Abstract
Introduction
Māmaki tea, Pipturus albidus (Hook. & Arn.) Gray, Nettle family (Urticaceae), a native Hawaiian plant species, 1 is well known for its medicinal uses. It is endemic to and only grows on the Hawaiian Islands.2,3 Māmaki tea is reported to contain diverse antioxidants, such as catechins, chlorogenic acid, and rutin,4,5 along with antibacterial and antiviral activities. 6 The medicinal uses of Māmaki tea range from antioxidant, mild natural laxative, anti-allergic effect, promoting cardiovascular and liver health, to decreasing stress levels.2,3 Antioxidants also play an important role in cancer chemoprevention and development in laboratory and animal model studies.7,8 As such, in addition to evaluating the antioxidant activity of Māmaki tea, we are also interested in investigating anticancer and chemopreventive potential associated with Māmaki tea extracts. 9 Specifically in this work, we examined various preparations and brewing methods (e.g., different temperatures, water and ethanol extraction, tea powders and leaves, and dehydrated or freeze-dried) and investigated which method may produce the best medicinal properties of Māmaki tea.
Results
Antioxidant Activity
The Ferric Reducing Antioxidant Power (FRAP) values of Māmaki tea and fruit extracts were determined in two triplicates (n = 6). Catechin was used as a positive control with a calculated FRAP value of 155.6 ± 16.7 µM/µg. The value for Māmaki tea extract was 40.0 μM/μg, cucumber 1.3 μM/μg, lilikoi 1.4 μM/μg, lemon 2.8 μM/μg, and papaya 4.6 μM/μg, with the Māmaki tea extract showing the highest level of antioxidant activity.
With anticancer and chemopreventive potential, all Māmaki tea extract samples (AS-1 to AS-25) were tested for inhibitory activity against nitric oxide (NO) production, nuclear factor kappa B (NF-κB) activation, and proliferation of two cancer cell lines. The results are shown in Table 1. Initial determinations for all these assays were carried out at a sample concentration of 20 µg/mL.
Activities of Māmaki Tea Extract Samples AS-1 to AS-25 Evaluated in Anticancer and Chemopreventive Assays.

Refer to Table 2 for detailed sample preparation information of AS-1 to AS-25.
IC50 values of N-tosyl-L-phenylalanine chloromethyl ketone (TPCK) in the NF-kB assay using human embryonic kidney HEK-293 cells.
IC50 values of L-NG-monomethyl arginine (L-NMMA) in the nitrite assay using LPS-stimulated RAW 264.7 murine macrophage cells.
IC50 values of staurosporine, a microbial alkaloid, in the cytotoxicity assay.
Abbreviation: NF-κB: nuclear factor kappa B.
NF-κB Inhibition
Among all the extract samples tested, overall the dehydrated Māmaki tea powders showed the highest inhibition activities in the NF-κB assay. For example, the room temperature, warm, and boiling tea samples using the dehydrated Māmaki tea powder showed over 60% of inhibition against NF-κB. Notably, the room temperature (AS-16) and boiling water (AS-18) samples from the dehydrated Māmaki tea powder had the highest percentages of inhibition of 73.7% and 75.0%, respectively. The freeze-dried Māmaki tea whole leaves (Sample AS-6), extracted from room temperature water, also showed 60.6% inhibition. Among the tea samples extracted with warm water (60 °C), the dehydrated Māmaki tea powder (AS-17) had the highest percentage of inhibition (62.7%). By comparing the freeze-dried Māmaki teas, samples AS-3, AS-4, and AS-5 extracted with boiling water and ethanol exhibited higher inhibition than those (AS-1 and AS-2) from room temperature and warm water. In addition, the 70% ethanol sample (AS-24) from dehydrated Māmaki tea whole leaves also showed 68.4% inhibition.
Nitric Oxide (NO) Inhibition
In general, all extract samples showed less than 60% inhibition of NO production in LPS-stimulated RAW 264.7 cells. However, the ethanol extract samples (e.g., the 100% ethanol extract from freeze-dried Māmaki tea sample, AS-10; the 70% ethanol extract from dehydrated Māmaki tea sample, AS-24; and the 100% ethanol extract from dehydrated Māmaki tea sample, AS-25) all showed over 50% inhibition when tested at 20 µg/mL. Interestingly, the room temperature water sample (AS-1) from freeze-dried Māmaki tea powders also showed the most potent percentage inhibition (55.5%).
Cytotoxicity Effect
When all Māmaki tea extract samples were tested at 20 µg/mL against two representative cancer cell lines (lung and breast cancer cells), most samples were largely inactive, exhibiting over 80% survival toward cancer cell proliferation. Cytotoxicity data are shown in Table 1 and reported as % survival with the higher % survival being less or not toxic. It is worthwhile mentioning that the warm and boiling water extract samples (AS-12 and AS-13, respectively) from green tea loose leaves showed the most potent cytotoxic activity with relatively low survival rates (73.9 and 77.5%). The room temperature 100% ethanol extract sample (AS-25) from dehydrated Māmaki tea leaves had the best cytotoxic activity with the lowest % survival (71.3%) against breast cancer cells.
Discussion
In the antioxidant study, fruits and vegetables were peeled, seeded, juiced, and then filtered. The water-soluble filtrates were collected and freeze-dried for testing. This sample preparation could cause lower than actual antioxidant activities observed, because antioxidants could be found in peels, seeds, and pulps with different distribution profiles. 10 Different brewing methods can also determine the amount of antioxidants extracted from Māmaki tea and, therefore, the chosen brewing method in this antioxidant study may not be the most optimal one. Consistent with the previous report describing the identification of flavonoids and polyphenols in Māmaki tea,4,5 the current data show Māmaki tea extract had the highest antioxidant activity (40.0 μM/μg) among test extract samples, suggesting it may contain a rich source of natural antioxidants such as ( + ) catechins and rutin. 11
With regard to suppressing NF-κB activation, the room temperature (AS-16) and boiling water (AS-18) samples from the dehydrated Māmaki tea powders had the highest percentages of inhibition of 73.7% and 75.0%, respectively, among all the test samples. In addition, the room temperature, warm, and boiling water tea samples using the dehydrated Māmaki tea powder, along with the freeze-dried Māmaki tea whole leaves (Sample AS-6), showed > 60% of inhibition. The dehydrated Māmaki tea powder (AS-17) exhibited the highest percentage (62.7%) of inhibition among the warm temperature tea samples. From the freeze-dried tea samples, AS-3, AS-4, and AS-5 from boiling water and ethanol showed higher inhibition than AS-1 and AS-2 from room temperature and warm water. Finally, AS-24, the 70% ethanol sample from dehydrated Māmaki tea whole leaves, showed 68.4% inhibition against NF-κB. In the NO inhibition assay, all samples generally showed < 60% of inhibition when tested at 20 µg/mL. In contrast, the ethanol extract samples such as AS-10 (100% ethanol extract from freeze-dried Māmaki tea), AS-24 (70% ethanol extract from dehydrated Māmaki tea), and AS-25 (100% ethanol extract from dehydrated Māmaki tea) all exhibited > 50% of inhibition at 20 µg/mL. Notably, green tea extracts have been previously evaluated in clinical studies as an effective supplement with cancer chemopreventive potential.12,13
In cell-based cytotoxicity assays against representative lung and breast cancer cells, most samples were largely inactive with > 80% survival toward cancer cell growth when tested at 20 µg/mL. The 100% ethanol extract sample from dehydrated Māmaki tea leaves, AS-25, showed the lowest survival percentage (71.3%) against breast cancer cells. The overall high % survival and low cytotoxicity activity may be attributed to weak activity and/or relatively low amounts of active compounds in the extract samples tested. On the basis of these data, there are no apparent correlations among chemopreventive NF-κB and NO inhibition and cytotoxic activity. Interestingly, polyphenols and/or tea beverage may also have a synergistic effect with chemotherapeutic agents to improve treatment outcome and reduce adverse effects in cancer chemotherapy. 14
Collectively, Māmaki tea is a Hawaiian plant that has been traditionally used by native Hawaiians as a health remedy. So far, very limited scientific and clinical studies have been done toward Māmaki tea and its health benefits. Thus, we see a window of opportunity to focus on Māmaki tea studies and obtain evidence-based scientific data to support its medicinal and therapeutic uses. This way, we can scientifically promote Māmaki tea, a unique product from Hawaii, to the rest of the world.
Conclusions
In conclusion, with regard to antioxidant evaluation, the antioxidant activity of Māmaki tea was measured as 40.0 µM/µg, showing the highest antioxidant level among the plants tested. In anticancer and chemopreventive studies, the Māmaki tea sample was generally more active than the green tea sample. Specifically, the room temperature and boiling water extract samples from the dehydrated Māmaki tea powder demonstrated the highest percentage inhibitions ranging from 73.7% to 75.0% in the NF-κB assay; the freeze-dried Māmaki leaf powders brewed at room temperature had the highest inhibitory activity in the nitrite assay. In the cytotoxicity assay, the 100% ethanol extract sample (AS-25) from dehydrated Māmaki tea leaves showed the best activity with the lowest survival against breast cancer cells (71.3%). Chemical and metabolomic profiling of these Māmaki tea extract samples are ongoing and will be reported in due course.
Materials and Methods
Testing Materials
Fresh Māmaki tea (P. albidus) leaves, fresh papaya, fresh cucumber, fresh lilikoi, and fresh lemon were purchased from Big Island Hilo Farmers Market. The Māmaki tea plant was identified by Dr. Leng Chee Chang. A voucher specimen (No. MKK. 002) was deposited in the Natural Product Chemistry Laboratory, the Daniel K. Inouye College of Pharmacy, University of Hawaii at Hilo.
Chemicals, Reagents, and Cell Cultures
Solvents and reagents were purchased from either Sigma-Aldrich or Fisher Scientific and used without further purification. These chemicals include ethanol, dimethylsulfoxide (DMSO), acetate buffer (300 mM, pH 3.6), 2,4,6-tripyridyl-s-triazine (TPTZ), FeCl3•6H2O, and FeSO4•7H2O; the Luciferase Reporter Assay System was purchased from Promega. Dulbecco's modified Eagle media (DMEM), antibiotic-antimycotic solution (100 × ), and fetal bovine serum (FBS) were purchased from Invitrogen, and phosphate-buffered saline (PBS), Griess reagent [1:1 mixture (v/v) of 1% sulfanilamide in H3PO4 and N-(1-naphthyl)ethylenediamine dihydrochloride solution], sodium nitrite, sulforhodamine B (SRB), trichloroacetic acid solution, acetic acid solution, lipopolysaccharide (LPS), and Tris·HCl from Sigma-Aldrich. The following cell lines were used: RAW 264.7 murine macrophage cells, human embryonic kidney 293 cells, and lung (LU-1) and breast (MCF-7) cancer cell lines. Unless otherwise stated, all cell lines were obtained from the American Type Culture Collection (ATCC), and grown in media supplemented with penicillin G (100 units/mL), streptomycin (100 mg/mL), and heat-inactivated FBS at 37 °C in 5% CO2 in a humidified incubator. Stock cell cultures were prepared and aliquots were stored in liquid nitrogen. During the course of the study, cells were used below a passage number of 20. No significant changes were noted in morphology or growth characteristics.
Supplies and Instruments
Project supplies and instruments used in this work include a 96-Well Microtiter Plate, Nesco® Professional Dehydrator, Ninja® Food Processor, GeneVac EZ-2 Evaporator, LABCONCO FreeZone Plus 12 Liter Cascade Console Freeze Dry System, Humidified Air Incubator, and ELY p800 Absorbance Micro Reader (BioTek Instruments). The luminometer (Lumistar Galaxy microplate reader) was programed to dispense 25 µL of the luciferase assay reagent into each well and the amount of light produced was measured.
Testing Materials Preparation
Antioxidant Study
Preparing Māmaki tea, Fruits, and Vegetables
Fresh Māmaki tea leaves, two papayas, four cucumbers, three medium-sized lemons, and five lilikoi were collected. Fresh Māmaki tea leaves were dried in a lyophilizer for 48 h. The dried Māmaki tea leaves (15 g) were soaked for 30 min in 300 mL of warm water (60 °C). After 30 min, the tea was filtered and poured into a separate bowl. The soaking and filtering processes were repeated twice. All of the filtrates was next combined and left in a freezer until it was completely frozen. The frozen tea filtrate was then left in the lyophilizer to freeze dry, producing Māmaki tea powders for experimental use. The fruit and vegetables were seeded, peeled, and then juiced in a slow juicer. The juice mixture was filtered, and the filtrate was left in the freezer until frozen. The frozen filtrate was lyophilized to let it freeze dry. Once the samples turned into powder and/or residue, they were ready to use. (On average, it took about 5 days to completely freeze dry the frozen juice.)
Sample Preparation
All the freeze-dried test materials were weighed and recorded, and subsequently transferred into vials, which were stored in a refrigerator for testing. Next, the Māmaki tea solution (10 mg/mL) was prepared by dissolving 10 mg of freeze-dried Māmaki tea in 1 mL of deionized water. The 1 mg/mL Māmaki tea solution was prepared by mixing 100 μL of the tea solution (10 mg/mL) with 900 μL of deionized water. The remaining 10 and 1 mg/mL vegetable and fruit sample solutions were prepared accordingly.
Anticancer and Chemopreventative Study
Preparing Māmaki tea Samples Using Different Brewing Methods
Portions of fresh Māmaki tea leaves were dried in a lyophilizer for 72 h. The rest of the fresh Māmaki tea leaves were dried in a dehydrator at the lowest temperature setting for around 3.5 h. Next, half of either the freeze-dried or dehydrated tea leaves were blended into powders using a food processor. The Māmaki tea samples used in this study include: (i) dehydrated Māmaki tea powders; (ii) dehydrated Māmaki tea leaves; (iii) freeze-dried Māmaki tea powders; and (iv) freeze-dried Māmaki tea leaves. Brewing methods included room temperature water (27 °C), warm water (60 °C), boiling water (100 °C), 70% ethanol in water, and 100% ethanol.
Twenty-five tea samples AS-1 to 25 were weighed and stored in tea bags with 1.5 g per sample. Specifically, samples AS-1 to 5 are freeze-dried Māmaki tea powders; AS-6 to 10 are freeze-dried Māmaki tea leaves; AS-11 to 15 are green tea loose leaves for comparison; AS-16 to 20 are dehydrated Māmaki tea powders; and AS-21 to 25 are dehydrated Māmaki tea leaves (Table 2).
A Summary of Tea Sample Preparation Types, Extraction Temperature, and Solvent Used.
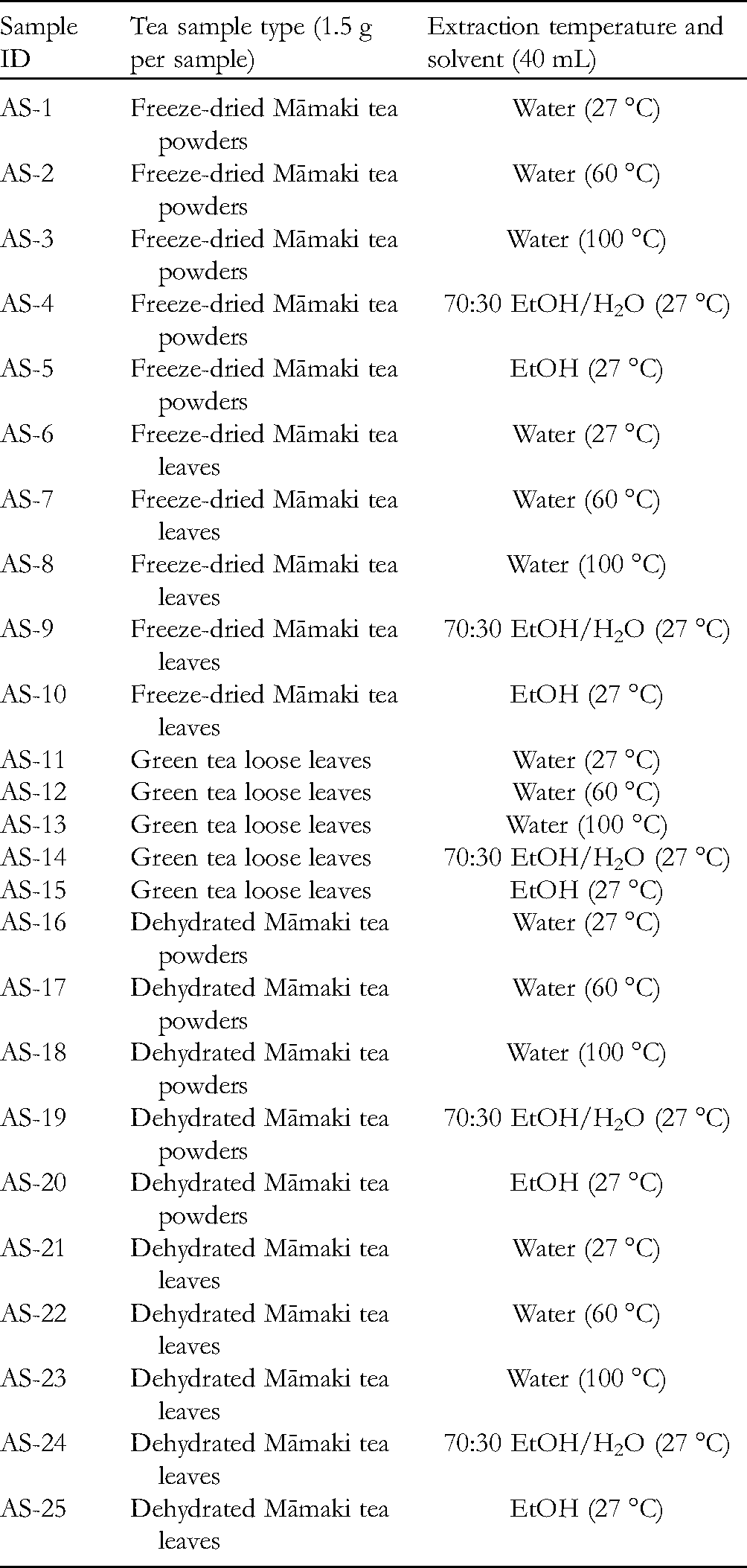
Forty mL of room temperature (27 °C) water was added to each polypropylene centrifuge tube containing samples AS-1, 6, 11, 16, and 21, respectively. The resulting centrifuge tube was shaken every 10 min during a 30 min period. After 30 min, the liquid was decanted, ensuring that most tea solution was drained from each tea bag. Tea solutions were collected and stored in labeled centrifuge tubes for later experiments. Accordingly, 40 mL of warm temperature (60 °C) water, boiling (100 °C) water, room temperature 70% ethanol in water, and room temperature 100% ethanol were added to each polypropylene centrifuge tube containing samples AS-2, 7, 12, 17, and 22; samples AS-3, 8, 13, 18, and 23; samples AS-4, 9, 14, 19, and 24; and samples AS-5, 10, 15, 20, and 25, respectively. Each of the resulting centrifuge tubes was shaken every 10 min during a 30 min period. After 30 min, the liquid was decanted, ensuring that most tea solution was drained from each tea bag. Tea solutions were collected and stored in labeled centrifuge tubes for later experiments.
Tea solutions from samples AS-1 to 25 were evaporated using a GeneVac evaporator and/or a lyophilizer instrument to remove water and/or ethanol. The dried powders from each sample were subsequently obtained and weighed.
Preparation of Samples for Assays
Test samples were weighed and dissolved in DMSO to make an initial sample concentration of 4 mg/mL; these were further diluted to give a final test concentration of 20 µg/mL. Briefly, test and control solutions (10 µL per well), with control groups containing a 10% DMSO aqueous solution, were plated in microtiter plates, followed by additional 1/20 dilutions by adding 190 µL cell solution into each well. Each test sample was evaluated in triplicate.
Assay Methods and Protocols
Antioxidant FRAP Assay
The ferric ion-reducing/antioxidant power (FRAP) assay is a simple, quick, and inexpensive protocol to quantify a sample's total antioxidant activity.15–17 The reducing ability of antioxidants is tested by reducing ferric tripyridyltriazine (TPTZ) complex to the formation of ferrous TPTZ form. The absorbance change of a given sample at 595 nm can then be used to calculate its antioxidant potential by comparing it with a standard. 18
The FRAP reagent was produced by mixing 300 mM acetate buffer (pH 3.6), 10 mM 2,4,6-tris(2-pyridyl)-s-triazine (TPTZ) solution, and 20 mM FeCl3•6H2O in a 10:1:1 ratio and heating to 37 °C just before use.15–17 Then, the standard solutions of FeSO4 for FRAP assay were prepared and diluted as follows: 2.7801 g of FeSO4•7H2O (molecular weight: 278.01 g/mol) was weighed and dissolved in 10 mL of deionized water to make a 1 M stock solution. The 1000, 800, 600, 400, 200, 100, and 50 μM solutions were prepared accordingly by serial dilution. The FRAP reagent (150 μL) was added to each well of the 96-well plate. A blank reading was taken at 595 nm using a microplate reader (BioTek ELY 800 Absorbance Reader). Twenty μL of the test sample in triplicate and 20 μL of standards of known Fe (II) concentrations in triplicate were added to the 96-well plate accordingly. After incubating at room temperature for 8 min, the plate was read at 595 nm. This process was repeated once so that six sets of data for each test sample were obtained. Catechin was used as a positive control.15–17
Chemopreventive and Cytotoxicity Assays
Chemopreventive potential was first measured using the luciferase NF-κB assay.19–21 This assay is determined by emitting light via the oxidative conversion of luciferin to oxyluciferin through electron transition. In addition, the Māmaki tea extract samples were also tested against two cancer cell lines in cell-based assays. The effect of extracts on proliferation of cultured cancer cells was tested using the cellular protein-staining SRB assay. 22 The chemopreventive property was also measured using the NO colorimetric assay.21,23 NO functions as an important physiological messenger and effector molecule and is present in diverse biological systems, such as neuronal, immunological, and cardiovascular tissues.24,25 One strategy to evaluate the NO formation is to test the level of nitrite (NO2−), one of two major products of NO.
NF-κB Luciferase Assay
This assay was performed using NF-κB reporter stably transfected human embryonic kidney cells 293 (HEK-293) from Panomics Inc. (Fremont, CA). This cell line contains chromosomal integration of a luciferase reporter construct, regulated by an NF-κB response element. The gene product, luciferase enzyme, reacts with luciferase substrate, emitting light, which can be detected with a luminometer. Recombinant, human tumor necrosis factor (TNF)-α, expressed in Escherichia coli, (Calbiochem, Gibbstown, NJ) was used as an activator at 2 ng/mL (0.14 nM in the incubation mixture). After a 6 h incubation with tested samples, the reaction was stopped by adding 50 µL Reporter Lysis Buffer (Promega) and frozen overnight at −80 °C. The cells were thawed and the inhibiting activity was measured using a luminometer LUMIstar Galaxy (BMG) and Luc assay system from Promega (Madison, WI). N-tosyl-L-phenylalanine chloromethyl ketone (TPCK) was used as a positive control. 20
Nitrite Assay to Measure NO Production in LPS-Stimulated RAW 264.7 Murine Macrophage Cells
Because of the instability of NO and its subsequent conversion to nitrite, the NO level in the cultured media was estimated by measuring the nitrite level. In brief, mouse macrophage RAW 264.7 cells from the American Type Culture Collection (ATCC®TIB-71TM) were incubated in 96-well plates at 37 °C, in a 5% CO2 humidified air incubator for 24 h. Cells were next treated with various tea extract samples for 15 min first, followed by treatment with or without LPS (1 μg/mL) for an additional 20 h. After the incubation, nitrite released in the cultured media was measured using Griess reagent [1:1 mixture (v/v) of 1% sulfanilamide in 5% H3PO4 and 0.1% N-(1-naphthyl)ethylenediamine dihydrochloride solution], and absorbance was measured at 540 nm with a microplate reader. The concentration of nitrite was calculated using a standard curve created with known concentrations of sodium nitrite. To evaluate the cytotoxic effects of test samples with RAW 264.7 cells under the same experimental conditions, the SRB assay was performed.21,23 L-NG-monomethyl arginine citrate was used as a positive control.
Sulforhodamine B (SRB) Cytotoxicity Assay
Māmaki tea extract samples were evaluated for cytotoxic activity using an in vitro SRB assay against breast (MCF-7) and lung (LU-1) cancer cell lines. 26 In brief, cancer cells (1 × 104 cells in 190 μL of the complete media) were plated in 96-well plates overnight and then treated with test samples and incubated at 37 °C in a 5% CO2 incubator for 72 h. All samples were assayed in triplicate. After the incubation, cells were fixed with 10% trichloroacetic acid solution for 30 min and stained with 0.4% SRB in 1% acetic acid solution for 30 min. After washing with 1% acetic acid solution, protein-bound SRB was dissolved in 10 mM Tris buffer (pH 10.0) and optical densities were determined at 515 nm. The effect of extract samples on cell survival was evaluated as % survival in comparison with vehicle (DMSO)-treated control cells.21,22
Footnotes
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
