Abstract
Zingiber cassumunar is widely distributed in Asia, and its rhizomes are used for asthma treatment in traditional medicine and as a spice. Five new phenylbutanoids, cassumunols I–M (
Zingiber cassumunar Roxb. (Family Zingiberaceae) is widely distributed in Southeast Asia, and its rhizomes are used for the treatment of asthma, bronchitis, and gastrointestinal distress in traditional medicine and as a spice in Thailand.1-4 We have reported the isolation and structure elucidation of various phenylbutanoids, including phlains I–VI and cassumunols A–H, from the methanolic (MeOH) extract of Z cassumunar rhizomes.5,6 Those constituents showed biofunctional effects, such as nitric oxide production inhibitory effects induced by lipopolysaccharide in mouse peritoneal macrophages and inhibitory effects on the invasion of human fibrosarcoma HT 1080 cells. In the course of our studies on the bioactive constituents from medicinal foods7-11 and Z cassumunar, we have isolated 5 new phenylbutanoids, cassumunols I–M (
The MeOH extract of the rhizomes of Z cassumunar was partitioned in an EtOAc-H2O (1:1, v/v) mixture to give EtOAc-soluble and H2O-soluble phases, as reported previously.5,6 The EtOAc-soluble fraction was subjected initially to normal- and reversed-phase column chromatography (CC) and finally to HPLC to give cassumunols I (

Constituents from rhizomes of Z cassumunar.
Cassumunol I (
13C NMR Spectroscopic Data of Cassumunols I–M (

Interchangeable.
Overlap.
As shown in Figure 2, DQF-COSY experiments on
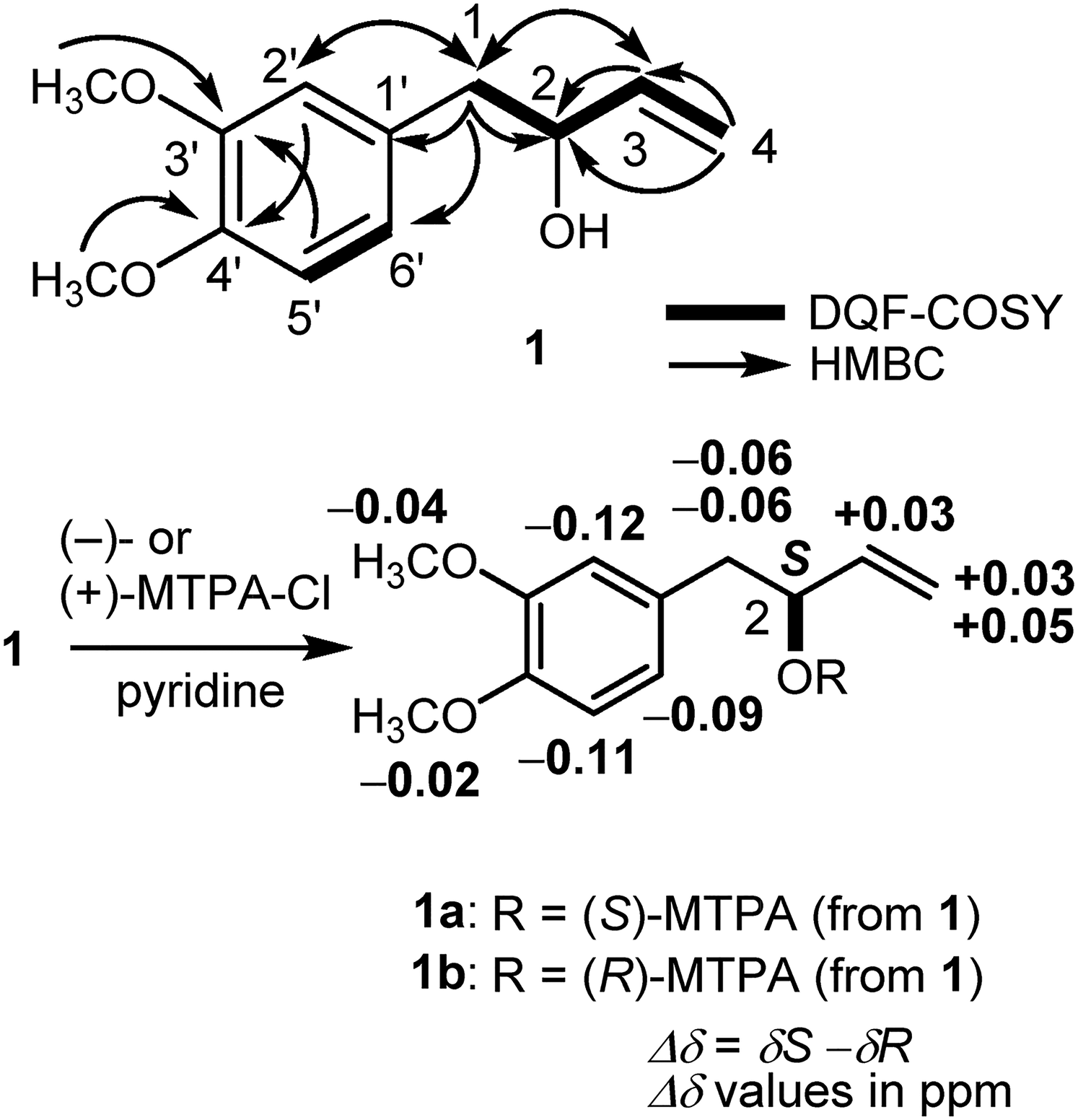
Important 2D NMR correlations and determination of absolute configuration at 2-position of
Cassumunol J (
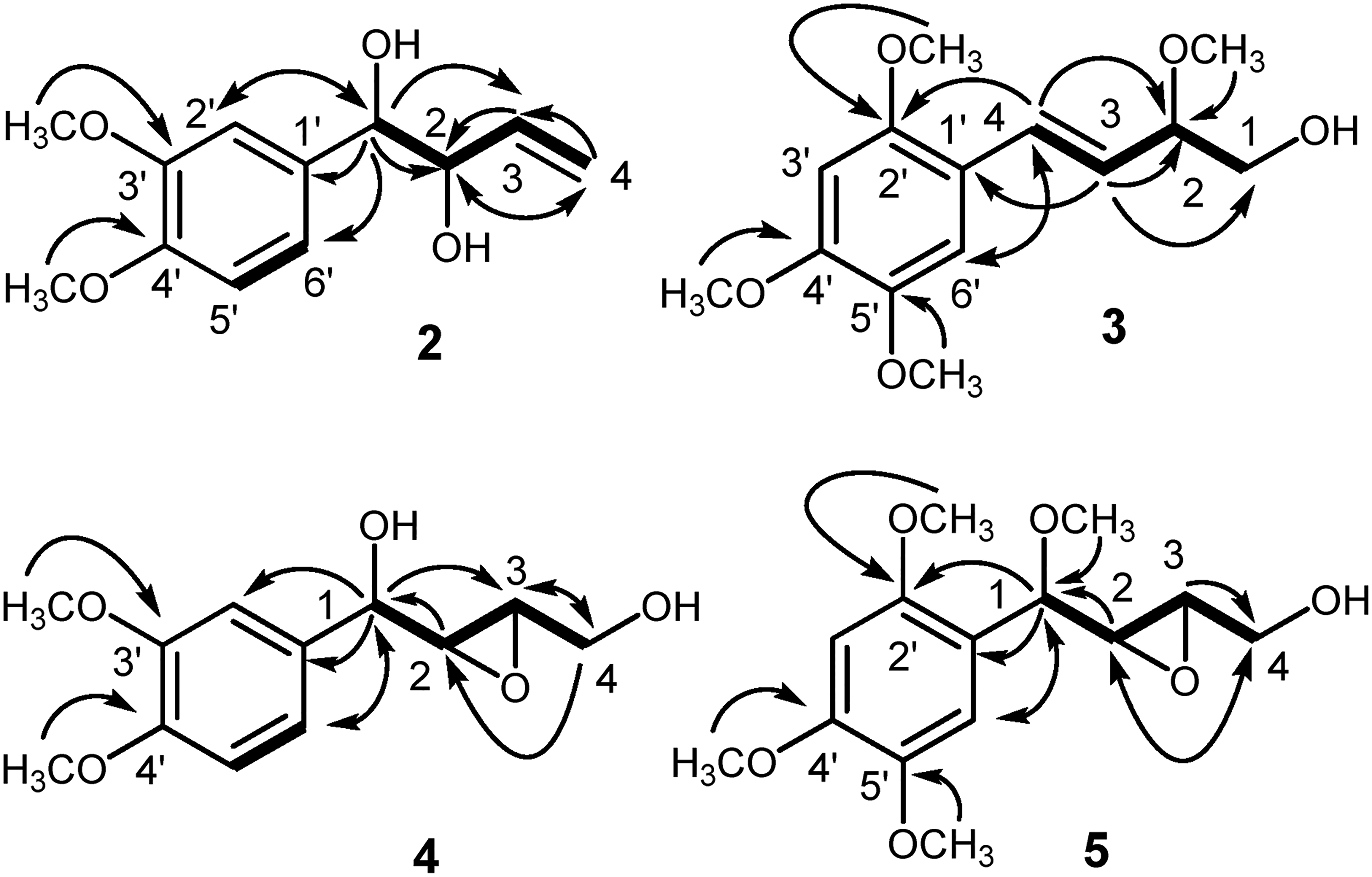
Important 2D NMR correlations of
Cassumunol K (
Cassumunols L (
In conclusion, 5 new phenylbutanoids, cassumunols I–M (
Experimental
General
UV spectra, Shimadzu UV-1600 spectrometer; IR spectra, Shimadzu FTIR-8100 spectrometer; specific rotations, Horiba SEPA-300 digital polarimeter (l = 5 cm); CIMS, EIMS, HRCIMS, and HREIMS, JEOL JMS-GCMATE mass spectrometer; 1H NMR spectra, JEOL JNM-LA 500 (500 MHz), and JEOL JNM-ECA 600 (600 MHz) spectrometers; 13C NMR spectra, JEOL JNM-LA 500 (126 MHz), and JEOL JNM-ECA 600 (151 MHz) spectrometers with tetramethylsilane as the internal standard; HPLC detector, Shimadzu RID-10A refractive index detector; and HPLC columns, YMC-Pack ODS-A (YMC, Inc., 250 × 4.6 mm i.d.) and (250 × 20 mm i.d.) columns for analytical and preparative purposes, respectively. The following materials were used for chromatography: normal-phase silica gel CC, Silica gel BW-200 (Fuji Silysia Chemical, Ltd, 150-350 mesh); reversed-phase silica gel CC, Chromatorex ODS DM1020T (Fuji Silysia Chemical, Ltd, 100-200 mesh).
Plant Material
Z cassumunar rhizomes were cultivated in Nakhon Si Thammarat, Thailand in 2007. The plant was identified by one of the authors (Y. P.). A specimen of the plant is stored in our laboratory.
Extraction and Isolation
A MeOH extract (340.0 g) was obtained from the rhizomes of Z cassumunar (1.4 kg). An aliquot of the extract was partitioned in an EtOAc–H2O (1:1, v/v) mixture to furnish an EtOAc-soluble phase (192.9 g, 16.7%) and an H2O-soluble phase (84.8 g, 7.4%). An aliquot (80.9 g) of the EtOAc-soluble fraction was subjected to normal-phase silica gel CC [2.4 kg, n-hexane-EtOAc → EtOAc → MeOH] to give 6 fractions [Fr. 1, Fr. 2 (15.5 g), Fr. 3 (5.1 g), Fr. 4 (28.6 g), Fr. 5 (10.0 g), Fr. 6 (16.6 g)] as reported previously.5,6
An aliquot (2.5 g) of Fr. 3 was subjected to reversed-phase silica gel CC [75 g, MeOH–H2O (70:30 →90:10, v/v) →MeOH] to give eight fractions [Fr. 3-1 (78 mg), Frs. 3-2 to 3-8]. Fr. 3-1 (78 mg) was purified by HPLC [MeOH–H2O (50:50, v/v)] to give cassumunol K (
Pale yellow oil [α]24D: + 12.4 (c 0.45, CHCl3). IR (film) vmax: 3450, 2930, 1655, 1474 cm−1. UV λmax [CHCl3, nm, (log ε)]: 281 (3.35), 241 (3.55). 1H NMR (500 MHz, CDCl3) δ 3.87, 3.88 [3H each, both s, CH3O-3′, 4′ (interchangeable)], 4.32 (1H, m, H-2), 5.14 (1H, dd, J = 1.4, 10.5 Hz, H-4a), 5.27 (1H, dd, J = 1.4, 17.0 Hz, H-4b), 5.94 (1H, ddd, J = 5.7, 10.5, 17.0 Hz, H-3), 6.76 (1H, d-like, J = 8.9 Hz, H-6′), 6.78 (1H, s-like, H-2′), 6.83 (1H, d, J = 8.9 Hz, H-5′). 13C NMR (126 MHz, CDCl3) δc: given in Table 1 and see Supplemental file. EIMS: m/z 208 (M)+; HREIMS m/z 208.1104 [Calcd for C12H16O3 (M)+, 208.1099]. Pale yellow oil [α]27D: + 7.1 (c 0.16, CHCl3). IR (film) vmax: 3450, 2925, 1655, 1465 cm−1. UV λmax [CHCl3, nm, (log ε)]: 279 (3.74), 231 (4.19). 1H NMR (500 MHz, CDCl3) δ 3.88, 3.89 [3H each, all s, CH3O-3′, 4′ (interchangeable)], 4.23 (1H, dd-like, J = 5.5, 5.5 Hz, H-2), 4.46 (1H, d, J = 5.5 Hz, H-1), 5.17 (1H, d-like, J = 11.0 Hz, H-4a), 5.28 (1H, d-like, J = 17.1 Hz, H-4b), 5.74 (1H, ddd, J = 5.5, 11.0, 17.1 Hz, H-3), 6.84 (1H, d, J = 8.6 Hz, H-5′), 6.89 (1H, dd, J = 1.8, 8.6 Hz, H-6′), 6.91 (1H, d-like, J = 1.8 Hz, H-2′). 13C NMR (126 MHz, CDCl3) δc: given in Table 1 and see Supplemental file. CIMS: m/z 225 (M + H)+; HRCIMS m/z 225.1135 [Calcd for C12H17O4 (M + H)+, 225.1127]. Pale yellow oil [α]23D: −7.4 (c 0.11, CHCl3). IR (film) vmax: 3450, 2925, 1648, 1509 cm−1. UV λmax [CHCl3, nm, (log ε)]: 323 (3.99), 265 (4.18). 1H NMR (600 MHz, CDCl3) δ 3.39, 3.84, 3.87, 3.90 (3H each, all s, CH3O-2, 2′, 5′, 4′), 3.65 (2H, m, H-1), 3.89 (1H, m, H-2), 5.90 (1H, dd, J = 7.8, 15.9 Hz, H-3), 6.50 (1H, s, H-3′), 6.93 (1H, d, J = 15.9 Hz, H-4), 6.98 (1H, s, H-6′). 13C NMR (151 MHz, CDCl3) δc: given in Table 1 and see Supplemental file. EIMS: m/z 268 (M)+; HREIMS m/z 268.1302 [Calcd for C14H20O5 (M)+, 268.1310]. Pale yellow oil [α]26D: + 5.2 (c 0.18, CHCl3). IR (film) vmax: 3450, 2962, 1458, 1262, 972, 841 cm−1. UV λmax [CHCl3, nm, (log ε)]: 280 (3.35). 1H NMR (500 MHz, CDCl3) δ 3.28 (1H, m, H-2), 3.41 (1H, m, H-3), 3.66 (1H, dd, J = 4.3, 12.8 Hz, H-4a), 3.88, 3.90 (s, CH3O-3′, 4′, interchangeable), 3.94 (1H, dd, J = 1.8, 12.8 Hz, H-4b), 4.88 (1H, d, J = 3.1 Hz, H-1), 6.87 (1H, d, J = 8.6 Hz, H-5′), 6.92 (1H, dd-like, J = 1.7, 8.6 Hz, H-6′), 6.93 (1H, d-like, J = 1.7 Hz, H-2′). 13C NMR (126 MHz, CDCl3) δc: given in Table 1 and see Supplemental file. EIMS: m/z 240 (M)+; HREIMS m/z 240.1005 [Calcd for C12H16O5 (M)+, 240.0998]. Pale yellow oil [α]23D: + 1.8 (c 0.33, CHCl3). IR (film) vmax: 3450, 2940, 1509, 1208, 972, 841 cm−1. UV λmax [CHCl3, nm, (log ε)]: 293 (3.69). 1H NMR (500 MHz, CDCl3) δ 3.19 (1H, dd, J = 2.4, 6.1 Hz, H-2), 3.24 (1H, dt-like, J = 2.4, 4.3 Hz, H-3), 3.34, 3.80, 3.86, 3.90 (all s, CH3O-1, 2′, 4′, 5′), 3.55 (1H, dd, J = 4.3, 12.9 Hz, H-4a), 3.88 (1H, m, H-4b), 4.48 (1H, d, J = 6.1 Hz, H-1), 6.53, 6.96 (1H each, both s, H-3′, 6′). 13C NMR (126 MHz, CDCl3) δc: given in Table 1 and see Supplemental file. EIMS: m/z 284 (M)+; HREIMS m/z 284.1268 [Calcd for C14H20O6 (M)+, 284.1260].
Preparation of (S)- and (R)-MTPA Esters (1a and 1b) From 1
A solution of
(S)-MTPA ester ( Pale yellow oil 1H NMR (500 MHz, CDCl3) δ 2.90 (2H, m, H2-1), 3.75, 3.85 [3H each, both s, CH3O-3′, 4′ (interchangeable)], 5.27 (1H, d-like, J = 11.0 Hz, H-4a), 5.37 (1H, d-like, J = 16.5 Hz, H-4b), 5.71 (1H, m, H-2), 5.85 (1H, m, H-3), 6.63 (1H, s-like, H-2′), 6.65 (1H, d, J = 7.8 Hz, H-5′), 6.71 (1H, d-like, J = 7.8 Hz, H-6′). EIMS: m/z 424 (M)+; HREIMS m/z 424.1492 [Calcd for C22H23O5F3 [M]+, 424.1497]. Pale yellow oil 1H NMR (500 MHz, CDCl3) δ 2.96 (2H, m, H2-1), 3.79, 3.87 [3H each, both s, CH3O-3′, 4′ (interchangeable)], 5.24 (1H, d-like, J = 10.3 Hz, H-4a), 5.32 (1H, d, J = 17.8 Hz, H-4b), 5.68 (1H, m, H-2), 5.82 (1H, m, H-3), 6.75 (1H, s-like, H-2′), 6.76 (1H, d-like, J = 8.0 Hz, H-5′), 6.80 (1H, d-like, J = 8.0 Hz, H-6′). EIMS: m/z 424 (M)+; HREIMS m/z 424.1502 [Calcd for C22H23O5F3 [M]+, 424.1497].
(R)-MTPA ester (
Supplemental Material
sj-docx-1-npx-10.1177_1934578X221077823 - Supplemental material for Chemical Structures of Phenylbutanoids From Rhizomes of Zingiber cassumunar
Supplemental material, sj-docx-1-npx-10.1177_1934578X221077823 for Chemical Structures of Phenylbutanoids From Rhizomes of Zingiber cassumunar by Seikou Nakamura, Junko Iwami, Yutana Pongpiriyadacha, Souichi Nakashima, Hisashi Matsuda and Masayuki Yoshikawa in Natural Product Communications
Footnotes
Acknowledgments
This work was supported by JSPS KAKENHI Grant Number 20K07109 (S.N.).
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the JSPS KAKENHI (grant number 20K07109).
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
