Abstract
Previously 3 sesquiterpenoids from Conocephalum conicum (L.) Dum. (Conocephalaceae) were found to modulate lymphocyte response to different stimuli, suggesting their immunomodulatory potential. Herein we evaluated the impact of low concentrations of these sesquiterpenoids on rat splenocyte viability and membrane permeability, as well as lactate dehydrogenase (LDH) activity, in order to, possibly, shed light on their mechanism of action. After a 24 h incubation of splenocytes with the sesquiterpenoids (from 10−8 to 10−6 M), MTT and trypan blue (TB) assays, as well as histochemical staining for LDH, were performed. The tested compounds were shown not to reduce the ability of cells to metabolize MTT; however, cell membrane permeability to TB was altered, suggesting that a certain percentage of cells were dead. Histochemical staining for LDH presence releveled that only 2, out of the 3 sesquiterpenoids, decreased the staining intensity, indicating either LDH leakage or its inhibition. In conclusion, having in mind the already proven modulatory potential of the tested sesquiterpenoids, the present results suggest that through the changes in the cell membrane function and leakage/inhibition of LDH in unaltered immune cells, some of the tested compounds could be considered promising candidates for further research as anticancer agents.
Introduction
The cell membrane represents a necessary unit that is associated with cell life. It enables communication of the cell, molecule transport, and some metabolic features. The main molecules of the membrane are amphiphilic, encompassing polar, hydrophilic, and apolar, hydrophobic, counterparts. 1 Under various physiological and pathological conditions, the cell membrane is exposed to stress which could arise from proteins, toxins, and mechanical activity, all forcing the cell to adapt to new conditions to survive. 2 Certain cell death mechanisms encompass cell membrane rupture, causing an inflammatory reaction and immunomodulatory effects. 2 During this stage, intracellular content is released into the surroundings, for example, extracellular fluid, plasma, and cell culture medium. One of the markers of damaged cell/cell membranes is lactate dehydrogenase (LDH), which has been used to monitor such events, even in clinical practice. 3
The trypan blue (TB) exclusion assay represents one of the first methods used to determine cell viability.4,5 This dye, with a molecular weight of ca. 1000-Da, cannot cross the cell membrane. In that sense, TB only enters cells with disrupted membranes and imparts a blue color to such cells. 6 It is well known that cell membrane integrity is one of the criteria for making the distinction between dead and living cells. 6 Thus, TB has been widely used as a vital dye, that is, it selectively penetrates the cytoplasm of dead cells, and is a very frequent assay for evaluating cytotoxicity. 6
Metastatic tumor cells are known to possess higher fluidity of the plasma membrane compared to non-metastatic cells. In that sense, restoring contact inhibition between cells through decreased membrane fluidity is effectuated by increasing rigidity at cell junctions.7,8 Hence, molecules possessing these features may cause slower cell division due to reduced membrane fluidity. Ferreira et al 9 described that the already known NSAIDs increase the fluidity of mouse splenocyte membranes in a concentration-dependent manner.
Recently, we isolated 3 new sesquiterpenoids, compounds

Structures of the tested sesquiterpenoids isolated from C conicum: (1Z,4E)-lepidoza-1(10),4-dien-14-ol (
Results and Discussion
Compounds

The percentage of viable cells was estimated using the MTT assay after 24 h of incubation with various concentrations of compounds
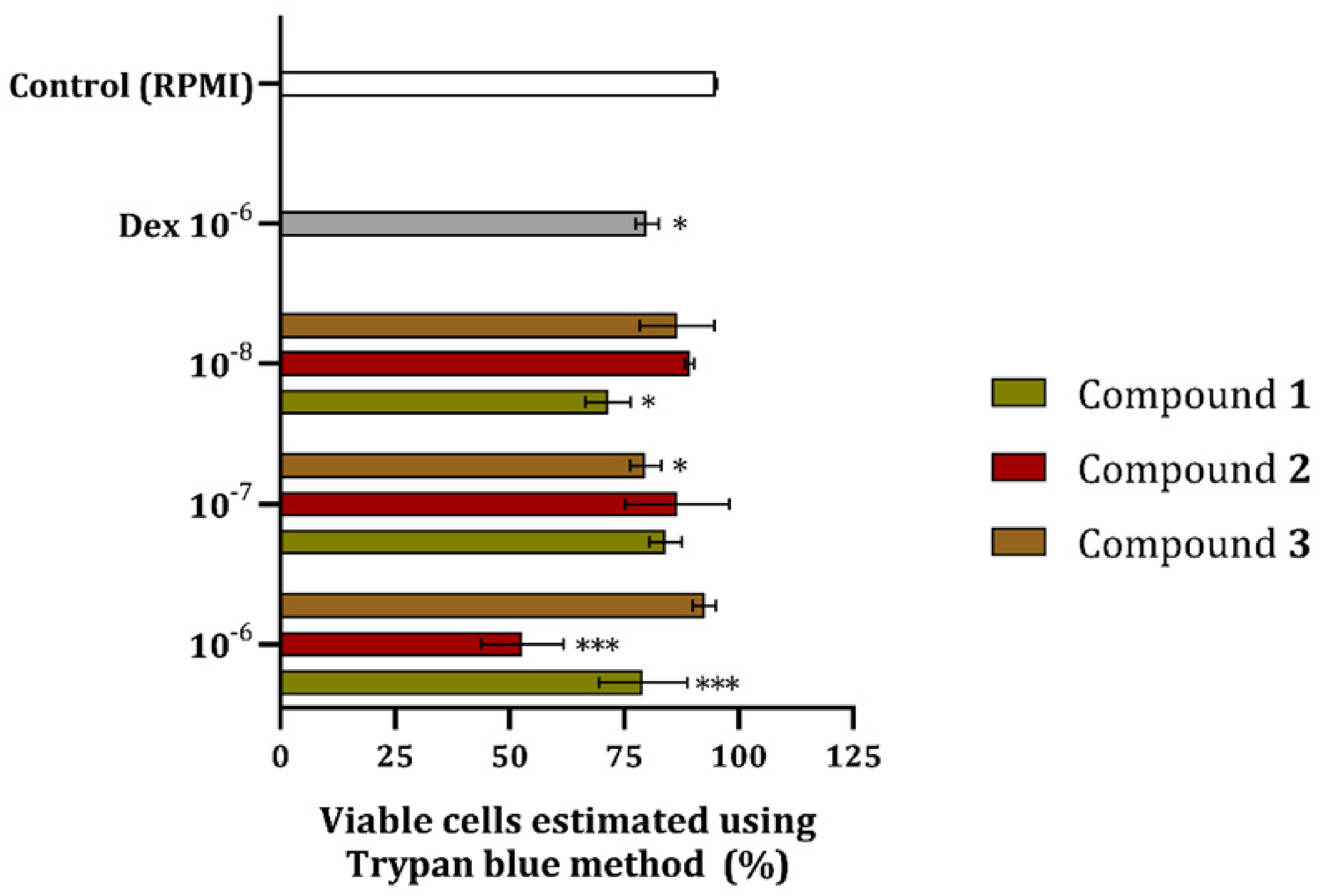
The percentage of viable cells was estimated using the Trypan blue method after 24 h incubation with various concentrations of compounds
Our previous study revealed that the investigated compounds
The results of the TB assay indicate that
When one compares the results from the MTT and TB assays, a clear discrepancy in the effects exerted by the test compounds could be seen (Figures 2 and 3), which was not the case for Dex. One of the possible explanations could be that compounds
Compared to the appearance of RPMI-treated cells, stained for LDH, the intensity of otherwise treated splenocytes was scored (Figure 4A). The application of

Microscopic appearance (magnification × 1000) of LDH stained cells obtained from the different treatments: (
The Effect of Compounds

In our study, we found that the application of Dex caused a decrease in LDH staining intensity with splenocytes (Table 1), which could arise from either decrease in LDH activity through its inhibition or a decrease in the amount of the enzyme within the cell after its leakage. In a study conducted on human chondrocytes, treatment with Dex, at concentrations of 0.5 and 1 μM, did not alter intracellular LDH activity, nor caused an increase in its leakage into the medium during 48 h of incubation. 16 On the other hand, a concentration of 0.5 μM Dex caused an increase in LDH leakage from the exposed hepatoma cells. 17 These differences might arise from different cell types treated with Dex, which include different cell membrane fluidity, seen in normal versus tumor cells. Also, splenocytes are rather sensitive to the action of corticosteroids, meaning that they might be more prone to cell death/damage, which is associated with increased membrane permeability and leakage of LDH.
A similar viewpoint as the one suggested for Dex could be transferred to the effect of the sesquiterpenoids isolated from the C conicum extract. Through an increase in membrane permeability, LDH could easily leak causing a decrease in staining intensity (Table 1). This mechanism could potentially be proposed for
In the end, our results might be viewed as interesting from the point of these compounds being potential anticancer agents since they might possess membrane damaging properties, as well as LDH inhibitory activity, all of which are desirable features of these types of drugs.18,19 Also, we should bear in mind that the earlier results showed that marchantin A isolated from liverworts is a potent inducer of apoptosis in MCF-7 human breast cancer cells. 20
Materials and Methods
Chemistry
The tested compounds were isolated in a previous study. 10 Their identity and purity were checked by 1H NMR and GC-MS, as described before, 10 prior to the performed in vitro assays. 10
Drugs and Chemicals
Cell culture medium (RPMI 1640, containing 10% fetal calf serum, and antibiotic/antimycotic solution) was acquired from Sigma-Aldrich, St. Louis, USA, dexamethasone (Dex), used as the positive control (a steroid drug with anti-inflammatory and immunosuppressant effects), from Galenika (Beograd, Serbia), and 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) and tetrasodium 3,3’-[(3,3’-dimethyl[1,1’-biphenyl]-4,4’-diyl)bis(azo)]bis[5-amino-4-hydroxynaphthalene-2,7-disulphonate] (trypan blue, TB), used for the estimation of cell viability, from Sigma-Aldrich (St. Louis, USA). All other chemicals were of analytical grade or better.
Animal Housing
Male Wistar rats (250-300 g), from the Vivarium of the Institute of Biomedical Research, Faculty of Medicine, University of Niš, maintained under standard laboratory conditions, were used in the present study. All experiments were performed following the Declaration of Helsinki and the European Community guidelines for the ethical handling of laboratory animals (EU Directive of 2010; 2010/63/EU) and the related ethics regulations of the University of Niš, Serbia (323-07-06862/2016-05/2).
Culture of Rat Splenocytes
Dissected rat spleens, cut into fine pieces, were passed through mesh cell sieves under sterile conditions. After centrifugation, the obtained splenocyte suspensions were treated with isotonic NH4Cl solution to remove red blood cells from them and washed with phosphate-buffered saline (PBS). Re-suspended cells in RPMI medium were adjusted to a 2 × 106 cells/mL density. These cells were further cultured in 96-well plates, in which the suspension was mixed with either RPMI (negative control) or compounds
Viability Assays (MTT Assay and TB Assay)
Splenocytes were incubated with compounds
TB viability assay was conducted according to an earlier described method, 11 where the cells from each well were mixed with TB solution (0.4%, w/w) in PBS (pH 7.2-7.3). Bright (viable) and dark blue (dead) cells were counted in a hemocytometer chamber under 40× lens magnification. The percentage of viable cells per 100 counted cells was reported.
Lactate Dehydrogenase Staining on Splenocyte Sections/Smears
Histochemical LDH staining was performed by incubating the cells attached to slides for 5 min at 37 °C. The staining solution was 100 mM phosphate buffer (pH 7.45), containing 17% polyvinyl alcohol, 3 mM NAD+, 150 mM sodium L-lactate, 0.32 mM 1-methoxyphenazine methosulphate, 5 mM NaN3 and 5 mM nitro-blue-tetrazolium. Control reactions were performed with the same staining solution, with the omission of sodium L-lactate and NAD+. 22 The positivity rate was represented based on LDH positive cells under 1000× lens magnification (100 cells counted). The intensity of staining was scored based on the slightly modified method described by Song et al, 23 and the staining intensity was scored as follows: 3 (strong intensity), 2 (moderate intensity), 1 (weak intensity), and 0 (negative).
Statistical Analysis
The obtained results were expressed as the mean ± standard deviation (SD). One-way analysis of variance (ANOVA), followed by Tukey’s post-hoc test for multiple comparisons (Statistics software Graphpad Prism version 5.03), was used to determine statistically significant differences. Probability values (P) less than .05 were considered to be statistically significant.
Footnotes
Acknowledgments
This study was supported by the Ministry of Education, Science and Technological Development of the Republic of Serbia (Contract numbers 451-03-68/2022-14/200124 and 451-03-68/2022-14/200113).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministarstvo Prosvete, Nauke i Tehnološkog Razvoja (grant number 451-03-68/2022-14/200124 and 451-03-68/2022-14/200).
Dedication
The authors wish to dedicate this work to the occasion of the 80th birthday of Professor Yoshinori Asakawa.
