an average survival time of only 5 to 10 years due to the high rate of relapse and resistance. Although the introduction of novel classes of drugs has improved the standard of care, new strategies are urgently needed. Plants are primary sources of bioactive natural compounds for cancer treatment. This study aimed at evaluating the in vitro inhibitory activity of the Inga edulis seed extract against multiple myeloma cells, as well as the compounds isolated from this extract. The ethanolic extract of the seeds of I. edulis inhibited 85% of RPMI 8226 cell proliferation at 20 µg/mL. Then, the extract was subjected to a bioassay-guided fractionation to afford 3 saponins known as concinnoside D (
Introduction
Multiple myeloma (MM) is a hematological disease characterized by uncontrolled proliferation of monoclonal plasma cells within the bone marrow. This leads to the accumulation of abnormal antibodies in the serum and/or urine and multiorgan dysfunctions.1,2 MM accounts for 10% of all hematological malignancies, and it still remains incurable in patients who relapse or become resistant to conventional regimens.3,4 However, in the last few decades, the clinical use of novel treatment options showed considerably marked progress in the longevity of MM patients with a median survival of over 5 to 10 years. 5 These treatments mainly consist of proteasome inhibitors, immunomodulatory drugs, histone deacetylase inhibitors, monoclonal antibodies, and exportin-1 inhibitors. 5 In the search for compounds active against MM, phytochemicals remain important players and are being extensively studied both as single agents and in combinations with other drugs.
Inga sp., Fabaceae, is native to tropical America, widely distributed in South and Central America, and common in regions near rivers and lakes.6‐9 The fruits are edible, cylindrical, and long, displaying a greenish color, while their seeds are black, and coated with the white pulp of the fruit.6,9,10 In popular medicine, the fruits of Inga sp. are used as an antidiarrheal,6,11 while the seeds are used to treat cancer.7,11 Such benefits have been related to the high antioxidant activity due to anthocyanins, flavonoids, 12 phenolic acids, catechin, 11 trypsin inhibitors,13,14 and saponins. 15 I. edulis Martius is the most widespread Inga species.12,16 However, its fruits are not widely cultivated and explored, which implies that there are only a small number of studies available in the literature on I. edulis.17‐19 Therefore, a detailed phytochemical investigation, as well as an evaluation of the antioxidant and antiproliferative activities of the seeds of I. edulis, was performed to assess its potential use.
Materials and Methods
General Experimental Procedures
NMR spectra were recorded on a Bruker Avance III HD 600 MHz NMR spectrometer equipped with a QCI 5 mm Cryoprobe and a SampleJet automated sample changer (Bruker BioSpin). Chemical shifts are reported in parts per million (δ) using the residual dimethyl sulfoxide (DMSO)-d6 signals (δH 2.50; δC 39.5) as internal standards for 1H and 13C NMR, respectively. HRMS was performed on a Q Exactive Focus Hybrid quadrupole-orbitrap mass spectrometer (Thermo Scientific) using electrospray in the positive mode. The spray voltage was set to 3.5 kV; the sheath gas flow rate (N2) to 50 units; the capillary temperature to 320 °C; the S lens RF level to 50; and the probe heater temperature to 425 °C. UHPLC was performed on an Acquity UPLC I-class System (Waters). The separation was performed on an Acquity BEH C18 UPLC column (50 × 2.1 mm i.d.; 1.7 μm, Waters), using a gradient (H2O and MeCN both containing 0.1% formic acid) of 5% to 98% MeCN in 6 min, followed by a washing step with 98% MeCN for 1 min. After the washing step, the column was equilibrated with 5% MeCN for 2 min before the next injection. The flow rate was set to 0.6 mL/min, the temperature to 40 °C, and the injection volume was 1 μL. Flash chromatography was performed on an Armen Spot preparative chromatographic system (Interchim) equipped with a quaternary pump, a UV detector, and a fraction collector. The flow rate was set to 40 mL/min, and UV absorbance was at 220 nm. Reverse phase semipreparative chromatography was performed on the same Armen Spot preparative chromatographic system as for flash chromatography. The flow rate was set to 20 mL/min, and UV absorbance was at 220 nm.
Plant Material
The fruits of Inga edulis Martius (570 g) were collected between June and August 2018 in the state of Espirito Santo, Brazil, Piuma (20°50′04.1″S 40°43′58.1″W). The specimens were identified by the botanist Luana Silva Braucks Calazans, and a voucher specimen is in the Herbarium of the Federal University of Espirito Santo - UFES (VIES) under the number VIES-40743.
Extraction and Isolation
The pericarp was removed manually and hygienically, and the pulp was separated from the seeds. Subsequently, the seeds were subjected to drying in an oven with forced air circulation at 40 °C for 24 h. Then, the seeds (82.5 g) were pulverized in a ball mill (model MA 350; Marconi). The samples were transferred to glass bottles, protected from light, and stored at −20 °C until extraction. The pulverized material was extracted by sonication at 60 Hz in two 60 min cycles using 150 mL of EtOH. After extraction, the solvent was removed using a rotary evaporator and lyophilized for 48 h in a lyophilizer (Terroni).
The extract of the seeds of I. edulis (800 mg) was submitted to fractionation using a flash column (Claricep/120 g, 20-35 μm C18, Phenomenex) with a gradient of 35% to 100% MeOH + 0.1% formic acid in 40 min to afford 13 fractions. Fraction 7 (33.4 mg), which showed a 97% inhibition of RPMI 8226 cell proliferation at 20 µg/mL, was submitted to semipreparative chromatography to isolate the bioactive compound(s). The isolation process was carried out with an X-Select CSH C18 column (250 × 19 mm, 5 µm; Waters) using a gradient of 25% to 40% MeCN + 0.1% formic acid in 80 min and yielded the known saponins concinnoside D (
Determination of the Antioxidant Activity by the DPPH, ABTS, and FRAP Methods
The radical 2,2-diphenyl-1-picrylhydrazyl (DPPH) was used to measure the radical scavenging activity, as described previously. 20 A quercetin calibration curve was obtained with concentrations ranging from 1.6 to 100 mg/mL (R2 = 0.988), and the results were expressed in µg quercetin equivalents/g (EQ/g). The ABTS method, based on the colorimetric reduction of 2,2′-azinobis-(3-ethylbenzthiazoline-6-sulfonic acid), was performed as previously described. 21 A quercetin calibration curve was obtained for concentrations ranging from 0.8 to 100 mg/mL (R2 = 0.988). The results represent the IR50 expressed in (EQ/g).
The FRAP method is based on direct measurement of the ability of antioxidants present in the sample in reducing, in an acid medium (pH 3.6), the Fe3+ complex with trypiridiltriazine (TPTZ), to form Fe2+, which displays an intense blue color, and was performed as described previously. 22 A quercetin calibration curve was obtained for concentrations ranging from 0.8 to 100 mg/mL (R2 = 0.985). The results represent the IR50 expressed in EQ/g.
Cell Culture
The multiple myeloma human plasma cells RPMI 8226 were purchased from American Type Culture Collection (ATCC). Cells were cultured at 37 °C in a 5% CO2 atmosphere in RPMI 1640 medium (Gibco) supplemented with 50 µg/mL streptomycin sulfate, 50 U/mL penicillin G sodium salt (Gibco), and 10% fetal bovine serum (FBS) (Biowest).
Antiproliferative Assay
The antiproliferative activity of fractions and compounds was measured using XTT (2,3-bis-(2-methoxy-4-nitro-5-sulfophenyl)) (Sigma). RPMI 8226 cells were seeded in 96-well plates (Costar) at a density of 15 000 cells per well and directly treated with the various samples. Fractions were tested at 20 µg/mL, and pure compounds were tested using increasing concentrations (0.31-20 µM) for 72 h. Bortezomib (BTZ) was used as a positive control and DMSO as the vehicle control (0.2% in culture medium). At the end of the treatment time, cells were incubated with 50 µL of XTT solution (15 mg/mL) for 4 h. The absorbance was read at 450 nm using a Cytation 3 plate reader (BioTek).
The percentage of cell proliferation was measured by comparing the absorbance of the sample treated with the vehicle control wells and multiplied by 100. The IC50 value was calculated through a nonlinear regression of 7 different concentrations using GraphPad Prism 9. All the fractions or compounds were tested in triplicate in 3 independent experiments. BTZ, the positive control, displayed an IC50 value of 0.0021 ± 0.0001 µM.
Statistical Analysis
Statistical analyses were performed using GraphPad Prism 9. Data are expressed as mean ± standard deviation (SD).
Results and Discussion
The extract of I. edulis seeds inhibited 85% of RPMI 8226 cell proliferation at 20 µg/mL, which was considered active. The antioxidant capacity of I. edulis seeds showed low activity in the DPPH (IR50 = 1291 ± 52 µg EQ/g), ABTS (IR50 = 827 ± 69 µg EQ/g), and FRAP (IR50 = 487 ± 48 µg EQ/g) assays.
The seed extract was subjected to bioassay fractionation affording 13 fractions. Fraction 7 (33.4 mg), depicted by the red ellipse in Figure 1, showed a 97% inhibition of RPMI 8226 cell proliferation at 20 µg/mL.
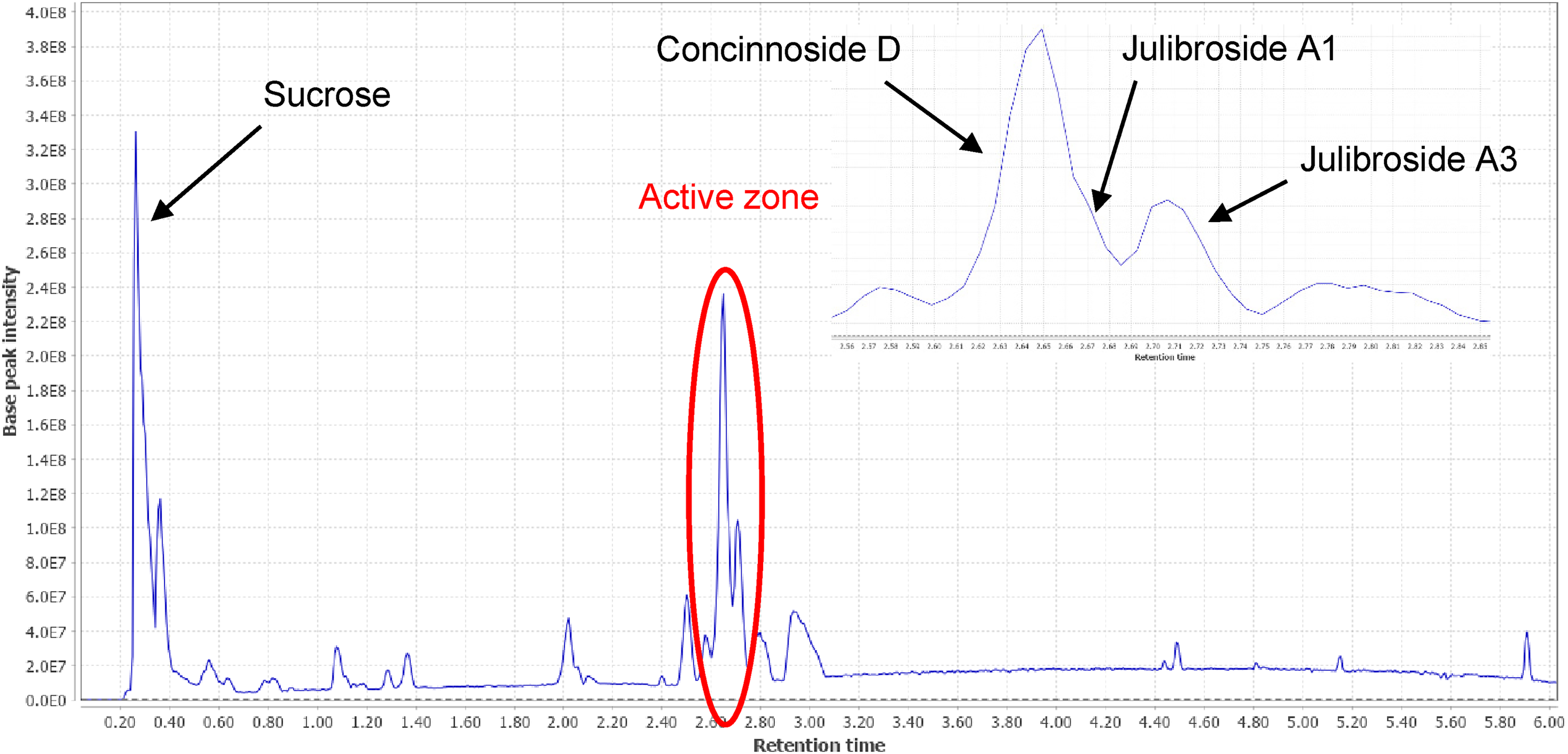
UHPLC-ESIMS chromatogram of the EtOH extract of Inga edulis.
Fraction 7 was submitted to semipreparative chromatography and yielded 3 saponins known as concinnoside D (

Chemical structures of
The 3 saponins23‐27 were previously isolated from the genus Albizia, Fabaceae, and are reported here for the first time in I. edulis. The seed extract of I. edulis showed low antioxidant activity. However, a study by Lima et al reported a moderate antioxidant activity for an aqueous fraction of I. edulis due to anthocyanins.
12
Our extract contained a high amount of sugar and the major compounds were saponins. This may explain the low antioxidant activity of our extract. More than 42 triterpenoid saponins have been reported in the Albizia genus, and several possessed cytotoxicity against various tumor cell lines.23,26,28 However, the saponins isolated in the present study were previously reported as inactive against the various cancer cell lines tested.23,26 Indeed,
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Leading House for the Latin American Region, Conselho Nacional de Desenvolvimento Científico e Tecnológico, Fundação de Amparo à Pesquisa e Inovação do Espírito Santo (grant numbers SMG1906, 310489/2019-9, 279/2021).
