Abstract
Two new abietane diterpenoids (4S, 5S, 10S)-12-(β-D-glucopyranosyl)oxy-11-hydroxyabieta-8,11,13-triene-19-oic acid β-D-glucopyranosyl-(1→6)-β-D-glucopyranosyl ester (
Introduction
Clerodendrum trichotomum Thunb., genus Clerodendrum family Verbenaceae, also called “chou wu tong” in Chinese owing to its bad smell,1,2 has been used as a folk medicine for the treatment of rheumatic arthritis, hypertension, and migraine. 3 The major constituents reported include phenylpropanoids, terpenoids, flavonoids and steroids.4–7 In earlier work,5, 7 the chemical constituents of the ethyl acetate fraction from the stems of C trichotomum have been studied systematically. However, relatively few papers have been devoted to the n-butanol extract so far.
Abietane diterpenoids are characteristic constituents of the genus Clerodendrum, which exhibit remarkable antitumor,
8
anti-inflammatory,
9
and insecticidal activities.
10
This paper focused on the study of the chemical constituents from the n-butanol extract of C trichotomum stems, and 2 new abietane diterpenoids, (4S, 5S, 10S)-12-(β-D-glucopyranosyl)oxy-11-hydroxyabieta-8,11,13-triene-19-oic acid β-D-glucopyranosyl-(1→6)-β-D-glucopyranosyl ester (

Structures of compounds
Results and Discussion
The n-butanol part of 85% ethanol extract from the stems of C trichotomum Thunb. was subjected to repeated column chromatography (CC) to yield 7 compounds
Compound
As shown in Supplemental Figure S5, there were 2 doublet methyl and 2 singlet methyl proton resonances at δH 1.09 (3H, d, J = 6.6 Hz, H-16), 1.11 (3H, d, J = 6.6 Hz, H-17), 1.30 (3H, s, H-18), and 1.20 (3H, s, H-20) in the 1H-nuclear magnetic resonance (NMR) spectrum. In addition, 3 doublet anomeric proton resonances at δH 4.30 (1H, d, J = 7.8 Hz, H-1′′′), 4.36 (1H, d, J = 7.8 Hz, H-1′), and 5.41 (1H, d, J = 7.8 Hz, H-1′′) showed the presence of 3 sugar units as a glycoside. Gas chromatography (GC) analysis (Supplemental Figure S4) showed that the sugar, after acid hydrolysis of
The 1H-1H homonuclear chemical-shift correlated spectroscopy (COSY) spectrum (Supplemental Figure S9) of

Key 1H-1H COSY, HMBC correlations for compound
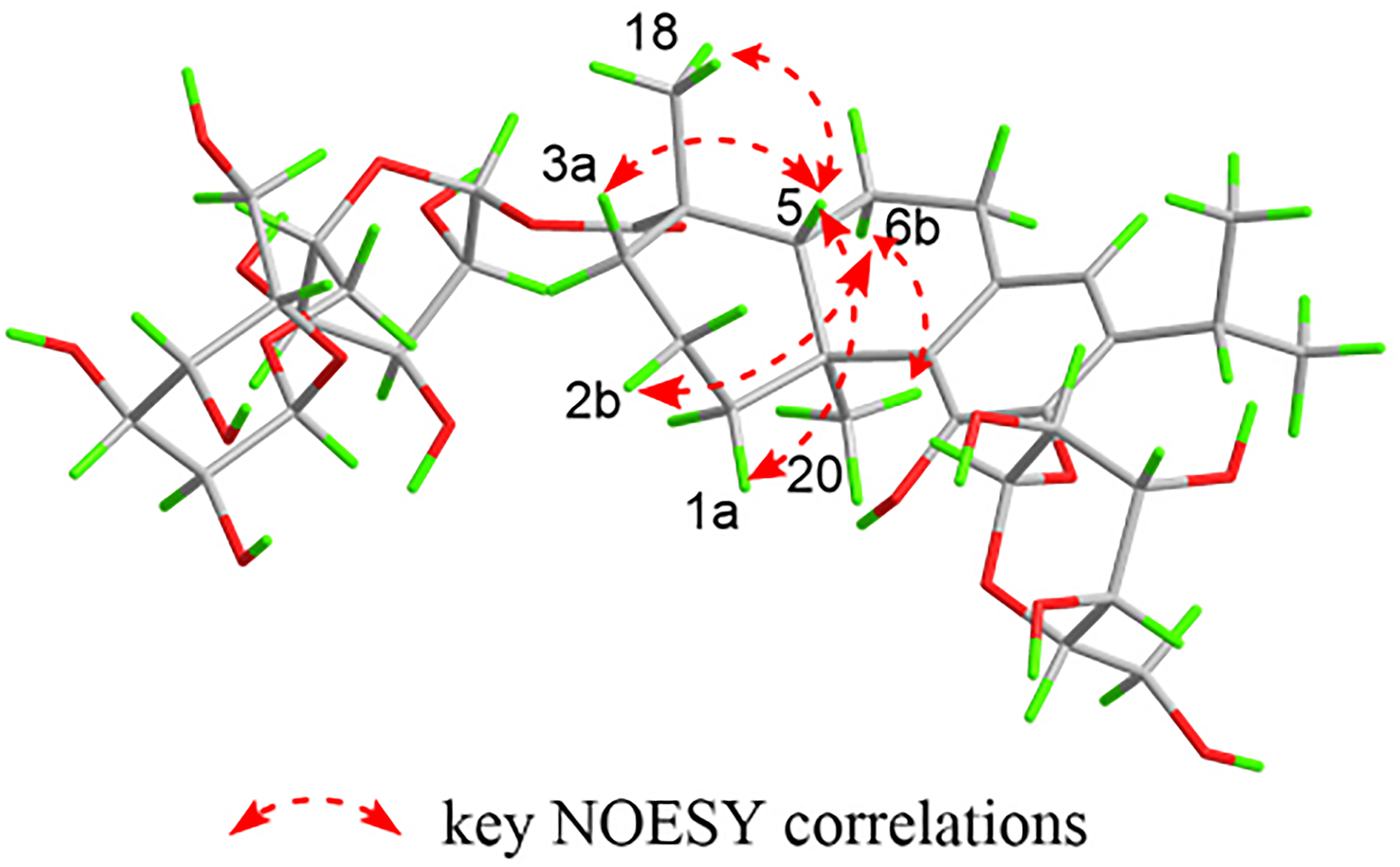
Key NOESY correlations for compound

Experimental and calculated electronic circular dichroism (ECD) spectra of compound
NMR Spectroscopic Data of Compound

Compound
In the 1H-NMR spectrum (Supplemental Figure S15), 1 doublet methyl and 3 singlet methyl proton resonances were displayed at δH 0.95 (3H, s, H-19), 1.12 (3H, s, H-18), 1.30 (3H, s, H-20), and 1.13 (3H, d, J = 7.2 Hz, H-17). In addition, 3 doublet anomeric proton resonances at δH 4.36 (1H, d, J = 7.6 Hz, H-1′), 4.46 (1H, d, J = 8.4 Hz, H-1′′), and 4.44 (1H, d, J = 7.6 Hz, H-1‴) showed the presence of 3 sugar units as a glycoside. The GC analysis spectrum (Supplemental Figure S14) showed that the sugar derivative in the acid hydrolysate of
In addition, the 1H-1H COSY spectrum (Supplemental Figure S19) of compound

Key 1H-1H COSY, HMBC correlations for compound

Key NOESY correlations for compound

Experimental and calculated electronic circular dichroism (ECD) spectra of compound
The antitumor activities of compounds
Experimental
General
HR-ESI-MS were measured on a Thermo Fisher Q Exactive-Plus mass spectrometer (Thermo Fisher Scientific), 1D and 2D NMR spectra on a BRUKER 600 NEO NMR spectrometer (Bruker Co. Ltd) and JEOL ECS 400 NMR spectrometer (Jeol), and UV spectra on an UV-2700 spectrometer (Shimadzu Co. Ltd). Optical rotation values were measured on a Bellingham-Stanley ADP 440 + polarimeter (Bellingham-Stanley Co. Ltd), and IR spectra on an IR Tracer-100 spectrophotometer (Shimadzu Co. Ltd). A JASCO J-715 spectrometer (Jasco) was used to record the ECD. GC analysis was carried out on a Shimadzu-2010 Plus gas chromatograph (Shimadzu Co. Ltd), using a ZB-5 capillary column (30 m × 0.25 mm i.d. × 0.25 μm), an flame ionization detector at 280 °C, and a column initial temperature of 200 °C. CC was performed with a macroporous resin (D101, Tianjin Haiguang Chemical Co. Ltd), silica gel (200–300 mesh and 300–400 mesh, Qingdao Haiyang chemical Co. Ltd), Toyopearl HW-40C (Tocho corporation), Toyopearl HW-40F (Tocho corporation), and Sephadex LH-20 (Pharmacia Biotech). For thin-layer chromatography, silica gel GF 254 plates were used (Qingdao Haiyang Chemical Co. Ltd).
Plant Material
C trichotomum Thunb. was collected from Nanjing, Jiangsu Province, and identified by Yao Gan researcher, Jiangsu Institute of Botany Chinese Academy of Sciences. The voucher specimens are preserved in the Engineering Research Center for the Development and Application of Ethnic Medicine and Traditional Chinese Medicine (Ministry of Education), Guizhou Medical University.
Extraction and Isolation
The stems of C trichotomum (15 kg) were extracted with 85% ethanol under reflux, and the extract was partitioned with petroleum, ethyl acetate, and n-butanol, sequentially. Three parts were obtained: the petroleum part (50 g), the EtOAc part (70 g), and the n-butanol part (470 g). The n-butanol part was dissolved in water and separated through a D101 macroporous resin, following elution in proper order by water, ethanol–water (30%, 60%, 95%, v/v).
The 60% ethanol eluted fraction was separated into 8 fractions (fr.1∼8) by octadecylsilyl silica gel (ODS) (MeOH/H2O 0:100→100:0) CC. Fr.2 was fractionated by silica gel CC eluting with EtOAc–MeOH (30:1 to 5:1) to yield compound
(4S, 5S, 10S)-12-(β-D-glucopyranosyl)oxy -11 -hydroxyabieta -8,11,13 -triene -19 -oic acid β -D -glucopyranosyl -(1→6) -
β
-D -glucopyranosyl ester (1)
Yellow solid; mp 182 to 185 °C; [α]
(3S, 5S, 10S, 15S)-3β-[β-D-glucopyranosyl-(1→6)-β-D-glucopyranosyl]oxy-12-(β-D-glucopyranosyl)oxyabieta-8,11,13-triene-11,16-diol (2)
Yellow solid; mp 174 to 176 °C; [α]
Acid Hydrolysis and Sugar Identification
Compounds
ECD Calculations
Conformational analysis of all the plausible stereoisomers of compounds
Cytotoxicity Assay
Antiproliferative activities were evaluated as IC50 against the K562 human leukemia, MCF-7 human breast cancer, A549 human lung cancer, and HepG2 human hepatoma cell lines, using the methyl thiazolyl tetrazolium (MTT) assay. The cell lines were cultured in humidified incubators (37 °C and 5% CO2). They were maintained in RPMI1640 medium (Sigma), supplemented with 10% fetal bovine serum (HyClone), 100 Unit/mL of penicillin G (Sigma), and 100 mg/mL of streptomycin (Sigma). The cells were seeded in a 96-well plate and cultured overnight, then treated with compounds
Conclusions
Two new abietane diterpenoids, (4S, 5S, 10S)-12-(β-D-glucopyranosyl)oxy-11-hydroxyabieta-8,11,13-triene-19-oic acid β-D-glucopyranosyl-(1→6)-β-D-glucopyranosyl ester (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221125053 - Supplemental material for Two New Abietane Diterpenes From the Stems of Clerodendrum trichotomum Thunb
Supplemental material, sj-docx-1-npx-10.1177_1934578X221125053 for Two New Abietane Diterpenes From the Stems of Clerodendrum trichotomum Thunb by Lin-Zhen Li, Yu Zhang, Liang Chen, Yin-Zhi Cen and Yang-Li Tu, Xiao-Sheng Yang, Yong-Jun Li in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, Science and Technology Cooperation Projects of Guizhou Province (grant numbers 81860689, and 20157359).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
