Abstract
Research on multitargeting drugs is emerging, focusing on the discovery of agents that simultaneously act on more than one biological target. Here, a novel synthetic route to access the fused-heterocycles 1,4-dihydropyrazolo[4,3-b]indoles (
Introduction
Indoles are widely distributed in nature and are attractive biological molecules. 1 Hybridization of indoles with other active moieties would be an important approach to generating new therapeutic agents with higher potency. Sunitinib, indomethacin, pindolol, physostigmine, delavirdine, metralindole, vincristine, and sumatriptan are indole-containing drugs approved by the Food and Drug Administration for the treatment of cancer, cardiovascular and neurologic disorders. 2 Among these drugs, heteroaryl-substituted or fused-indoles frequently form privileged architectural units of the molecules. 3 Recently, 1,4-dihydropyrazolo[4,3-b]indoles have been designed as structural scaffolds of new Topo I/II inhibitors based on the merging of potent anticancer pharmacophores carbazoles, pyrazolocarbazoles, and N-acetyl pyrazolines.4-6 This structural architecture was also confirmed by their preliminary docking into the proteins, which account for their proposed inhibitory activity. 7
Inflammation is a complex pathophysiological process in which a variety of signaling molecules in excess levels are produced by macrophages, mononuclear phagocytes, eosinophils, and neutrophils. 8 These are inflammatory mediators such as nitric oxide (NO), prostaglandin E2, and cytokines, such as interleukin-1β (IL-1β), interleukin-6, and tumor necrosis factor (TNF-α). 9 An overproduction of proinflammatory cytokines and NO characterizes the pathogenesis of many inflammation-derived diseases such as rheumatoid arthritis, inflammatory bowel diseases, atherosclerosis, diabetes, and cancer. 10
Effective cancer management often recommends a combination of multiple drugs with different mechanisms. Thus, research on multitargeting drugs is emerging, focused on the discovery of agents that simultaneously act on more than one biological target. Here, we report the synthesis of a new 1,4-dihydropyrazolo[4,3-b]indole and its antiproliferation effect on acute myeloid leukemia cells, and selective inflammatory cytokines inhibition in comparison with pyrazolo[4,3-c][2,1]benzothiazine 4,4-dioxide.
Results and Discussion
Only a few synthetic methods to access either pyrazolo[4,3-b] or [3,4-b]indole derivatives have been reported.11-13 Here, a new synthetic route for the novel 1,4-dihydropyrazolo[4,3-b]indole is shown in Scheme 1. 2,1-Benzothiazine 2,2-dioxide (

Synthesis of compounds
A large number of biological studies have been undertaken on pyrazolobenzothiazines, such as on their antiinflammatory15,16 and anticancer
17
activities, but nothing has been reported so far about the cytotoxic/antiproliferative/antiinflammatory activity of these compounds, which prompted us to investigate the novel pyrazolobenzothiazine (
In Figure 1, it is shown that 24 h of treatment with 10 and 20 µg/ml of compound

Compounds
Analysis of the cell cycle revealed a significant increase in cells in the G0/G1 phase with 5, 10, and 20 μg/ml of compound

Shows the analysis of the cell cycle in the same experiments. There was a significant block of cells in the G0/G1 phase of the cell cycle, with a consequent significant decrease of cells in the S and G2/M phases of the cell cycle.
The antiinflammatory activity of pyrazolobenzothiazines prompted us to test, by real-time polymerase chain reaction (PCR), the ability of compounds
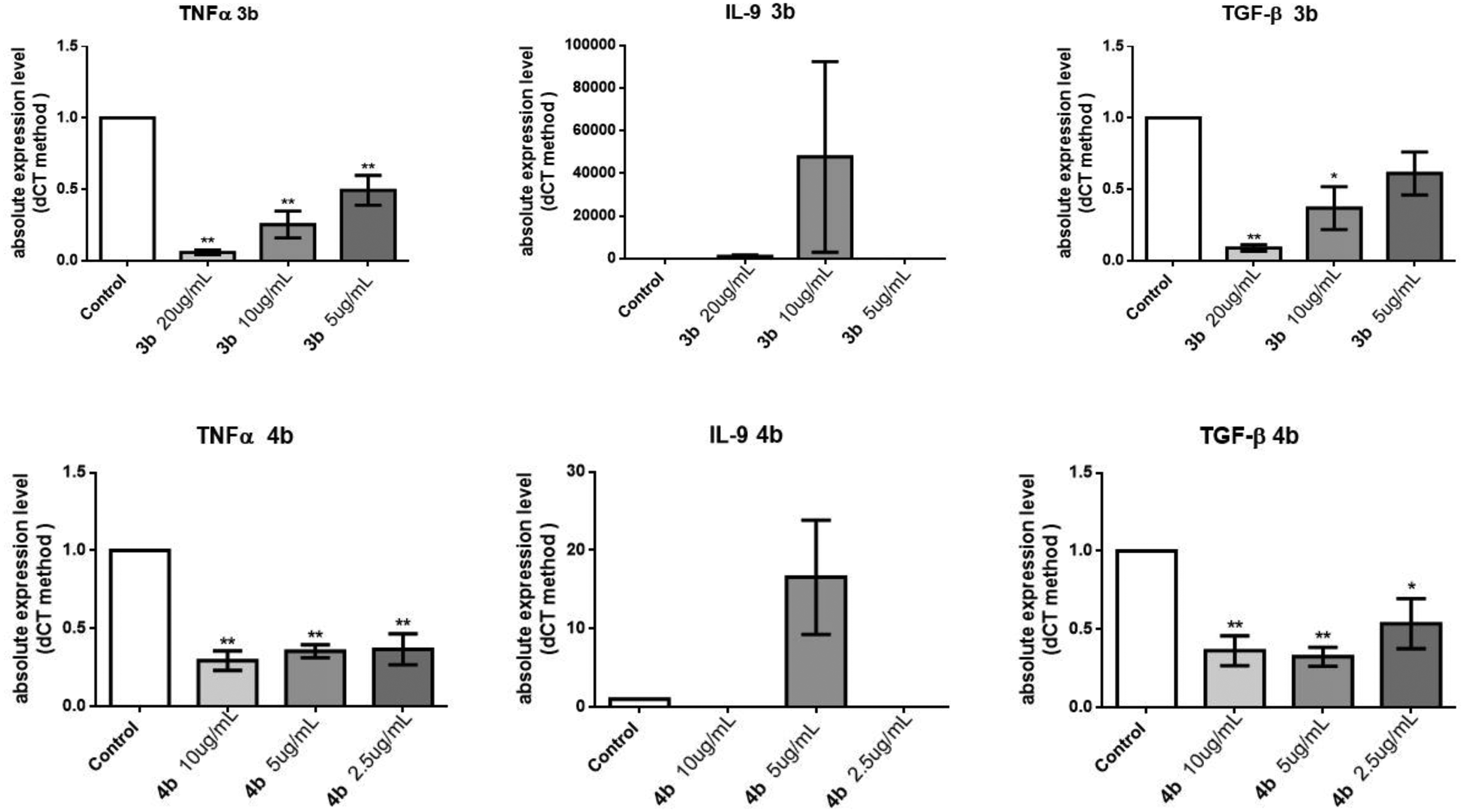
The cytokine analysis by real-time PCR for
Conclusion
We report here a novel synthetic approach to access 1,4-dihydropyrazolo[4,3-b]indols from pyrazolo[4,3-c][2,1]benzothiazine 4,4-dioxide via [H2O–SO2] elimination and intramolecular ring-closing reaction. The significance of compounds
Materials and Methods
General
All the reagents were purchased from commercial suppliers and used without further purification. Melting points were measured with a Krüss M5000 melting-point apparatus and are uncorrected. IR spectra were recorded with a Perkin Elmer Spectrum Two Fourier transform-infrared spectrometer by dispersion of the sample in KBr. The 1H NMR (500/600 MHz) and 13C NMR (125/150 MHz) spectra were recorded in CDCl3 or dimethylsulfoxide (DMSO)-d6 using Bruker AV-500 and AV-600 spectrometers with trimethylsilane as an internal reference. The J values are given in Hertz. High-resolution electrospray ionization (ESI) mass spectra were obtained on a SCIEX-X500R QTOF LC/MS system. Column chromatography was performed using silica gel (Kieselgel 60, 70-230 mesh and 230-400 mesh, Merck), and thin-layer chromatography was performed using precoated silica gel 60 F254 (0.25 mm, Merck).
Synthesis
(2,2-dioxido-1H-benzo[c][1,2]thiazin-3-yl)(p-tolyl)methanone (
(2,2-Dioxido-3,7b-Dihydro-1aH-Benzo[c]oxireno[2,3-e][1,2]thiazin-1a-yl)(p-Tolyl)methanone (2 )
Aroyl sultam
General Procedure for the Synthesis of 3a-hydroxy-1,3a,5,9b-tetrahydropyrazolo[4,3-c][2,1]benzothiazine 4,4-dioxides (3 )
A mixture of epoxide
3a-Hydroxy-1-Phenyl-3-(p-Tolyl)-1,3a,5,9b-Tetrahydropyrazolo[4,3-c][2,1]Benzothiazine-4,4-Dioxide (3a )
Applying the above experimental procedure using phenylhydrazine, compound
3a-Hydroxy-3-(p-Tolyl)-1-(4-(Trifluoromethyl)Phenyl)-1,3a,5,9b-Tetrahydropyrazolo[4,3-c][2,1] Benzothiazine-4,4-Dioxide (3b )
Applying the above experimental procedure using 4-(trifluoromethyl)phenylhydrazine,
General Procedure for the Synthesis of 1,4-dihydropyrazolo[4,3-b]indoles (4)
A solution of
1-Phenyl-3-(p-Tolyl)-1,4-Dihydropyrazolo[4,3-b]Indole (4a )
7
Light yellow solid, yield: 43%; m.p. 222.3-223.1; IR (KBr, cm−1): ν 3144, 1596, 1503, 1249; 1H NMR (DMSO, 600 MHz): δ (ppm) 11.51 (s, 1H), 8.02 (d, J = 8.0 Hz, 2H), 7.95 (d, J = 7.4 Hz, 2H), 7.90 (d, J = 8.0 Hz, 1H), 7.66 (t, J = 8.0 Hz, 1H), 7.59 (d, J = 8.2 Hz, 1H), 7.43–7.35 (m, 4H), 7.17 (t, J = 7.5 Hz, 1H), 2.40 (s, 3H). 13C NMR (150 MHz, DMSO) δ 144.92, 140.92, 137.59, 134.24, 131.85, 130.31, 130.22, 129.95, 129.86, 126.47, 126.02, 125.05, 120.56, 119.39, 119.31, 113.63, 112.83, 21.39. HR-MS (ESI) calcd. For C22H18N3 [(M + H)+]: 324.1501; found: 324.1485.
3-(p-Tolyl)-1-(4-(Trifluoromethyl)Phenyl)-1,4-Dihydropyrazolo[4,3-b]Indole (4b )
Light yellow solid, yield: 34%; m.p. 190.3-190.6; IR (KBr, cm−1): ν 3484, 3316, 1615, 1509, 1325, 1254, 1101; 1H NMR (DMSO, 500 MHz): δ (ppm) 11.64 (s, 1H), 8.17 (d, J = 8.4 Hz, 2H), 8.06-7.98 (m, 5H), 7.60 (d, J = 8.2 Hz, 1H), 7.44-7.35 (m, 3H), 7.20 (t, J = 7.6 Hz, 1H), 2.40 (s, 3H). 13C NMR (125 MHz, DMSO) δ 144.35, 143.22, 137.60, 135.12, 131.17, 130.17, 129.46, 128.81, 127.06 (d, J = 3.8 Hz), 125.72, 125.43, 125.31, 124.74, 124.16 (q, J = 245.3 Hz), 119.79,119.10 (d, J = 25.3 Hz), 113.23, 112.18, 20.87. HR-MS (ESI) calcd. For C23H17F3N3 [(M + H)+]: 392.1375; found: 392.1350.
OCI-AML Culture Conditions 23
The OCI-AML3 cell line was from the German Collection of Microorganisms and Cell Cultures (Braunschweig), cultured in 24-well plates at logarithm growth. RPMI 1640 medium was used with 10% fetal bovine serum, 100 U/ml penicillin, and 100 μg/ml streptomycin at 37 °C in 5% CO2. Cultures were grown at a concentration of 2 × 105 cells/ml, and treated with various doses of DMSO (the higher concentration 0.6 μl/ml). The compounds were added at concentrations of 2.5, 5, 10, and 20 μg/ml, and cells were analyzed after 24, 48, and 72 h.
Analysis of Cell Number, Apoptotic Cell Death, and Cell Cycle Progression 23
Cells were counted using a hemocytometer. Cell viability and cell cycle were assessed by flow cytometry to determine the propidium iodide (PI)-stained DNA content of cell nuclei after gating out of necrotic cells by size (forward scatter [FSC]). Briefly, cells were centrifuged and washed in phosphate-buffered saline (PBS). Cells were incubated in PBS containing 50 µg/ml PI for 30 min at 4 °C. Cells were then analyzed by flow cytometry using Coulter Epics XL-MCL equipment (Beckman Coulter Inc.). Doublet discrimination was made with the Coulter Epics XL-MCL™ Flow Cytometer SYSTEM IITM Software, which can detect >90% cellular doublets in cells ≥7 μm, using peak versus integral discrimination. A peak fluorescence signal was assigned to AUX to measure peak versus integral fluorescence.
Quantitation of Cytokine Levels by Enzyme-Linked Immunosorbent Assay 24
Cytokine secretion was evaluated by enzyme-linked immunosorbent assay in a medium in which OCI-AML cells, untreated or treated with experimental compounds, were cultured. High-binding 96-well microtiter plates (Nunc Maxisorb) were adhered with 100 ml anticytokine capture antibody (diluted in 0.1 M NaHCO3, pH 8.3) and incubated overnight at 4 °C. Plates were flushed three times with PBS–0.05% Tween and treated with 200 ml of PBS–10% fetal calf serum (FCS) for 1 h at RT. After having washed the plates, standard samples were added at 100 ml/well, and incubated for 2 h at RT. After washing with PBS–0.05% Tween plates were incubated with a biotinylated anti-cytokine antibody for 60 min at RT. After additional washes with PBS–0.05% Tween, horseradish peroxidase avidin D (Vector Laboratories) (1:2000 dilution in PBS–10% FCS) was added (100 ml/well), and plates were incubated for 30 min at RT, followed by additional washes in PBS–0.05% Tween. The antibodies utilized for the IL-4, IFN-γ, IL-9, IL-12, TNF-α, and TGF-β assays were rat anti-mouse cytokine mAbs (capture antibody) and biotinylated rat antimouse cytokine mAb (detection antibody) (Pharmingen)
Statistical Analysis
Statistical analysis was performed by the Mann–Whitney U-test. Statistically significant differences were in agreement with the criteria: *p<.05; **p<.01; ***p<.001.
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221105692 - Supplemental material for New 1,4-Dihydropyrazolo[4,3-b]indoles Induce Antiproliferation of Acute Myeloid Leukemia Cells and Inhibition of Selective Inflammatory Cytokines
Supplemental material, sj-docx-1-npx-10.1177_1934578X221105692 for New 1,4-Dihydropyrazolo[4,3-b]indoles Induce Antiproliferation of Acute Myeloid Leukemia Cells and Inhibition of Selective Inflammatory Cytokines by Vo Ngoc Binh, Sabrina Adorisio, Domenico V. Delfino and Quoc Anh Ngo in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Institute of Chemistry, Vietnam Academy of Science and Technology (VAST), funded by the Graduate University of Science and Technology (GUST.STS.ĐT2019-HH01), granted funding for this research (grant number GUST.STS.ĐT2019-HH01).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
