Abstract
In the current paper, secondary metabolites separated from Tupistra plants have been reviewed. Approximately 200 phytochemicals, classified in various chemical classes of bioactive compounds, have been compiled, along with their sources and references. The most striking feature is that the most frequently isolated compounds have been spirostanol sapogenins, and spirostanol and furostanol saponins, most of which are new in nature. The application of both Tupistra plant extracts and isolated compounds in biological assays is also one of the crucial aims in pharmacological discoveries. Tupistra constituents have demonstrated valuable properties in the field of pharmacology, such as antioxidative, antimicrobial, antidiabetic, and antihepatic activities, but their cytotoxic and anti-inflammatory actions can be considered as the more remarkable. In vivo cancer-related activities of the tested Tupistra samples were mostly based on apoptosis. Further phytochemical investigations, together with extensive assessments of the biological profiles and mechanism of action studies of the components of Tupistra species are to be expected.
Introduction
Natural products have been shown to possess pharmacological and biological properties that might be beneficial in the treatment of diseases. They can also serve as a source of inspiration for the development of possible new medications. Natural products have been extensively researched in recent decades for their potential applications in medicinal chemistry and molecular science, and as pharmaceuticals. 1
Tupistra (family Asparagaceae) is a genus of flowering plants that can be found in Asia. It contains about 26 species. 2 Plants of this genus are of great value in folk medicine, for example, the rhizome of Tupistra chinensis Baker is always recognized as one of the ingredients for pain attenuation, blood stasis dissipation, detoxification, traumatic injury, and rheumatoid arthralgia. 3 Tupistra nutans Wall. ex Lindl., locally named “Nakima,” is native to India, Nepal, Bhutan, China, and Laos. Its root decoction is used to treat body pain and weakness. 4 Studies on Tupistra plants have shown therapeutically critical evidence, such as cytotoxic, anti-inflammatory, antimicrobial, and antidiabetic activities.4–10 Regarding phytochemical reports, steroids, flavonoids, phenols, and aromatic derivatives can be considered as major secondary metabolites.4–10
Although experimental reports are now abundant, none of the overviews has been discussed so far. The current review aims to provide useful information on the phytochemistry and pharmacology of the genus Tupistra. The data have been retrieved from various resources, but primarily from Google Scholar, Sci-finder, ISI Web of Science, Scopus, Reaxys, and systematic publishers. The terms “Tupistra,” “phytochemistry,” and “pharmacology” were the most meaningful keywords used to search for references.
Phytochemistry
Phytochemical and pharmacological studies of medicinal plants have made substantial progress in recent years, with an emphasis on novel molecules with therapeutic qualities.11–17 The focus of this section is on publications over the last 40 years that deal with phytochemical investigations of Tupistra species. Up to now, approximately 200 compounds with diverse chemical structures have been isolated from the Tupistra genus, but steroid derivatives have predominated. Among the 20 Tupistra species, T. aurantiaca (Baker) Wall. ex Hook. f., T. nutans Wall. ex Lindl., T. wattii (C. B. Clarke) Hook. f., T. yunnanensis F. T. Wang & S.Yun Liang and T. chinensis Baker have been the most studied, especially the last. Table 1 outlines the compound name and the species from which it has been isolated, and Figures 1 to 5 show the chemical structures of these secondary metabolites.

Spirostanol sapogenins from genus Tupistra.
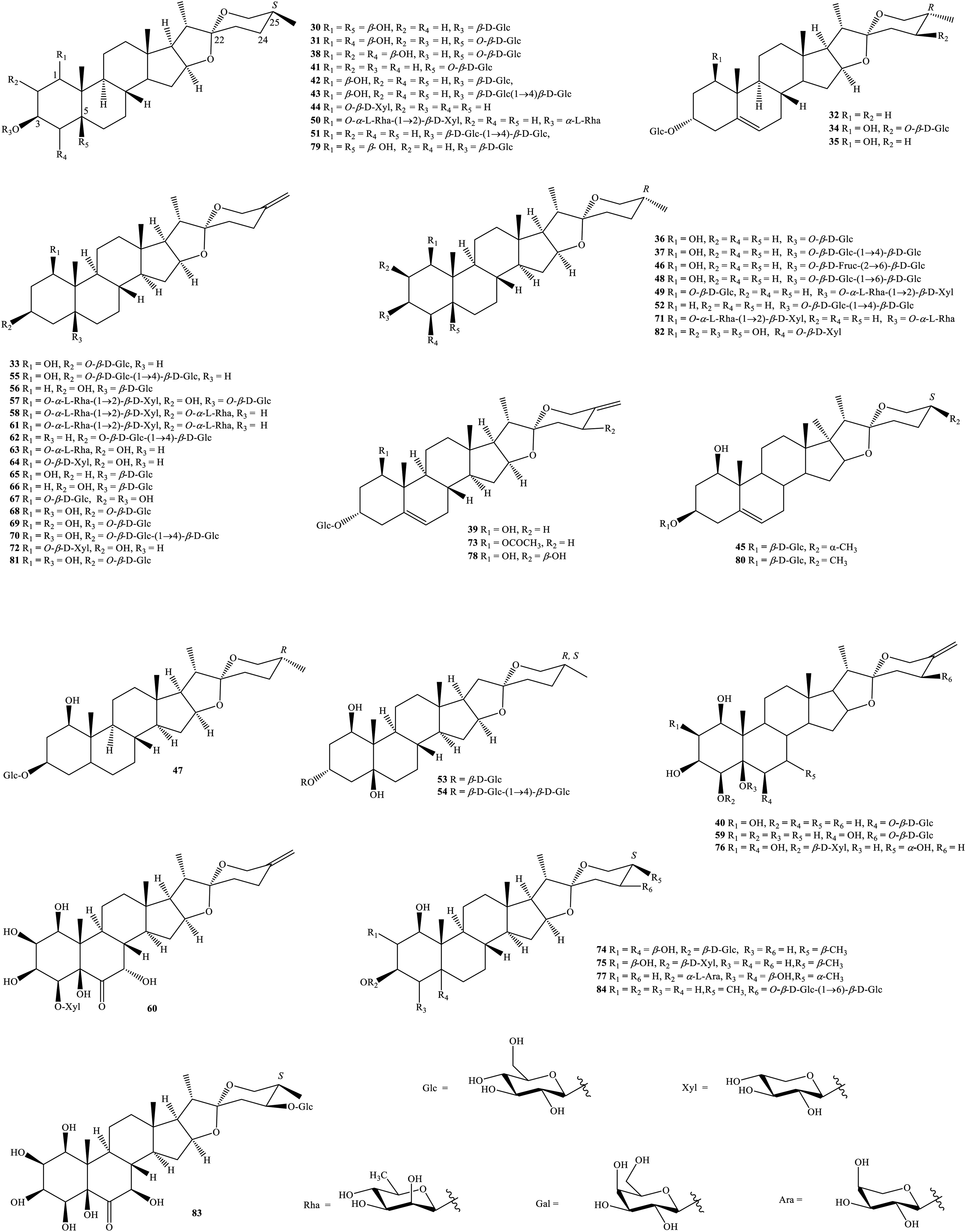
Spirostanol saponins from genus Tupistra.
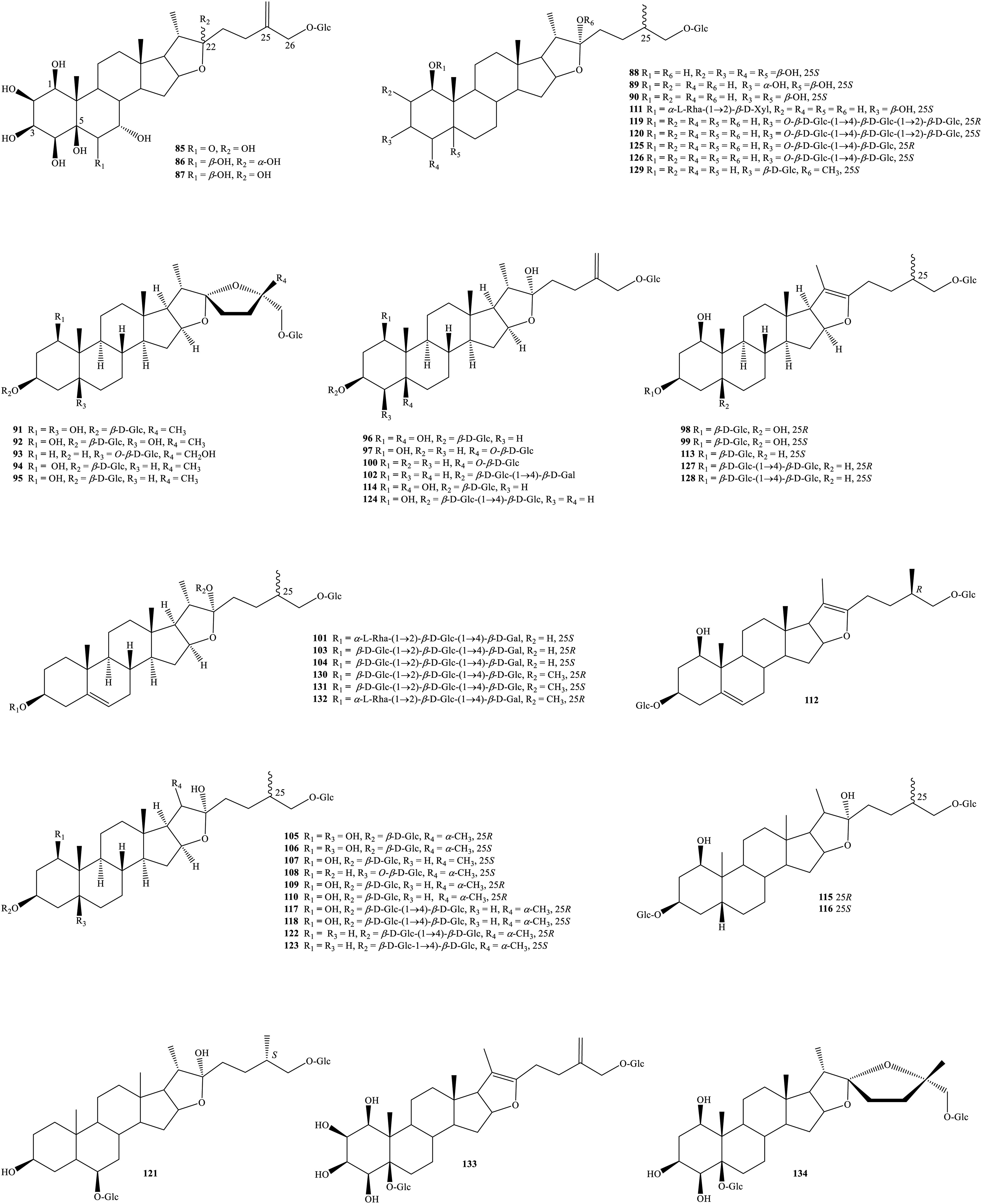
Furostanol saponins, and other types of steroids from genus Tupistra.

Flavonoids, phenols, and aromatic derivatives from genus Tupistra.

Alkaloids, lignans, amides, and triterpenoid from genus Tupistra.
Chemical Constituents From Tupistra Species.
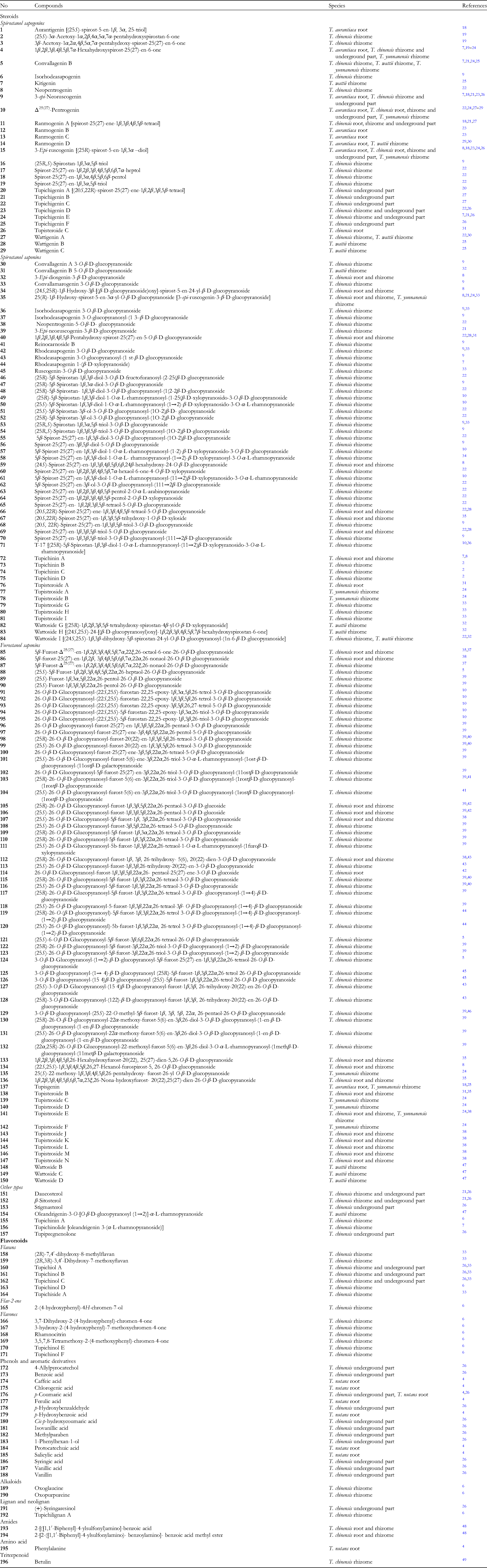
Spirostanol Sapogenins
Steroids structurally formulated with a sterane nucleus are present in both animals and plants. The compounds in this chemical class possess various skeletons such as brassinosteroids, cucurbitacins, bufadienolides, withanolides, sapogenins, and bile acids.
50
Sapogenins are recognized as aglycones, or non-saccharides, some of which can serve as a starting point for semisynthetic steroidal hormones.
51
Sapogenin-type spirostanols are likely to be the main class of secondary metabolites found in Tupistra species. Spirostanol sapogenin derivatives are highly concentrated in the rhizome or underground parts of T. aurantiaca, T. chinensis, T. wattii, and T. yunnanensis; 29 metabolites (
Compound
In 2000 and 2003, Pan et al successfully isolated 5 new sapogenins, trivially named as tupichigenins A-E (
Spirostanol and Furostanol Saponins
Saponins derived from Tupistra plants have 2 parts in their structure. The aglycone has a spirostanol or furostanol nucleus, whereas the glycosyl part is formed of either monosaccharides such as glucopyranose, galactopyranose, xylopyranose, rhamnopyranose, and arabinopyranose or disaccharides such as β-D-glucopyranosyl-(1→4)-β-D-glucopyranoside or α-L-rhamnopyranosyl-(1→2)-β-D-xylopyranoside (Figures 2 and 3). T. chinensis, T. wattii, and T. yunnanensis are main sources of spirostanol and furostanol saponins. Sixty-five compounds (
Taking spirostanol saponins into consideration, glycosylation generally happens at C-1, C-3, C-5, and C-24. Similar to the above spirostanol sapogenins, the saponins have either an R- or S-configuration at carbon C-25. In the genus Tupistra, convallagenin A 3-O-β-D-glucopyranoside (
Thus, the root and rhizome of T. chinensis may be thought of as a rich resource of new spirostanol saponins. In another phytochemical report, apart from the known compound rhodeasapogenin 1-(β-D-xylopyranoside) (
Regarding furostanol saponins, the chemical structures of the isolated compounds (
In 2012 and 2013, Liu et al used silica gel CC and HPLC to isolate and identify the structures of many new saponins from the ethanolic extract of T. chinensis rhizome, such as (25S)-5β-furost-1β,2β,3β,4β,5β,22α,26-heptaol-26-O-β-D-glucopyranoside (
Another report recorded a new furostanol saponin, tupisgenin (
Other Steroids
Other types of steroids have also been proved during the search for bioactive compounds from Tupistra species (Figure 3). Together with major components spirostanol sapogenins, T. chinensis underground part, collected from Taiwan, contained 3 well-known sterols, daucosterol (
Flavonoids, Phenols, and Aromatic Derivatives
Flavonoids play a great role in food chemistry and pharmacology, and terrestrial plants are the main resource of this chemical class.10,17,52,53 Flavonoid derivatives from Tupistra species are present in 3 skeletons, flavans (
Phenols and aromatic derivatives have also been recorded for Tupistra species. Herein, a list of 17 isolated compounds (
Miscellaneous
Other phytochemicals from Tupistra species include 2 alkaloids, oxoglaucine (
Pharmacological Activities
There have been numerous studies using both Tupistra secondary metabolites and extracts for pharmacological examinations, which may be classified into: cytotoxic,3,7,8,10,22,23,26,28,32–36,38,39,41,48,54,55 anti-inflammation,9,10,22,37,39,44,56–58 antimicrobial,4,19 antioxidant, 4 antidiabetic, 4 and other beneficial effects. 56 An overview of previous pharmacological evaluations of Tupistra plants are discussed in detail and outlined in Table 2.
Pharmacological Activities of Isolated Compounds and Plant Extracts From Genus Tupistra.

Abbreviation: LPS, lipopolysaccharide.
Cancer-Related Activities
A great deal of research has shown a strong correlation between saponins and cytotoxic activities, with several specific structural motifs targeting different cancer cells.
59
Herein, both isolated compounds and plant extracts of Tupistra species have been evaluated as cytotoxic candidates due to the high amount of spirostanol saponins. Saponins convallagenin B 5-O-β-D-glucopyranoside (
Spirostanol saponins
Spirostanol saponins seem to be better than furostanol saponins in cytotoxicity assays. Only spirostanol saponin
Besides saponins, steroid derivatives also showed good inhibitory effects on cancer cell lines. Spirostanol sapogenins
Some small molecules derived from Tupistra plants also showed cytotoxic properties. The flavan aglycone exhibited more cytotoxic activity than its glucoside when compared with the inhibitory effect against Human LoVo and BGC-823 cancer cells {
Unrevealing the cytotoxic mechanisms of Tupistra constituents is also crucial in pharmacological studies. Yi et al (2018) proved that 5β-spirost-25(27)-en-1β,3β-diol-1-O-α-L-rhamnopyranosyl-(1→2)-β-D-xylopyranosyl-3-O-α-L-rhamnopyranoside (
Anti-Inflammatory Activities
Anti-inflammatory activity provides basic evidence to confirm the traditionally medicinal application of Tupistra species for the treatment of pharyngolaryngitis and rheumatic diseases. Using the lipopolysaccharide (LPS) stimulated inflammatory syndrome in the abdominal cavity of C57BL/6 mice, furostanol saponins
In the continuing search for potential anti-inflammatory agents from Tupistra species, furostanol saponins, once again, were demonstrated to serve as important components responsible for such activity. Compound
Similar to the cytotoxic activity, the anti-inflammatory results for spirostanol saponins were found to be better than those for furostanol saponins. For example, significant IC50 values ranging from 3.1 to 4.4 μM were assigned to 5 spirostanol saponin derivatives (
Both spirostanols and their saponins derived from T. chinensis species could be considered as promising anti-inflammatory agents. As shown in Table 2, among 23 tested compounds, the spirostanol saponin with the most potential (
Antimicrobial Activities
Recently, bioassay and fractionation of the 95% EtOH extract of T. chinensis rhizome led to the isolation of 4 spirostanol sapogenins
Pharmacological Activities of Extracts
The saponin extract of T. chinensis at a concentration of 10 μg/mL controlled the growth of HeLa (81% inhibition) and HL-60 (92% inhibition) cancer cells. 41 In another study, saponin extract obtained from T. chinensis was investigated for its ability to inhibit the proliferation of ovarian cancer cells by using the MTT assay. The results indicated that this extract, in a dose-dependent manner, significantly inhibited the proliferation of SKOV3 cancer cells through the apoptotic mechanism by suppression of the Wnt/β-catenin signaling pathway. 3 With a high total saponin percentage of 80%, the saponin extract of T. chinensis has also shown cytotoxic activity toward A549, MCF-7, and HeLa cancer cells, with IC50 values of 4.11, 6.47, and 7.78 μg/mL, respectively. The underlying mechanism in human A549 cells was explained by the mitochondria-dependent apoptotic pathway. 54 For sarcoma S-180 cells, T. chinensis saponin extract was examined for in vitro and in vivo inhibitory effects. The in vitro results showed that this extract significantly inhibited S-180 cell growth at doses of 160 and 320 µg/mL after 48 h treatment, while its in vivo mechanism is due to NF-kB signaling suppression in a dose-dependent manner. 55
T. chinensis is renowned for treatment of various diseases such as pharyngolaryngitis, rheumatism, hepatitis, bruises, and stomach pain. 60 This may be due to its anti-inflammatory effects. Saponin extract from T. chinensis at doses of 200 and 400 mg/kg in 5 days treatment prevented mice from LPS-induced death by downregulation of IL-1β and TNF-α expressions. 58 In both in vivo and in vitro models, the 70% EtOH extract of T. chinensis, in a concentration-dependent manner (75, 150, 300 mg/kg), caused a decrease in levels of inflammatory cytokines TNF-α and IFN-γ, and also controlled serum transaminases and lactic dehydrogenase levels in Con A-induced hepatitis in mice. 57
T. chinensis water extract at a dose of 10 g/kg was reported to significantly inhibit acetic acid-induced capillary permeability and xylene-induced ear edema in mice. 60 In another pharmacological experiment, against isoproterenol-induced myocardial injury in mice, the n-butanol and ethyl acetate extracts of T. chinensis plant showed a higher effect in comparison to those obtained from water and light petroleum extracts. 56 Therefore, it can be concluded that medium polar extracts of T. chinensis containing phenols and aromatic derivatives seem better than strong or weak polar extracts.
Among Tupistra species, T. nutans has strong antioxidative activity due to its high phenolic content. The DPPH radical scavenging capacity of T. nutans extracts was in the order ethyl acetate (IC50 21.31 µg/mL) > n-butanol (65.15 µg/mL) > n-hexane (168.88 µg/mL) > methanol (184.05 µg/mL) > water (309.41 µg/mL). 4
T. nutans is also a potential antimicrobial agent. Its 70% MeOH root extract at concentrations of 0.12 to 0.25 mg/mL induced inhibitory zones between 10.17 and 19.16 mm against 7 microorganisms, Staphylococcus aureus, Salmonella enterica, Escherichia coli, Candida albicans, Aspergillus niger, A. flavus, and A. fumigatus; mycostatin (1 mg/mL), the positive control, produced inhibitory zones of 8.00 to 18.00 mm. 4
To date, T. nutans is the only Tupistra species that has been tested for antidiabetic activity. Among 5 extracts (n-hexane, ethyl acetate, n-butanol, methanol, and water), its ethyl acetate extract established the best α-glucosidase inhibitory percentage of 81.68%; the rest showed 60.01% to 73.49% inhibition, as compared to that of the positive control acarbose (98.10%). 4 The ethyl acetate extract has the highest phenolic (51.20 mg/g GAE) and flavonoid (15.20 mg/g QE) contents. 4
Conclusion
For the first time, the current review has provided a general overview of the phytochemistry and pharmacology of the genus Tupistra. Phytochemical studies of Tupistra species have resulted in the isolation and structural elucidation of about 200 secondary metabolites. These compounds are of various chemical classes, but steroids, flavonoids, and simple phenols have been considered as major components. The characteristic steroids are spirostanol sapogenins, and spirostanol and furostanol saponins. The different parts of T. chinensis have been used the most frequently in both phytochemical and pharmacological studies. Tupistra saponins are promising agents for cancer related and anti-inflammatory activities, and spirostanol saponins are generally found to be better than furostanol saponins for these properties. Some spirostanol saponins were better than the positive controls in assays, for example, saponins 25(R)-1β-hydroxy-spirost-5-en-3α-yl-O-β-D-glucopyranoside (
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
