Abstract
Stainless steel (316L SS) is commonly used to build coronary artery stents for the treatment of occluded arteries. However, tissues in contact with the stent may develop inflammation that can lead to restenosis. The natural substances derived from normal diet provide a pool of candidates that have potential to treat cardiovascular diseases. (-)-Epigallocatechin-3-O-gallate (EGCG), a polyphenolic flavonoid present in green tea, has antioxidant, antithrombogenic, and anti-inflammatory effects, and may reduce the risk of cardiovascular diseases. This study aimed to investigate whether EGCG has an anti-inflammatory effect on human umbilical vein endothelial cells (HUVECs) attached to the surface of 316L SS. We evaluated cell proliferation using the dimethyl thiazolyl tetrazolium bromide method in HUVECs after treatment with EGCG. Enzyme-linked immunosorbent assay (ELISA) assessed the level of inflammatory cytokines, including interleukin 6 (IL-6) and tumor necrosis factor (TNF) in HUVECs. We further investigated the regulatory mechanisms of the signal transducer and activator of transcription 3 (STAT3)/NF-κB signaling pathway in HUVECs by Western-blot analysis. We found that HUVECs cultured on 316L SS had increased cell proliferation and inflammation, and these can be inhibited by treatment with EGCG. EGCG reduced the secretion of IL-6 and TNF and decreased the expression of STAT3 and NF-κB in HUVECs cultured on 316L SS. Consequently, our study demonstrated that EGCG treatment ameliorates the proliferation of HUVEC when cultured with 316L SS, potentially by modulating the inflammation responses via the STAT3/NF-κB signaling pathways.
Introduction
Neointimal hyperplasia, characterized by proliferation and migration of vascular smooth muscle cells (VSMCs) to the tunica intima, is the main cause of in-stent restenosis (ISR) and has limited the long-term effectiveness of the treatment for vascular occlusions. In addition, the properties of the stent material and surface also contribute to the formation of acute thrombus and ISR. 1 The bare metal stents which are commonly used perform well with respect to mechanical stability. However, these stents increase the risk of inflammation, fibromuscular proliferation and thrombosis in the contacted-tissues, which lead to restenosis. 2 The bare metal stents are generally made from 316L SS. The hypersensitivity reaction to metals may be part of the inflammatory process and one of the triggering factors of ISR. 3 Interleukin 6 (IL-6) and tumor necrosis factor (TNF)-α are involved in the occurrence and development of ISR. 4 Recently, much progress has been made on the mechanisms of metals-induced inflammatory responses. NF-κB causes transcriptional regulation, thereby altering the expression of genes involved in cytokine release. 5
Drug-eluting stents (DESs) refer to those stents coated with synthetic polymers which can act as drug reservoirs to elute drugs over a long period of time. DESs are widely used clinically to decrease the risk of restenosis. Additionally, DES can deliver drugs that can inhibit inflammation, VSMC proliferation and thrombus formation. 6 DESs, such as paclitaxel-eluting stents and sirolimus-eluting stents, have substantially reduced the rate of restenosis. 7 Given that, chemotherapeutic solvents, such as dimethyl sulfoxide can inhibit thrombus formation, co-coating them onto paclitaxel-eluting stents has been suggested to improve the design of DESs. 8
To reduce effectively the risk of ISR, novel designs of DESs have been investigated extensively. Naturally occurring substances present in ordinary diet provide a novel source for drug development in cardiovascular therapy. (-)-Epigallocatechin-3-O-gallate (EGCG), purified from green tea, has multiple pharmacological properties and can be used to inhibit oxidant stress, cell proliferation, tissue inflammation, atherogenesis, and thrombus formation.9–12 EGCG can also benefit the cardiovascular system through its pleiotropic effects including antioxidant, antithrombogenic, and anti-inflammatory properties.13–18 Of these, the anti-inflammatory effect of EGCG suggests that it might also reduce the chance of developing ISR.
The aim of this study was to investigate the effect of EGCG on human umbilical vein endothelial cells (HUVECs) cultured on the surface of 316L SS. The proliferation, viability and inflammatory response (IL-6 and TNF) of HUVECs were tested. We also investigated the regulatory mechanisms of the signal transducer and activator of transcription 3 (STAT3)/NF-κB signaling pathway in HUVECs.
Materials and Methods
316L Stainless Steel
The composition of 316L SS was: 0.025; Cr: 17.5; Mn: 1.06; Mo: 2.66; Ni: 13.07; Si: 0.6; S: 0.008; P: 0.02 wt%, and Fe in balance. To melt 316L SS, a 25 kg vacuum induction melting furnace was used according to ASTM F138-2003. All the samples were cut from the rolled materials and machined into 32 1 mm slices, to fit into the 6-well culture plates. For cell culture, 316L SS slices were cleaned ultrasonically using ethanol and then rinsed in distilled water before sterilization at 121 °C for 20 min.
Cell Culture
The method of cell culture was the same as that described previously. 19 HUVECs used in this study were purchased from KeyGEN Biotech (KG419). Cells were maintained in RPMI-1640 medium (P004-1, Nanjing Jiancheng Bioengineering Institute) supplemented with 10% fetal bovine serum (FBS) (Gibco), 100 IU/mL penicillin and 100 mg/mL streptomycin and incubated at 37 °C in a humidified incubator with 5% CO2.
Group
HUVECs cultured on the surface of 316L SS were divided into 5 groups: 316L (without EGCG); 316L + EGCG (12.5 μmol/L); 316L + EGCG (25 μmol/L); 316L + EGCG (50 μmol/L); 316L + EGCG (100 μmol/L). Untreated cells cultured without 316L SS were used as the control group.
EGCG was purchased from Shanghai Yuanye Biotechnology Co., Ltd (S27863).
Determination of Cell Proliferation by Dimethyl Thiazolyl Tetrazolium Bromide Method
HUVECs at the exponential phase were collected and disassociated in 0.25% trypsin-EDTA. Cells were resuspended in RPMI-1640 media supplemented with 10% FBS and quantified using a hemocytometer. Cells were seeded onto 316L stainless steel-containing 6-well tissue culture plates at a density of 1 × 105 cells per well. Cell cultures were maintained in RPMI-1640 media supplemented with 10% FBS in 5% CO2 at 37 °C for 12 h until the cells were attached. Different concentrations of EGCG were added to 3 wells in parallel, respectively. For controls, 1 group only contained media without any cells, and the other group contained cells without any treatment.
Dimethyl thiazolyl tetrazolium bromide (MTT) assay was carried out on days 1, 3, and 5 postseeding. The method was the same as that previously described. 20 EGCG-treated cells were washed with PBS twice before incubation in 2 mL RPMI-1640 media and 200 μL 5 mg/mL MTT working solution (M1020, Solarbio life Science) at 37 °C for 4 h. After removing the liquid from each well, 2 mL DMSO was added to dissolve the precipitate completely and the samples were put on a shaker at low speed for 10 min. 300 μL of the samples were added into 96-well plates. The optical density was measured at 490 nm using a microplate reader and the relative growth rate of the cells was calculated.
ELISA
The levels of IL-6 and TNF in the supernatant of HUVECs were determined using a flow-based enzyme linked immunosorbent assay (ELISA) system (SEKH-0013 for IL-6 and SEKH-0047 for TNF-α, Solarbio Life Science) after 1 and 3 days of either treatment or incubation. The ELISA assay was carried out as previously mentioned. 21
Western Blot
The total proteins in HUVECs from all groups were extracted using ice-cold cell lysis buffer, as described previously. 22 Bicinchoninic assay was used to determine the concentration of total proteins in HUVECs. The proteins were separated on 12% SDS-PAGE gels for 3 h at 90 V and then transferred onto PVDF membranes for 1 h at 200 mA. The membrane was blocked for 1 h at room temperature in 3% fat-free milk dissolved in distilled water. Targeted proteins were detected using primary antibodies against NF-κB 1 (1:1000; A14754, ABclonal), NF-κB 2 (1:1000;A3108, ABclonal), STAT3 (1:1000; AP0365, Bioworld Technology Inc.), and GAPDH(1:2000, AC001, ABclonal). The membranes were incubated for 1 h at room temperature with the secondary antibody conjugated with horseradish peroxidase (#7074, CST, USA). Proteins were visualized using enhanced chemiluminescence reagents (Biosharp). Image J was used to quantify protein bands. The levels of intensity in bands were normalized to the corresponding levels of GAPDH. At least 3 independent repeats were conducted for each experiment.
Statistical Analysis
Data analysis was carried out using SPSS software. All data were displayed as means ± standard deviation (SD) (n = 3). One-way analysis of variance followed by LSD was used for statistical analysis. A P value < .05 was considered statistically significant.
Results
The Effect of Different Concentrations of EGCG on the Survival Rate of HUVEC
To investigate how EGCG may affect the survival rate of cells, we carried out an MTT experiment. After 24 h posttreatment, the survival rate of HUVECs decreased when the concentration of EGCG increased. However, there was no significant difference in the survival rate of cells treated with different doses of EGCG after either 72 h or 120 h of stimulation and there was no dose-responsive effect (P > .05, Table 1). There was no significant difference in the survival rate of HUVECs after 24 h, 72 h and 120 h of stimulation with EGCG and there was no time-responsive effect (P > .05, Table 1).
The Changes in Cell Viability Rate of Each Group in MTT Assay. (%).

Control group: HUVEC group; E1: HUVEC treatment with 12.5 μmol/L EGCG; E2: HUVEC treatment with 25 μmol/L EGCG; E3 HUVEC treatment with 50 μmol/L EGCG; E4: HUVEC treatment with 100 μmol/L EGCG. Abbreviations: MTT, dimethyl thiazolyl tetrazolium bromide; HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate.
EGCG Inhibits the Proliferation of HUVECs in Culture With 316L SS
The proliferation of HUVECs, as indicated by the MTT assay, was decreased in the 316L + 50 μmol/L EGCG group in comparison to the 316L group after 2 472 and 120 h of stimulation or incubation (P < .05, Table 2). There was no significant difference in the proliferation rate of each dose group with the increase of administration time, and there was no obvious time-effect relationship (P > .05, Table 2). In each time period, with the increase in dose, the value-added rate of each administration group decreased significantly, and the difference was statistically significant at 24, 72 and 120 h (P < .001, P < .001, P < .01, respectively).
The Changes in Cell Viability Rate of Each Group in MTT Assay. (%).
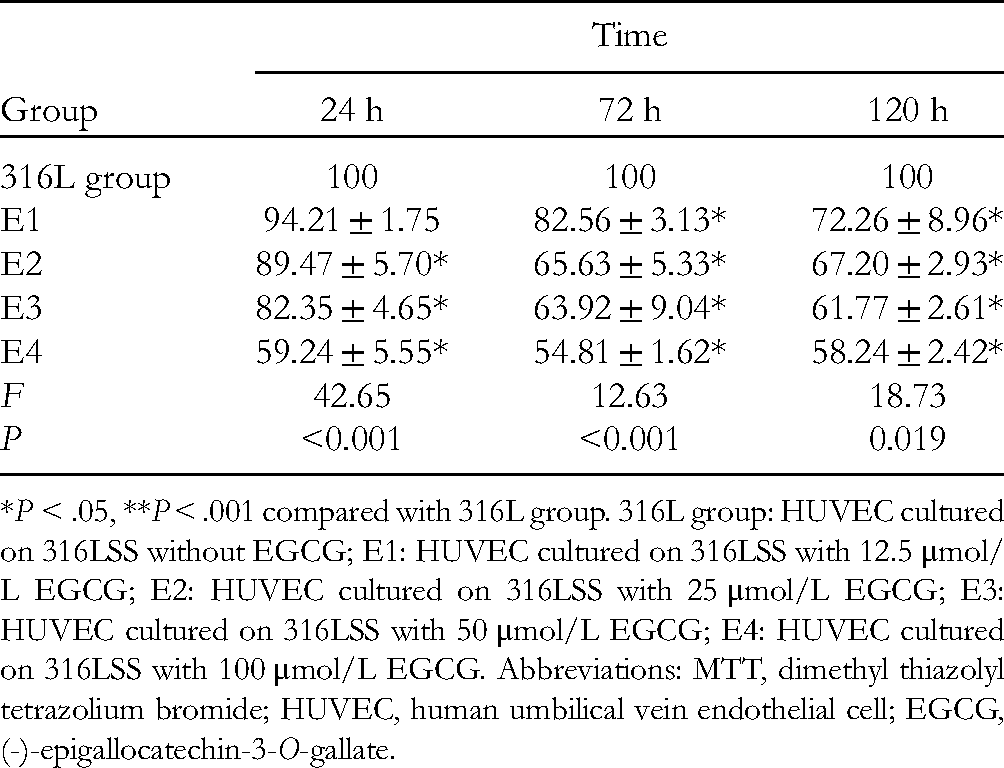
*P < .05, **P < .001 compared with 316L group. 316L group: HUVEC cultured on 316LSS without EGCG; E1: HUVEC cultured on 316LSS with 12.5 μmol/L EGCG; E2: HUVEC cultured on 316LSS with 25 μmol/L EGCG; E3: HUVEC cultured on 316LSS with 50 μmol/L EGCG; E4: HUVEC cultured on 316LSS with 100 μmol/L EGCG. Abbreviations: MTT, dimethyl thiazolyl tetrazolium bromide; HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate.
The Effect of Different Concentrations of EGCG on the Cytokine Levels of HUVEC
As shown in Table 3, the level of IL-6 was decreased in EGCG-treated HUVEC after 24 h compared with the untreated control (P < .001). After 48 h of stimulation with EGCG, the levels of IL-6 in cells treated with either 12.5 μmol/L or 50 μmol/L of EGCG were decreased compared with the untreated control. However, at this time point, the levels of IL-6 in cells treated with other concentrations of EGCG were increased compared with the untreated control (P < .001). After 72, 96 or 120 h of EGCG stimulation, the levels of IL-6 in EGCG-treated cells were decreased compared with the untreated control (P < .05).
The Effects of EGCG Treatment on IL-6 in HUVEC Cell Supernatant (pg/mL).

Control: HUVEC group. 316L group: HUVEC cultured on 316LSS without EGCG; E1: HUVEC cultured on 316LSS with 12.5 μmol/L EGCG; E2: HUVEC cultured on 316LSS with 25 μmol/L EGCG; E3: HUVEC cultured on 316LSS with 50 μmol/L EGCG; E4: HUVEC cultured on 316LSS with 100 μmol/L EGCG. Abbreviations: MTT, dimethyl thiazolyl tetrazolium bromide; HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate.
*P < 0.05 versus 316L group;
Similarly, as shown in Table 4, after 24, 48, or 72 h of stimulation with EGCG, the level of TNF in EGCG-treated cells was decreased compared with the untreated control (P < .001). After 96 h of stimulation with EGCG, only cells treated with 12.5 μmol/L EGCG secreted a higher level of TNF compared with the untreated control, whereas cells treated with other concentrations of EGCG secreted lower levels of TNF compared with the untreated control (P > .05). After 120 h of stimulation with EGCG, cells treated with all 4 doses of EGCG secreted lower levels of TNF as compared with the untreated control (P < .001).
The Effects of EGCG Treatment on TNF-α in HUVEC Cell Supernatant (pg/mL).

Control: HUVEC group ,316L group: HUVEC cultured on 316LSS without EGCG; E1: HUVEC cultured on 316LSS with 12.5 μmol/L EGCG; E2: HUVEC cultured on 316LSS with 25 μmol/L EGCG; E3: HUVEC cultured on 316LSS with 50 μmol/L EGCG; E4: HUVEC cultured on 316LSS with 100 μmol/L EGCG. Abbreviations: MTT, dimethyl thiazolyl tetrazolium bromide; HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate; TNF, tumor necrosis factor.
*P < 0.05 versus 316L group;
EGCG Inhibits the Expression of STAT3 in the HUVECs Culture With 316L SS
As shown in Figure 1, the upregulation of STAT3 protein was confirmed in the 316L group compared with the control group. The 316L + EGCG (50 μmol/L) group exhibited significantly lower STAT3 compared with 316L SS group. Meanwhile, the expression of NF-kB1 and NF-kB2 in the 316L + EGCG (50 μmol/L) group was also decreased compared with the 316L SS group.

Effect of EGCG on STAT3, NF-kB1 and NF-kB2 protein expression in HUVECs. E1 = EGCG (316L + 12.5 μmol/L);E2 = EGCG (316L + 25 μmol/L); E3 = EGCG (316L + 50 μmol/L); E4 = EGCG (316L + 100 μmol/L). Abbreviations: HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate; STAT3, signal transducer and activator of transcription 3.
Discussion and Conclusion
ISR is closely associated with inflammatory extracellular matrix deposition and the migration of endothelial and VSMCs. The recruitment and infiltration of inflammatory cells are the hallmarks of ISR. Activated by the damage in the lumen of the vessel caused by surgery and the stimulation of the stent to the vessel after surgery, monocytes, platelets, neutrophils, eosinophils, T-lymphocytes and endothelial cells produce and secret a variety of inflammatory cytokines (IL-6, TNF), leading to the migration and proliferation of VSMCs and accumulation of the extracellular matrix, and thus restenosis. Green tea, prepared from dried leaves of Camellia sinensis, is a popularly consumed beverage throughout the world. Recent studies have focused on the green tea extract, called EGCG, and explored its application in disease therapy due to its antioxidant, anti-inflammatory, and anticancer properties.23–26 In this study, we proved that EGCG inhibits proliferation of HUVECS, which was increased by 316L SS.
The inflammatory pathway can be driven by IL-6, which is a member of the gp130 family. The IL-6 receptor (IL-6R) is activated after binding to IL-6 and promotes the homodimerization of gp-130 leading to activation of the Janus kinase (JAK)/STAT and the MAPK pathway. EGCG suppressed the expression levels of IL-6 and TNF-α. 27 A previous study has indicated that EGCG inhibited the production of reactive oxygen species, TNF and IL-6 in MC-LR-stimulated HUVECs. 28 Our study demonstrates similar findings that 316L SS stimulated the HUVEC to release inflammatory cytokines (IL-6 and TNF-α), whereas the levels of cytokines were reduced after treatment with EGCG. Our study also found that 316L SS promoted the proliferation of HUVEC after 3 days of co-culturing. Moreover, we found that EGCG (50 μmol/L) inhibited the 316L SS-induced overproliferation of HUVEC. Therefore, EGCG can reduce the inflammatory stimulation of 316L SS to HUVEC and thus reduce the cell proliferation.
EGCG has also been demonstrated to regulate multiple signal transduction pathways involved in tumorigenesis. The key factors in these pathways that can be modulated by EGCG include PI3K, MAPKs (ERK, p38 MAPK, and JNK), STAT3, Smad2/3, and nuclear factor-κB.29–33 STAT3 is accumulated and phosphorylated on Tyr705 in response to pro-inflammatory cytokines, particularly IL-6. Then, nuclear translocation of phosphorylated STAT3 (pSTAT3) activates gene transcription of inflammatory and apoptotic factors. 34 IL-6 stimulation activates the associated JAKs. Activated JAKs further recruit and activate STAT3 and STAT1. 35 Wang et al 36 found that IL-6 is upregulated in colorectal carcinoma and the activation of STAT3 is enhanced. This could be the result of the sustained activation of STAT3 by IL-6, which further enhances the transcription and expression of STAT3. Therefore, the IL-6/STAT3 pathway is involved in the inflammatory reaction, and inhibition of this pathway can reduce the inflammatory response. EGCG inhibits STAT3 activation which contributes to the inhibition of keloid formation. 37 It was also reported that IL-6 and TNF-α synergistically activate STAT3 and NF-kB to promote colorectal cancer cell growth. 38 We also found that the secretion level of IL-6 and the expression level of STAT3 were increased in HUVEC stimulated with 316L SS compared with the untreated control. However, in 316L SS-stimulated cells, EGCG inhibited the secretion level of IL-6 and the expression level of STAT3 and NF-kB. Overall, EGCG inhibits the inflammatory response by inhibiting the STAT3/NF-kB pathway and thus reduces the production of pro-inflammatory cytokines, including IL-6 and TNF-α.
The findings of the present study demonstrated that EGCG treatment ameliorates the proliferation of HUVEC when cultured with 316L SS, potentially through modulating the inflammation responses via the STAT3/ NF-κB signaling pathways (Figure 2). We demonstrated that EGCG can serve as a novel therapeutic option for developing DESs.
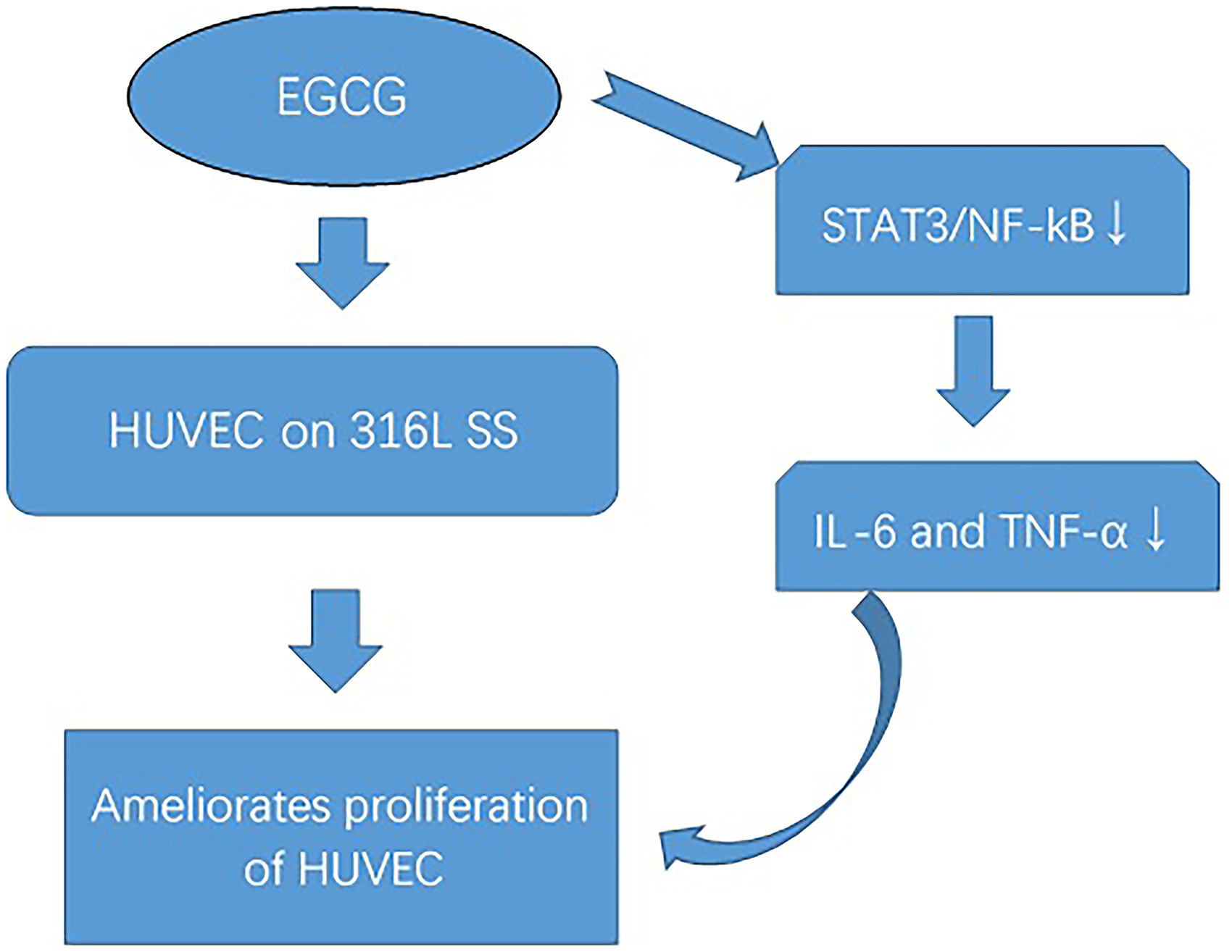
A scheme of the possible biological pathways. EGCG treatment ameliorates proliferation of HUVEC when cultured with 316L SS, potentially through modulating the inflammation responses via the STAT3/NF-κB signaling pathways. Abbreviations: HUVEC, human umbilical vein endothelial cell; EGCG, (-)-epigallocatechin-3-O-gallate; STAT3, signal transducer and activator of transcription 3.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the the Education Department of Jilin Province, Jilin Province Development and Reform Commission, National Natural Science Foundation of China, the Health and Family Planning Commission of Jilin Province, the Development of Science and Technology, Jilin Province (grant numbers JJKH20180239KJ, 2020C038-7, 81401721, 2017J074, 2018010195JC). Beijing Key Laboratory of environmental toxicology, Capital Medical University (2021hjdI03).
