Abstract
Most biologically natural compounds are very sensitive and easily degradable under drying conditions. This study aimed to investigate the effect of drying temperature on the contents of phenolic compounds and in vitro antioxidant and antidiabetic activities of wild guava leaves. Wild guava leaves were dried at 40 °C, 50 °C, 60 °C, 70 °C, and 80 °C, as well as sun drying (SD). The total phenolic and flavonoid contents (TPC and TFC) were measured using the colorimetric method. Antioxidant activity was determined by the 2,2-diphenyl-2-picrylhydrazyl hydrate (DPPH) and 2,2-azino-bis(3-ethylbenzothazoline-6-sulfonic acid (ABTS) radical scavenging activity assays. In vitro antidiabetic activity was measured by assessing α-glucosidase and α-amylase inhibitory activities. The contents of individual phenolic compounds were determined using ultra-performance liquid chromatography (UPLC). The principal component analysis (PCA) results revealed that hot air drying at different temperatures had significantly different effects on the bioactive compounds and biological properties of guava leaves. Drying at high or low temperatures resulted in a higher degradable rate of bioactive compounds leading to a weakening of the biological properties. Drying at 50 °C resulted in high TPC and TFC as well as the highest in vitro antioxidant and antidiabetic activities. Furthermore, the highest contents of gallic acid, catechin, rutin, quercetin, apigenin, quercitrin, vitexin, and apigenin-7-O-D-glucopyranoside were found in samples dried at 50 °C, whereas the highest contents of chlorogenic acid, epicatechin, and ellagic acid were observed in samples dried at 60 °C. Therefore, drying at 50 °C was the suitable temperature for retaining valuable biological compounds in wild guava leaves.
Introduction
Psidium guajava L. (Guava) belongs to the family Myrtaceae and grows widely in tropical and subtropical regions, particularly in dry climates. Wild guava grows naturally in M’Drak District, Dak Lak Province, Vietnam. Wild guava leaves have been used by local people to control diarrhea, correct gastric problems, and regulate blood glucose levels in diabetic patients. A previous report indicated that guava leaves are rich in essential oils, triterpenes, tannins, kaempferol, and other compounds, such as flavonoids, malic acid, gallic acid, chlorophyll, and mineral salts. 1 Guava leaves are also well known for their antimicrobial, antioxidant, anti-inflammatory, and antidiabetic activities.2,3 Decoction, infusion, and boiling are the common ways to prepare either oral or topical agents depending on the illness, as reviewed by Elixabet et al. 4
Drying is a key technique to reduce moisture content in fresh materials, as it stops biochemical changes, inhibits microbial activities, preserves phytochemical compounds, extends the shelf life of fresh materials, and minimizes the costs of packaging and transportation. In addition, drying also changes the physical microstructure of the plant tissues leading to an increase in extraction yield.5,6 However, most bioactive compounds are very sensitive and are easily degraded by drying methods and conditions, such as high temperature, oxygen-rich air, and microwave/ultraviolet radiation. 7 The effect of a specific drying method on the preservation of raw quality cannot be predicted, as it is dependent on the type of chemical compounds present and the plant species.8,9 According to the results of Kausilya et al, 10 oven drying at 40 C results in better physicochemical characteristics of Carica papaya L. leaves among the investigated methods. Nguyen and Hoang 11 reported that oven drying at 80 °C is a better temperature for drying roselle (Hibiscus sabdariffa L.). Fahad et al 9 indicated that Kinnow mandarin and Eureka lemon seeds dried at 60 °C have higher antioxidant activity and total polyphenolic content (TPC), whereas drying at 70 °C is better for Orlando orange seeds. Furthermore, different drying methods, such as freeze drying, hot air drying, vacuum drying, and microwave drying, have significant effects on the phytochemicals and antioxidant properties of samples.7,12–14 Nguyen et al 15 reported that the microwave drying technique is the most suitable for drying cacao pod husks. Saifullah et al 16 showed the highest bioactive compound contents in lemon myrtle leaves after freeze drying. In contrast, Papoutsis et al 13 reported that the antioxidant and TPC were higher in lemon pomace dried by hot air or vacuum than those dried by freeze drying. Moreover, the contents of individual phenolic compounds are completely different in lemon pomace dried using different methods. 13 Therefore, there is a need to identify a suitable drying technique to minimize processing loss. This study aimed to investigate the effect of different drying temperatures on the content of bioactive components and in vitro antioxidant and antidiabetic activities of wild guava leaves.
Results and Discussion
Effect of Drying Temperature on Total Phenolic and Flavonoid Contents as well as the Antioxidant and Starch-Hydrolyzing Enzyme Inhibitory Activities of Wild Guava Leaves
The drying time, TPC and total flavonoid contents (TFC), and the bioactivities of the samples significantly differed with respect to the drying temperature (Table 1). The highest TPC and TFC (145.38 mg GAE/g dw and 21.06 mg QE/g dw) were observed in samples dried at 50 °C, followed by those dried at 40 C, 60 °C, 70 °C, and 80 °C. Previous studies have reported that guava leaves contain relatively high levels of phenolic compounds that contribute to biological activities, including antioxidant and starch-hydrolyzing enzyme inhibitory activities,3,17 whereas phenolic compounds are susceptible to different processing conditions, such as high temperature and oxygen. 18
Drying Time, Moisture, Total Phenolic and Flavonoids Contents, Antioxidant Activities, and In Vitro Antidiabetic Activity of Dried Wild Guava Leaves.

Abbreviations: DPPH, ABTS: DPPH and ABTS radical scavenging activities; SD, sun drying; TE, Trolox equivalent; TFC, total flavonoid content; TPC, total polyphenolic content.
HD40, HD50, HD60, HD70, HD80: hot air drying at 40 °C, 50 °C, 60 °C, 70 °C, 80 °C, respectively. Different letters above the values in each row indicate a significantly difference (P < .05).
The drying temperature has been reported to cause significant degradation of phenolic compounds in medicinal plants and herbs, such as roselle, 11 Vernonia Amygdalina leaves, 19 and citrus seeds. 20 In this study, a significant decrease in TPC and TFC of dried guava leaves was found after drying at 60 °C, 70 °C, and 80 °C. Drying at 40 °C and sun drying were carried out at the lowest temperature, and the long drying time led to material long exposure to heat, light, and oxygen that resulted in greater degradation of those compounds compared to short-term drying at high temperatures (Table 1). The high TPC and TFC contents in guava leaves dried at 50 °C may have been caused by the use of a middle drying temperature together with a relatively short drying time (10 h), which was shorter by 50% and 47% compared to that of drying at 40 °C and sun drying, respectively. Drying at 50 °C also yielded much higher TPC in guava leaves according to Nantitanon et al. 21 The TPC of 2 varieties of guava leaves reported by Elixabet et al 17 were 113.34 mg GAE/g dw (pomifera variety) and 86.12 mg GAE/g dw (pyrifera variety) which were significantly lower than that of wild guava leaves dried at 50 °C (145.38 mg GAE/g leaves dw) in this study. Nantitanon et al 21 observed more polyphenols after sonication-assisted extraction of guava leaves, and the maximum detected content was 136.02 mg GAE/g dw. Partial differences in TPC in this study compared to previous studies may be due to growing conditions, differences in varieties, climatic factors, and drying conditions, such as moisture content of the material and temperature. Since the moisture content of fresh materials influences the drying time to obtain the storage moisture of product.
The thermal drying process significantly reduces the antioxidant activity of antioxidant compounds in medicinal plants. 22 A variation in 2,2-diphenyl-2-picrylhydrazyl hydrate (DPPH) and ABTS radical scavenging activities via Trolox equivalents (TE) of dried guava leaves was observed for TPC and TFC. Table 1 shows that guava leaf extracts dried at 50 °C had higher ABTS and DPPH radical scavenging activities than those dried at other temperatures. Drying at 40 °C and sun drying maintained higher TPC and TFC than drying at 70 °C or 80 °C but possessed lower DPPH radical scavenging activity. This may be due to the difference in individual antioxidant compounds in guava leaves dried at 40 °C and sun drying compared to samples dried at 70 °C and 80 °C. The main antioxidant compounds may degrade more during exposure to a long drying time, light, and oxygen than to a high temperature during a short drying time. Moreover, higher temperatures increase the force to break down cellular structures and improve the release of specific phenolic compounds bound to cell wall macromolecules. 23 In addition, the lowest IC50 values for α-amylase and α-glucosidase inhibitory activities were detected in samples dried at 50 °C, followed by those that were sun-dried and hot air-dried at, 40 °C, 70 °C, and 80 °C. These results indicate that guava leaves dried at high or low temperatures increase the degradation of α-amylase and α-glucosidase inhibitory compounds, resulting from heat-labile active compounds destroyed by high temperature 18 or by exposure to long drying 21 during the thermal process.
Effect of Drying Temperature on Phenolic Compounds Content in Wild Guava Leaves
The changes in the phenolic compound contents analyzed by ultra-performance liquid chromatography (UPLC) due to the drying temperature are presented in Table 2. The 11 known phenolic compounds in this study were found in guava leaves dried at different temperatures, including gallic acid, catechin, epicatechin, rutin, ellagic acid, quercetin, apigenin, quercitrin, vitexin, apigenin-7-O-D-glucopyranoside, and chlorogenic acid (Table 2). Among them, the most predominant phenolic compound was catechin (16.46-30.07 mg/g dry leaves), which had a much higher content than the others, followed by chlorogenic acid (1.69-6.80 mg/g dry leaves), apigenin (2.55-6.36 mg/g dry leaves), and rutin (2.29-3.2 mg/g dry leaves). Gallic acid, ethyl gallate, ellagic acid, quercetin, quercetin, vitexin, and apigenin-7-O-D-glucopyranoside were present at lower concentrations in the wild guava leaf extract (Table 2). The results of Liu et al 24 indicated that catechin and epicatechin had the highest contents, whereas gallic acid, chlorogenic acid, quercetin, caffeic acid, and epigallocatechin gallate were present at lower concentrations in the guava leaf extract. Catechin, ellagic acid, and gallic acid are the major compounds in a guava leaf extract reported by Rahman et al. 25 Another study reported that quercetin, rutin, gallic acid, avicularin, and isoquercitrin in guava leaves accounted for about 65% of the total peak area on the chromatogram. 26 The differences in the composition and contents of phenolic compounds are due to differences in the guava variety, climatic conditions, maturity stage, and other factors, such as the extraction method and conditions, preparation method, and analytical technique.
The Contents of Phenolic Components Analyzed by ultra-performance liquid chromatography (UPLC) in Wild Guava Leaves Dried at Different Temperatures.
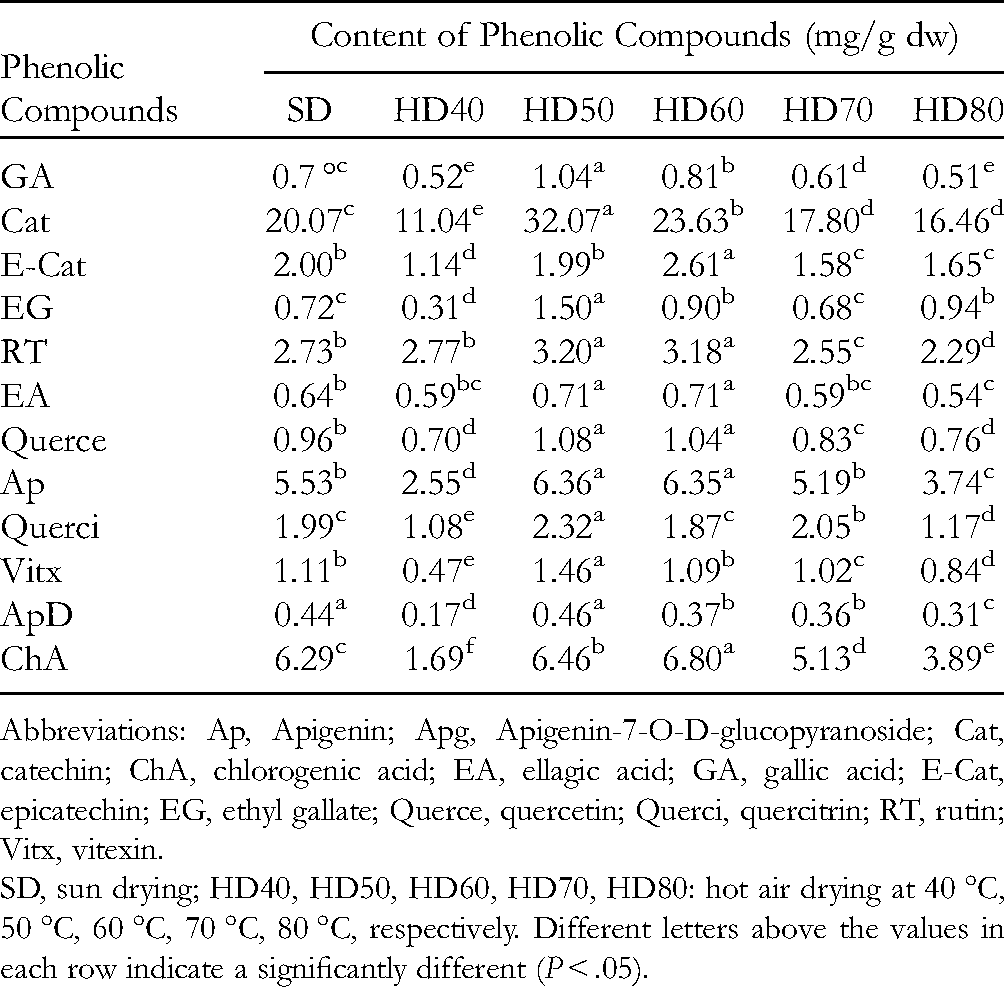
Abbreviations: Ap, Apigenin; Apg, Apigenin-7-O-D-glucopyranoside; Cat, catechin; ChA, chlorogenic acid; EA, ellagic acid; GA, gallic acid; E-Cat, epicatechin; EG, ethyl gallate; Querce, quercetin; Querci, quercitrin; RT, rutin; Vitx, vitexin.
SD, sun drying; HD40, HD50, HD60, HD70, HD80: hot air drying at 40 °C, 50 °C, 60 °C, 70 °C, 80 °C, respectively. Different letters above the values in each row indicate a significantly different (P < .05).
The results show that the drying temperature affected the contents of phenolic compounds. The contents of almost all of the phenolic compounds were highest in samples dried at 50 °C except for epicatechin, ellagic acid, and chlorogenic acid, which were found at their highest contents in samples dried at 60 °C and the lowest in samples dried at 40 °C, 70 °C, and 80 °C. The results shown in Table 2 demonstrate that thermal drying at 50 °C was suitable for dehydrating guava leaves in this study. Drying at higher or lower temperatures significantly increased the degradation rate of phenolic compounds in the wild guava leaves. These results agree with those of Katsube et al 27 indicating that the contents of chlorogenic acid, rutin, astragalin, kaempferol-3 (6-malonylglucoside), and quercetin-3 (6-malonylglucoside) were higher in mulberry leaves dried at 60 °C than in samples dried at 79 °C, 80 °C, or 110 °C. This result suggests that phenolic compounds are destroyed by high temperatures 18 and by exposure to a long drying process. 21
Correlation Analysis and the Principal Component Analysis
The Pearson's correlations (r) between TPC and α-glucosidase and α-amylase inhibitory activities, as well as DPPH and ABTS radical scavenging activities, were observed and arranged from moderate to very high (Table 3). The results demonstrated that phenolic compounds are the main active compounds in guava leaves. TFC was highly positively correlated with TPC (r = 0.96, P < .001) suggesting that flavonoids comprised most of the phenolic compounds. Moreover, TPC and TFC were highly negatively correlated with the IC50 value of α-amylase inhibitory activity (r = − 0.80, P < .001, and r = − 0.80, P < .001, respectively), indicating that the compounds strongly contributed to the α-amylase inhibitory activity present in guava leaves. The other compounds, except epicatechin, apigenin, apigenin-7-O-D-glucopyranoside, and chlorogenic acid, were correlated with TPC. Among them, highly positive correlations were detected between TPC and gallic acid (r = 0.85, P < .001), rutin (r = 0.79, P < .001), catechin (r = 0.74, P < .001), and ellagic acid (r = 0.72, P < .001) which have been reported as possessing anti-hyperglycemic 28 and antioxidant21,28 activities in guava leaves. This study also observed that rutin, ellagic acid, gallic acid, and quercetin were strongly negatively correlated with the IC50 values of α-amylase inhibitory activity with r (correlation coefficient) values of − 0.92, − 0.90, − 0.80, and − 0.76 at P < .001, respectively. These results suggest that rutin, ellagic acid, gallic acid, and quercetin are the main compounds expressing α-amylase inhibitory activity in guava leaves. Similar results were reported by Lingfeng Wu et al, 29 Antonella et al, 30 Li et al, 31 and Oboh et al. 32 Ethyl gallate, catechin, and vitexin were relatively highly negatively correlated with the IC50 value of α-glucosidase inhibitory activity (r = − 0.92; − 0.77, and − 0.74 with P < .001, respectively). This result indicates that ethyl gallate, catechin, and vitexin are responsible for the α-glucosidase inhibitory effect in wild guava leaves. The literature on α-glucosidase inhibitors shows that the α-glucosidase activity can be inhibited or slowed down by catechin,33,34 rutin, and astragalin. 35 Moreover, the combination of rutin, catechin, ellagic acid, and gallic acid is highly potent against glucosidase, as reported by Antonella et al. 30
Pearson's Correlation Matrix Among Total Phenolic and Flavonoid Contents (TPC and TFC), DPPH and ABTS Radical Scavenging (DPPH and ABTS) Activities, α-Glucosidase and α-Amylase Inhibitory Activities (AlphAI and AlphGI), and Phenolic Compounds of Wild Guava Leaves.

Abbreviations: Ap, Apigenin; Apg, Apigenin-7-O-D-glucopyranoside; Cat, catechin; ChA, chlorogenic acid; EA, ellagic acid; E-Cat, epicatechin; EG, ethyl gallate; GA, gallic acid; Querce, quercetin; Querci, quercitrin; RT, rutin; TFC, total flavonoid content; TPC, total polyphenolic content; Vitx, vitexin.
In addition, the results show that vitexin, gallic acid, catechin, and ethyl gallate were highly positively correlated with DPPH radical scavenging activity with r values of 0.77, 0.82, 0.88, and 0.92, respectively. Moderate correlations with DPPH radical scavenging activity were detected for apigenin (r = 0.59), quercitrin (r = 0.60), and quercetin (r = 0.64), whereas a good correlation with ABTS radical scavenging activity was observed for rutin (r = 0.73) and moderate correlations were found for ethyl gallate (r = 0.62; gallic acid (r = 0.59), and quercitrin (r = 0.52). Moreover, good correlations were detected between α-amylase inhibitory and ABTS radical scavenging activities as well as α-glucosidase inhibitory and DPPH radical scavenging activity. Consistent with these findings, the literature on phenolic-rich extracts reveals that enzyme inhibitory activity is highly correlated with antioxidant activity, which has been attributed to the phenolic content in samples.36,37
Principal component analysis (PCA) was used to identify the relationship between the drying temperature, the correlation between extract bioactivities, and the contents of individual compounds. Figure 1 is a representation of the 6 drying treatments for all of the contents of the individual compound and extracts bioactivities. The first dimension, denoted Dim.1, explained 68.5% of the total variability, whereas the second dimension, denoted Dim.2, explained 15.5% of the total variability. The PCA score biplot demonstrated that the individual compound contents and bioactivities were separated into two clusters without any outliers (Figure 1). The results indicate the strong contribution of the principal component axes (Dim.1 and Dim.2) to the total variability (83.7%) of the plotted data (Figure 1). The principal component (Dim.1) exhibited the greatest sample variation, followed by Dim.2 with the percentage contribution of variance of 68.5% and 15.5%, respectively. Of the 11 bioactive compounds concentrated on Dim.1, vitexin, catechin, gallic acid, ellagic acid, apigenin, and quercetin predominated on Dim.1 (scores > 0.6); thus, causing greater variability among these compounds. The total components (Dim.1 × Dim.2) described 83.7% of the total variance of the data and provide biased data strongly related to the effects of drying temperature on the contents of the compounds analyzed and the bioactivities evaluated (r2 = 0.999; P < .0001).

Principal component analysis (PCA) biplot for total phenolic and flavonoid contents (TPC and TFC), antioxidant activity, enzyme inhibitory activity, and phenolic compounds of dried wild guava leaves. Abbrieviation: HD, hot air (HD) at 40 °C, 50 °C, 60 °C, 70 °C, 80 °C; SD, sun drying ;Ap, Apigenin; Apg, Apigenin-7-O-D-glucopyranoside; ChA, chlorogenic acid; Cat, catechin; EA, ellagic acid; E-Cat, epicatechin; EG, ethyl gallate; GA, gallic acid; Querce, quercetin; Querci, quercitrin; RT, rutin; Vitx, vitexin.
As shown in Figure 1, the PCA-biplot of ABTS and DPPH radical scavenging activities via TE was directed to the positive side of the biplot, which was the most active area and closest to the analyzed compounds. In contrast, the IC50 values of α-glucosidase and α-amylase inhibitory activities were directed to the negative sides of the biplot, indicating that these compounds were positively correlated with ABTS and DPPH radical scavenging activities as well as α-glucosidase and α-amylase inhibitory activities.
Moreover, the samples dried at 50 °C and 60 °C as well as the sun-dried samples appeared on the positive side of the biplot with a strong correlation between the bioactive compounds and bioactivities (r2 = 0.999, P < .0001). The highest values on the positive sides of the x-axis were found in samples dried at 50 °C, followed by those dried at 60 °C and the sun-dried samples, suggesting that drying guava leaves at 50 °C is a suitable temperature to retain the bioactive compounds leading to high DPPH and ABTS radical scavenging as well as α-glucosidase and α-amylase inhibitory activities. In contrast, samples dried at 40 °C, 70 °C, and 80 °C were on the negative side of the x-axis, indicating that these drying conditions induced degradation of the bioactive compounds. The distribution in the negative area of Figure 1 indicated that samples dried at 40 °C retained higher contents of the compounds that possessed ABTS and α-amylase inhibitory activities, whereas higher contents of compounds that possessed DPPH and α-glucosidase inhibitory activities were found in samples dried at 80 °C. The PCA determined that hot air drying at different temperatures had significantly different effects on the bioactive compounds and bioactive properties.
Conclusion
The findings of this study indicate that wild guava leaves are a good source of phenolic compounds, including gallic acid, catechin, epicatechin, rutin, ellagic acid, quercetin, apigenin, quercitrin, vitexin, apigenin-7-O-D-glucopyranoside, and chlorogenic acid, which have antioxidant and hypoglycemic activities. In general, the drying process had a significant effect on the bioactive compounds as well as the bioactive properties of wild guava leaves. Drying at 50 °C was the optimal temperature for retaining the valuable bioactive compounds. Wild guava leaves can be effectively used to recover bioactive compounds with useful functional properties and can be applied as functional foods for patients with diabetes. The processing conditions should be optimized to ensure the maximum benefit from these leaves.
Materials and Methods
Chemical and Reagents
The Folin–Ciocalteu phenol reagent, sodium carbonate, sodium bicarbonate, metaphosphoric acid, phenolic compounds (eg gallic acid, ethyl gallate, catechin, epicatechin, chlorogenic acid, rutin, quercetin, quercitrin, apigenin, apigenin, astraginin, apigenin-7-O-D-glucopyranoside, and ellagic acid), DPPH, ABTS, α-amylase, α-glucosidase, and methanol were purchased from Sigma-Aldrich (St. Louis). Ethanol, methanol, and acetic acid were obtained from Xilong Scientific Co., Ltd (Guangdong Province Shantou City, China). Other chemicals and reagents were of analytical grade.
Sample Preparations
Wild guava leaves were collected from a forest in M’drak, Dak Lak Province in August 2020. The fresh leaves were contained in a gunny bag to transfer to the laboratory. Afterward, the leaves were pre-washed in tap water, dried with a towel, and cut into small pieces. The leaves were dried in a hot air oven at 40 °C, 50 °C, 60 °C, 70 °C, and 80 °C (Memmert) or sun-dried at 28 °C to 35 °C. In the current research, all samples were dried until they reached a moisture content <8%, which is the recommended value for storing dried leaves and producing a powder. 38
Extract Preparation
Wild guava leaf powder (5 g) was soaked in 50% ethanol in water at a ratio of (1:10) for 2 min and then sonicated for 20 min at 28 kHz (Vietsonic, VS28H, Vietnam) with a micro-tipped probe (diameter 10 mm) immersed 1 cm into the solution. The extracts were centrifuged at 15,000 × g for 15 min, and the supernatants were filtered through Whatman filter paper. The residues were extracted twice as mentioned above and then the separate extract solutions were combined and measured in a 150 mL volumetric flask. The samples were stored at − 30 °C until further use.
Amounts of Phenolic and Flavonoid Compounds
The determination of TPC in the plant methanol extracts followed the method of Nguyen and Eun. 39 To prepare the calibration curve, 1 mL aliquots of 0.024, 0.075, 0.105, and 0.3 mg/mL ethanol gallic acid solutions were separately mixed with 5 mL of Folin–Ciocalteu reagent (diluted 10-fold) and 4 mL (75 g/L) of sodium carbonate. The absorption values were read after 30 min at 20 °C and 765 nm, and the calibration curve was drawn. One milliliter of the plant extract was mixed with the reagents described above, and absorption values were measured after 30 min to determine the TPC. All determinations were performed in triplicate. TPC of the plant extracts was expressed as gallic acid equivalents (GAE)/g dw.
Flavonoid contents were measured using a modified colorimetric method described by Nguyen and Eun. 39 The extract solution was added to a test tube containing 1.25 mL of distilled water. Sodium nitrite solution (0.075 ml, 5%) was added to the mixture, followed by 5 min of incubation. Then, 0.15 ml of 10% aluminum chloride was added. After 6 min, 0.5 mL of 1 M sodium hydroxide was added, and the mixture was diluted with 0.275 ml of distilled water. The absorbance of the mixture was measured immediately at 510 nm and compared to a quercetin standard curve. Flavonoid contents are expressed as mg of quercetin equivalents (QE)/g dw.
DPPH Radical Scavenging Activity
The DPPH free radical scavenging activities of the guava leaf extracts were measured by a method modified slightly from Nguyen et al. 15 A stock solution of 0.24 g/L (w/v) DPPH in methanol was prepared and stored at − 18 °C until use. Before the DPPH reaction, a working solution was prepared by diluting 1.0 mL of stock solution with 45 mL of methanol to achieve an absorbance of 1.1 ± 0.02 at 517 nm. The reaction was prepared with 0.1 mL of the guava extract solution and 3.9 mL of the working solution, which was held in the dark at room temperature for 30 min. The absorbance of the mixture was measured at 517 nm using a UV-VIS spectrophotometer (V630, Jasco, Tokyo, Japan). Ethanol and Trolox were used as the control and standard, respectively. The DPPH radical scavenging activity of the guava leaves extracts was expressed as mg TE/g dw.
ABTS Radical Scavenging Activity
The ABTS radical scavenging activity of the guava leaf extract was measured by a method slightly modified from Nguyen et al. 15 Briefly, a stock solution was prepared by mixing 7.4 mM ABTS* + (2,2′-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid)) with 2.6 mM K2S2O8 at a ratio of 1:1 (v/v), followed by a 12-hour incubation in the dark at room temperature. Then, the solution was stored at − 18 °C until use. A working ABTS solution was prepared by mixing 1 mL of stock solution with 60 mL of methanol to obtain an absorbance of 1.1 ± 0.02 at 734 nm. A mixture of 0.15 mL of the extract and 2.85 mL of working solution was thoroughly stirred and kept in the dark for 2 h at room temperature. The absorbance of the mixture was measured at 734 nm using a UV-VIS spectrophotometer. Ethanol and Trolox were used as the control and standard, respectively. The ABTS radical scavenging activity of the guava leaf extracts was expressed as mg TE/g dw.
In Vitro Enzymatic Inhibition Assays
α-Glucosidase Inhibition Assay
The inhibition of α-glucosidase activity was measured by a method slightly modified from Apostolidis et al. 40 Briefly, 50 μL of guava leaf extract solution and 100 μL of 0.1 M phosphate buffer (pH 6.9) containing α-glucosidase (1.0 U/mL) were incubated in a 96-well plate at 25 °C for 10 min. Then, 50 μL of 5 mM p-nitrophenyl-α-D-glucopyranoside solution in 0.1 M phosphate buffer (pH 6.9) was added to each well at timed intervals. The reaction mixtures were incubated at 25 °C for 5 min. Before and after the incubation, absorbance readings were recorded at 405 nm using a microplate reader (Bio-Rad Laboratories, Inc.) and compared to a control that had 50 μL of buffer in place of the extract. The α-glucosidase inhibitory activity was expressed as inhibition (%) and was calculated as follows:
Inhibition (%) = (A0 − A1)/A0 × 100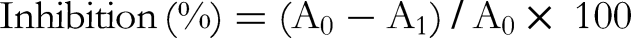
Porcine pancreatic α-amylase inhibitory activity assay
The inhibition of porcine pancreatic α-amylase activity was measured by a method slightly modified from Kwon et al. 41 Two hundred microliters of sample solution at different concentrations were mixed with 500 µL of 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M sodium chloride) and α-amylase solution (0.5 mg/mL) and incubated at 25 °C for 10 min. Then, 500 µL of a 1% starch solution in 0.02 M sodium phosphate buffer was added and incubated at 25 °C for 10 min. The reaction was stopped with 1.0 mL of dinitrosalicylic acid and the color reagent. The reaction mixture was boiled in a water bath for 5 min and cooled to room temperature. Finally, the reaction mixture was diluted by adding 10 mL of water, and absorbance was measured at 540 nm with the microplate reader. These data were submitted to linear regression, and a straight line equation was obtained for the IC50 calculation.
Phenolic Compound Analysis by UPLC
The extract solution was filtered through a 0.22 µm membrane before injecting into the UPLC system. Each sample was injected in triplicate at a volume of 5 µL. The phenolic compounds were separated using the Thermo-Ultimate 3000 UPLC system (Thermo Scientific) equipped with a 250 mm × 4.6 mm, particle size 5 µm (BDS Hypersil C18 column) and connected to a UV detector monitoring at 265 nm. A binary solvent system was used that was comprised of (A) methanol and (B) Milli-Q water acidified with 0.1% H3PO4. Separations were performed using a segmented gradient as follows: 97% B (0-0.5 min); 97% to 83% B (0.5-8.0 min); 83% to 70% B (8.0-10.0 min); 70% to 55% B (10.0-15.0 min); 55% to 5% B (15-20 min); 5% to 97% B (20-22 min), and 97% B (22-23 min) at a flow rate of 0.7 mL/min. Calibration curves were prepared from the signals obtained from the standard solutions based on the ratio of the chromatographic peak area for each to that of the corresponding internal standard. The phenolic compounds were quantified from a specific calibration curve of the standards.
Data Analysis
The one-way ANOVA combined with the LSD post hoc test (P < .05) were used to assess the differences among the treatments. The results were graphically presented by mean ± standard deviation (SD) for the group determinations. In addition, the PCA was used to assess the typology of individuals (ie drying treatments) in one part and relationships between variables (ie biological compounds). The PCA was performed using R software version 4.1.2 42 with the package FactoMineR 43
Footnotes
Author Contributions
Quang-Vinh Nguyen: Conceptualization, Methodology, Data analysis, Writing—original draft, Investigation, Formal analysis, Validation, Visualization, Writing—review and editing.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by the Ministry of Education and Training under grant number of B2021-TTN-03 and partially funded by the Vietnam National Foundation for Science and Technology Development (NAFOSTED) under grant number 106.99-2020.17. Minh-Trung Nguyen was funded by Vingroup JSC and supported by the Master’s, PhD Scholarship programme of Vingroup Innovation Foundation (VINIF), Institute of Big Data, Code VINIF.2021.TS.157.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.

