Abstract
Piceatannol is a stilbenoid, which has shown bioactivities in various cell culture models. However, its stability in cell culture medium is not clear. Here, UPLC-MS/MS was applied in situ to analyze the degradation products of piceatannol in Dulbecco's Modified Eagle's Medium (DMEM) and cell culture to investigate the compound's stability in DMEM. During the incubation with cell culture medium (at 4 and 37 °C), several piceatannol derivatives, such as an oxidation product (
Introduction
Piceatannol (
Results
Stability of Piceatannol in Dimethyl Sulfoxide and Methanol
Piceatannol was found to be stable in dimethyl sulfoxide (DMSO) at 37, 4 and −20°C for 2 weeks. As expected, the higher the storage temperature, the more unstable was the piceatannol. Piceatannol was more unstable when kept in methanol than in DMSO. Derivatives of piceatannol with a

TIC chromatogram of piceatannol in DMSO and methanol at different temperatures. (A) Mass range 80.00 to 1500.00 (Max relative abundance: 15 000 000). (B) Mass range 245.00 to 245.10 (Max relative abundance: 5000 000). Abbreviations: TIC, total ion current; DMSO, dimethyl sulfoxide.
the Stability of Piceatannol in DMEM
An in situ analysis was performed to determine the stability of piceatannol in DMEM at 37 °C. The samples were automatically injected into the UPLC-MS-MS system every 35 min. The changes are shown in Figure 2. The peak at
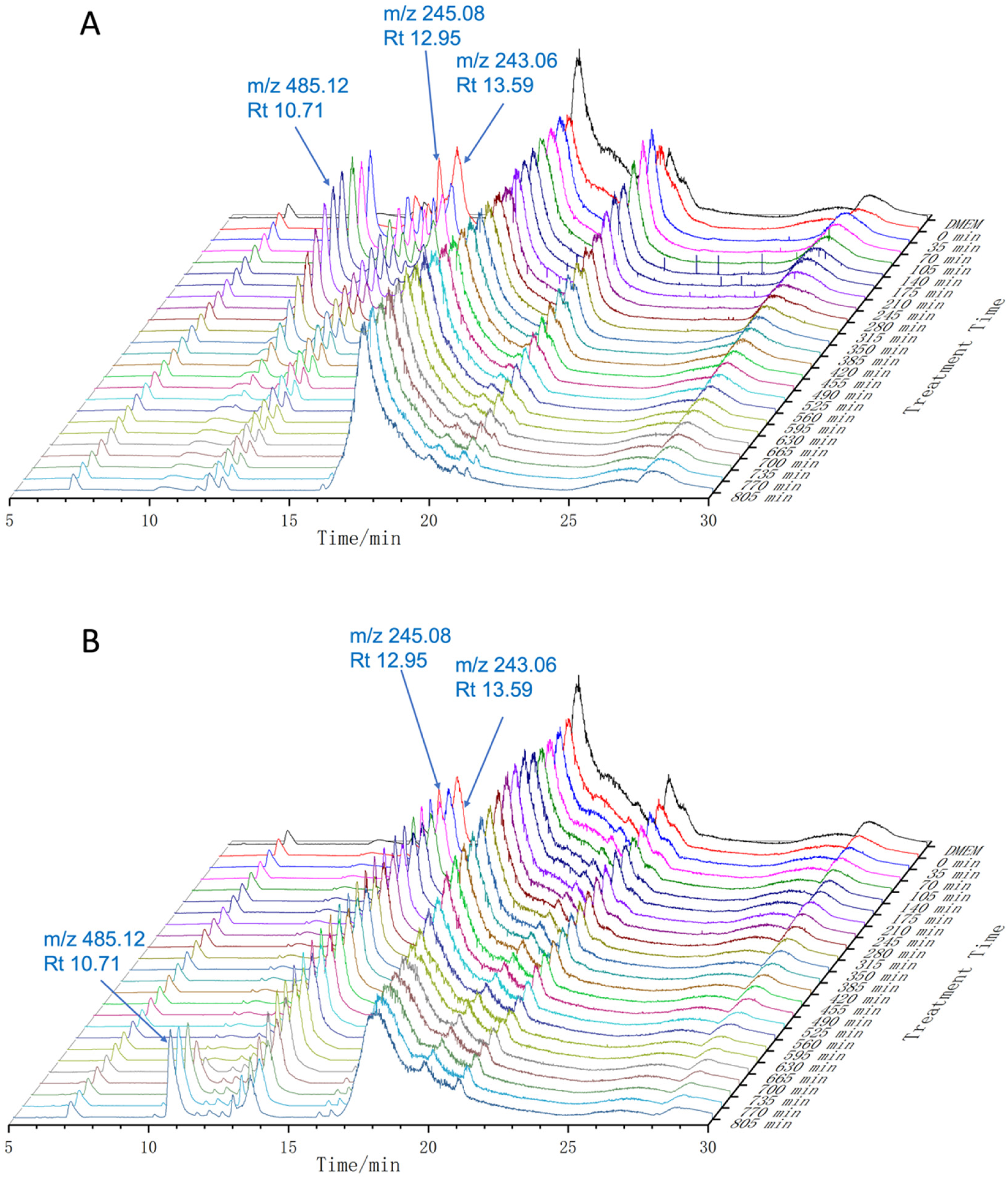
TIC chromatogram (mass range 80.00-1500.00) of piceatannol in DMEM at 37 °C in the absence (A) and presence (B) of ascorbic acid. Max relative abundance 30 000 000. Abbreviations: TIC, total ion current; DMEM, Dulbecco’s Modified Eagle’s Medium.

TIC chromatogram of piceatannol in DMEM and blank DMEM at 37 °C in the absence (A, C, E, G, I, K) and presence (B, D, F, H, J, L) of ascorbic acid. Abbreviations: TIC, total ion current; DMEM, Dulbecco’s Modified Eagle’s Medium.
The Appearance and Disappearance Time Points of Piceatannol Derivatives in Dulbecco’s Modified Eagle’s Medium (DMEM) Medium.

Effect of Ascorbic Acid on the Stability of Piceatannol in DMEM
In the presence of ascorbic acid, the peak of piceatannol (
The peak at
The intensity of peaks at
Characterization of New Products of Piceatannol in DMEM
Several new peaks appeared due to the degradation of piceatannol in DMEM at 37 °C. These derivatives were grouped by their molecular ion peaks [M + H] at
Comparison of Actual MS2 and Predicted MS2 of

Compound A (m/z 243.06)
Compound A (

The MS2 and predicted structure of (A) compound A (
Compound B (m/z 247.09)
The peaks of compound B (
Compound C (m/z 485.12)
The peaks of compound C (

The structure of piceatannol and the predicted structures of piceatannol derivatives.
Dimers of Piceatannol (m/z 487.14)
The peaks at
Comparison of Actual MS2 and Predicted MS2 of

There were several ways for piceatannol to form a dimer by losing 2 hydrogen atoms. Because the oxidized piceatannol is dehydrogenated and rearranged to different radicals, the coupling of different piceatannol radicals would form dimers. An 8-
Trimers of Piceatannol (m/z 727.18)
The peaks at
Comparison of Actual MS2 and Predicted MS2 of

Previous studies have shown that piceatannol derivatives could have multiple biological activities. Eight piceatannol dimers synthesized through biotransformation of piceatannol showed their potential for α-glucosidase inhibition activity. 15 Scirpusin B, a dimer of piceatannol, was reported to show a better vasorelaxant effect in rat thoracic aorta than piceatannol via NO derived from the endothelium. 18 Cyperusphenol D, a trimer of piceatannol, was found to have an effect in suppressing the growth of human T-cell leukemia Jurkat cells. 19
Conclusions
As there must be a relatively long time between sample collection and UPLC-MS/MS analysis, the stability of piceatannol was determined when it was kept in different solutions at different temperatures for 2 weeks. The result proved that the storage temperature and solution could affect the stability of piceatannol through long-term storage, and the importance of sample storage for the biological study is beyond doubt.
Since most cell culture experiments are performed at 37 °C, the stability of piceatannol was studied in DMEM at 37 °C; piceatannol was unstable under these conditions. Previous studies showed that other polyphenols, such as quercetin and EGCG, were unstable in the cell culture medium.20,21 It is interesting to note that in most of the cell culture studies, the activity might not be due to the compound added to the cell culture system, but to its degradation products. As piceatannol is a potent antioxidant, it can be oxidized easily, especially at higher temperatures. The result from the current study showed that piceatannol might be oxidized to a quinone form or multiple piceatannol molecules might form different types of dimers or trimers (Table 5). Earlier studies have shown that dimers and trimers of piceatannol could have multiple biological activities.
The Source of Predicated Structure of Piceatannol Derivatives.

Methods and Materials
Chemicals
Piceatannol (98%) was obtained from Macklin Bio Chem Technology Co. Ltd. HPLC grade methanol, acetonitrile, acetic acid (>99%, AR) and DMSO were purchased from Merck, hydrochloric acid (34%-37%, AR) from Fisher Scientific, DMEM high glucose liquid medium from Life Technologies (Gibco BRL), and fetal bovine serum, trypsin-EDTA, penicillin-streptomycin from Professional Health Trading Co. Ltd. Deionized water was prepared using a Milli Q Integral water purification system (Millipore). Ascorbic acid was provided by Aladdin Industrial Co. Ltd.
Stability of Piceatannol in DMSO and Methanol
Piceatannol was dissolved in either DMSO or methanol to obtain a solution of 5 mM in autosampler vials. The solutions were placed at −20, 4 and 37 °C for 2 weeks, respectively. Before analysis, the solutions were diluted to a concentration of 0.5 mM with methanol. An aliquot (5 μL) was automatically injected into a Thermo Scientific LTQ Orbitrap XL hybrid FT Mass Spectrometer.
Stability of Piceatannol in DMEM
Piceatannol was dissolved in DMSO to obtain a standard solution (5 mM), which was kept at −20 °C in the dark. An in situ analysis based on UPLC-MS/MS was used to analyze the stability of piceatannol in DMEM at 37 °C. DMEM (900 μL) and 100 μL of piceatannol standard solution (5 mM) were mixed in an autosampler vial and immediately placed in the UPLC autosampler, which was kept at 37 °C. Every 35 min, a 5 μL aliquot was automatically injected into a Thermo Scientific LTQ Orbitrap XL hybrid FT Mass Spectrometer.
Effect of Ascorbic Acid on the Stability of Piceatannol in DMEM
Ascorbic acid was dissolved in MilliQ water to obtain a solution of 10 mM before being used. In an autosampler vial, 850 μL of DMEM and 50 μL of ascorbic acid solution (10 mM) were mixed, then 100 μL of piceatannol standard solution (5 mM) was added and mixed well. Immediately, an aliquot (5 μL) was automatically injected into a Thermo Scientific LTQ Orbitrap XL hybrid FT Mass Spectrometer.
UPLC-LTQ Orbitrap XL Hybrid FT Mass Analysis
For qualitative analysis, the assay was performed using an ultimate 3000 hyperbaric liquid chromatography system coupled to a Thermo Scientific LTQ Orbitrap XL hybrid FT Mass Spectrometer via an ESI interface. The chromatography system consisted of an autosampler, a diode array detector, a column compartment and 2 pumps. Xcalibur and Mass Frontier 7.0 software packages were used for data collection and analysis. Liquid chromatographic separations were performed using a Waters ACQUITY UPLC HSS T3 column (2.1 × 150 mm, 1.8 µm). The mobile phase consisted of 0.1% formic acid in water (solvent A) and acetonitrile (solvent B). The samples were eluted with the following linear gradient: 15% B at 0 to 5 min, 15% to 25% B at 5–10 min, 25% to 40% B at 10 to 20 min, 40% to 45% B at 20 to 25 min, 45% to 15% B at 25 to 30 min. After that, the mobile phase was returned to the initial condition and held for 5 min so that system could equilibrate for the next injection. The flow rate was 0.3 mL/min. The temperature-controlled column oven was set at 25 °C and the autosampler was kept at different temperatures. Positive ionization (ESI) modes were used in the analysis. The capillary temperature was 350 °C, sheath gas (N2) flow rate 40 psi, aux gas flow rate 10 psi, and the ion spray voltage was set at 3.5 kV. In the FT cell, full MS scans were acquired in the range of
Footnotes
Acknowledgements
The authors would like to thank Professor Lunzhao Yi from Kunming University of Science and Technology, China for chemical structure analysis.
Authors’ Contributions
Meng Sam Cheong contributed toward the stability assay and the preparation of the manuscripts. Hui Cao carried out data analysis. Wai San Cheang and Jianbo Xiao supervised the experiments and checked the descriptions in the manuscript. Lutfun Nahar and Satyajit D Sarker revised the manuscript. All authors have read and approved the final manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work is supported by Ramón y Cajal grant (RYC2020-030365-I). Lutfun Nahar gratefully acknowledges the financial support of the European Regional Development Fund-Project ENOCH (No. CZ.02.1.01/0.0/0.0/16_019/0000868).
