Abstract
Abstract
The main objective of this study was to investigate the antibacterial activity and mechanism of Litsea cubeba essential oil (LCEO) against Acinetobacter baumannii. The antibacterial activity was examined by a serial dilution method and growth curves. The essential oil showed strong activity against A. baumannii. The mechanism of the antibacterial action was evaluated by the integrity and permeability of the membrane, scanning electron microscopy (SEM) and SDS-PAGE. The change in permeability of the cell membrane and leakage of cell intracellular biomacromolecules verified that LCEO has an obvious effect on the cell membrane. SEM showed the damaging effect of the essential oil on cells since the morphology of the treated bacteria was significantly changed. Different expression of proteins indicated the effect of the essential oil on protein synthesis. It is suggested that LCEO with both emulsifying and antibacterial activities, has a potential to serve as an inhibitor against A. baumannii.
Multidrug resistance among nosocomial pathogens is a major threat to public health and poses a huge economic burden on global health care. 1 Antibiotic resistance kills about 7 00 000 people each year worldwide. 2 Especially the Gram-negative carbapenem-resistant Acinetobacter baumannii raises fears of untreatable infections. The WHO declared A. baumannii as one of the most serious ESKAPE organisms (Enterococcus faecium, StaphylococcAus aureus, Klebsiella pneumoniae, Acinetobacter baumannii, Pseudomonas aeruginosa, and Enterobacter species) that effectively escape the effects of antibacterial drugs. 3 Therefore, finding new antimicrobial drugs is desperately needed. In recent years, essential oils have attracted lots of scientific interest because they exhibit a wide spectrum of bioactivities, such as antibacterial, antifungal, antiviral, antioxidant, and insecticidal activities.
Litsea cubeba is a plant widely distributed in Southeast Asia and Southern China. 4 L. cubeba essential oil (LCEO), extracted from the fruit peel, has an intense lemon-like aroma; the oil is insoluble in water. 5 LCEO has been widely used in the food, medical, chemical and other industries. 6 Due to either the synergistic or antagonistic effects of the constituent components of LCEO, it has antibacterial, 7 antiseptic, 4 anti-oxidant, 8 and other biological activities, and so has a good application prospect in the field of food preservation. In addition, LCEO has anticancer, 9 -11 and insect repellent effects. 12 -14 However, there are few reports on the inhibitory effect and mechanism of the oil on Acinetobacter baumannii and other pathogenic bacteria.
The aim of this study was to evaluate the antibacterial activity of LCEO, and explore its mechanisms of antibacterial action against multidrug-resistant A. baumannii by scanning electron microscopy (SEM), growth curve assays and cell membrane permeability, as well as the integrity of the cell membrane. We also studied the effect of LCEO on bacterial protein expression by SDS-PAGE.
Results
Antibacterial Activity of LCEO
The surfactant Tween 80 and absolute ethanol had no effect on the growth of A. baumannii. The MIC of LCEO was determined as 1.04 mg/mL, and the MBC 4.17 mg/mL.
Growth Curve Studies
Figure 1 shows the growth curves of A. baumannii determined by measuring the optical density (OD) at 600 nm. The growth curve for the control group shows an “S” trend, with a fast increase in bacterial numbers. On the contrary, the OD values of the groups which were treated with the essential oil (1 × MIC, 2 × MIC) were significantly lower than those of the control group, with few changes in the curve. The results showed that the different concentrations of LCEO can almost completely inhibit the growth of the target bacterium.

Growth curves of A. baumannii.
The Effect of LCEO on Morphological Change
The effect of LCEO on morphological change was evaluated using SEM; the results are shown in Figure 2. Compared with the untreated bacteria, the LCEO treated cells had obvious morphological change. The cells which had been exposed to LCEO at 1 × MIC showed a considerable morphological change, with damaged cell walls, shriveled cell morphology, and irregular shape. The effect was enhanced with increasing concentration of the essential oil as the cells treated with LCEO at 2 × MIC were more severely damaged. In contrast, the untreated bacteria showed regular and intact morphology, with a plump surface, and were uniform in size and shape.
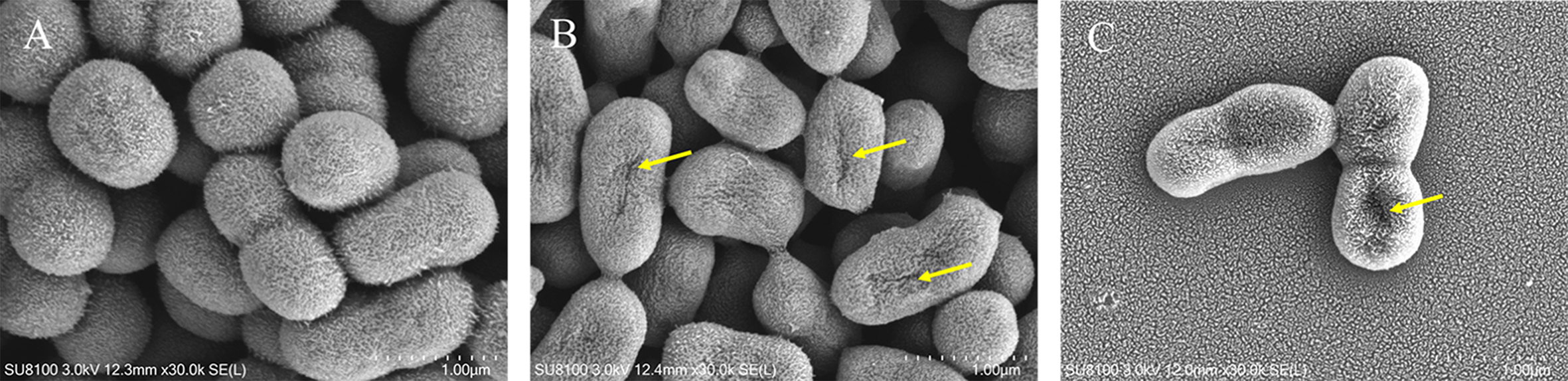
Effect of LCEO on morphology of A. baumannii. (A) Untreated (B) Treated with LCEO at 1 × MIC; (C) Treated with LCEO at 2 × MIC. Arrows show the shriveled appearance and holes on the cell surface.
The Effect of LCEO on AKP Concentration
Figure 3 shows the AKP concentration in the bacterial culture solution after treatment with LCEO for increasing periods of time; this indicated the extent of damage to the cell wall of the bacteria by LCEO. Compared to the control group, the amount of AKP that leaked into the culture medium increased, and reached a maximum after 2 hours in the groups to which different concentrations of LCEO had been added. The leakage from each treatment group was higher than that of the control group and the amount leaked into the culture medium of the 2 × MIC group was the highest at every time point. In the control group, the leakage of AKP was at a low level and had no significant changes.
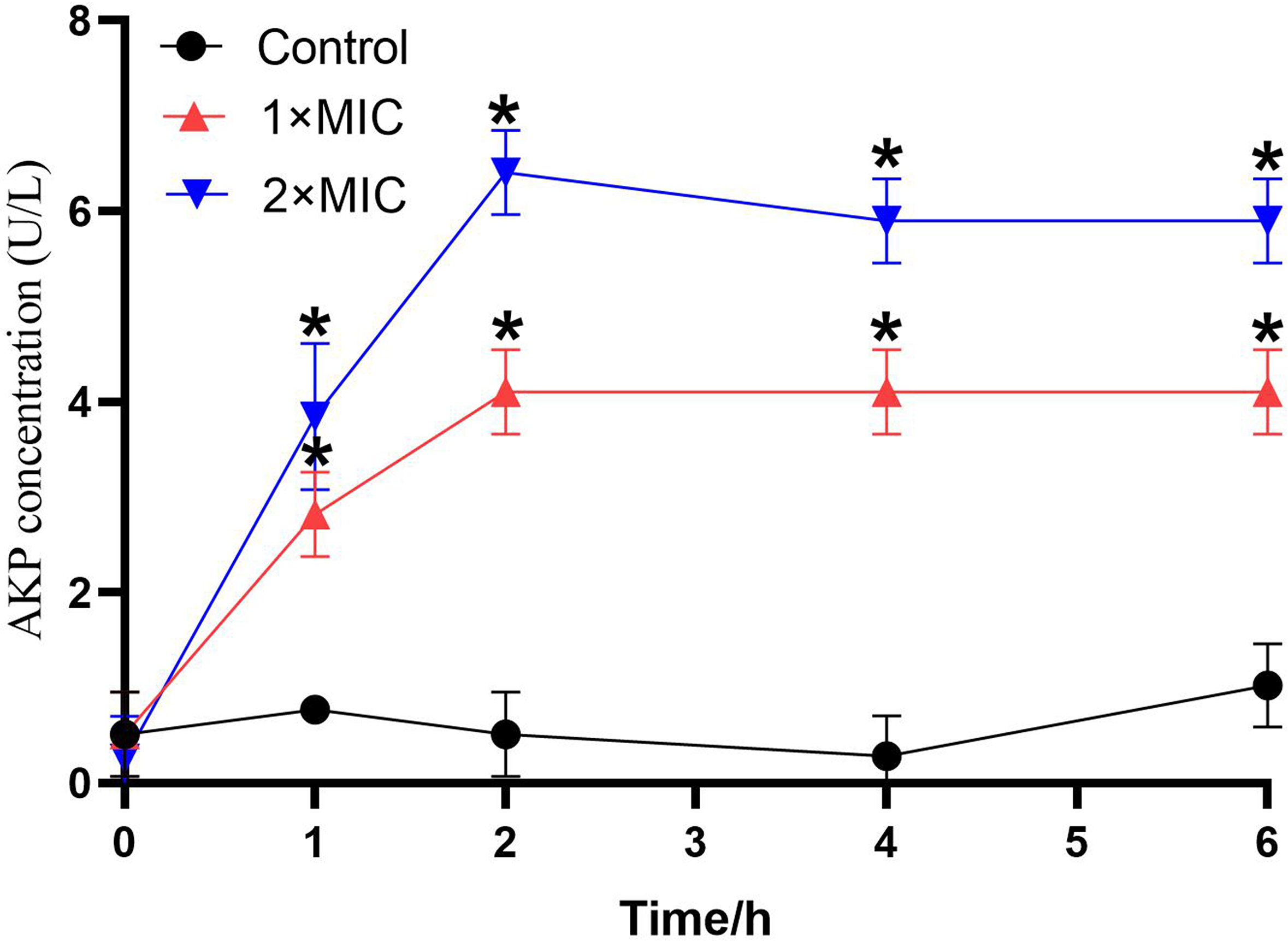
The effect of LCEO on A. baumannii cell wall. *P < 0.05 vs the control groups.
The Effect of LCEO on Cell Membrane Permeability
The change in the permeability of the cell membrane can be reflected by the change in the conductivity of the bacterial suspension. As shown in Figure 4, compared to the treatment groups, the increase in relative electric conductivity of the control was much lower. The relative electric conductivity of the treatment groups kept increasing and there was a sharp increase in the first 2 hours, which corresponded to the leakage of AKP, as shown in Figure 3. The increase in relative electric conductivity of the bacteria treated with LCEO at 2 × MIC was higher than that treated with LCEO at 1 × MIC.
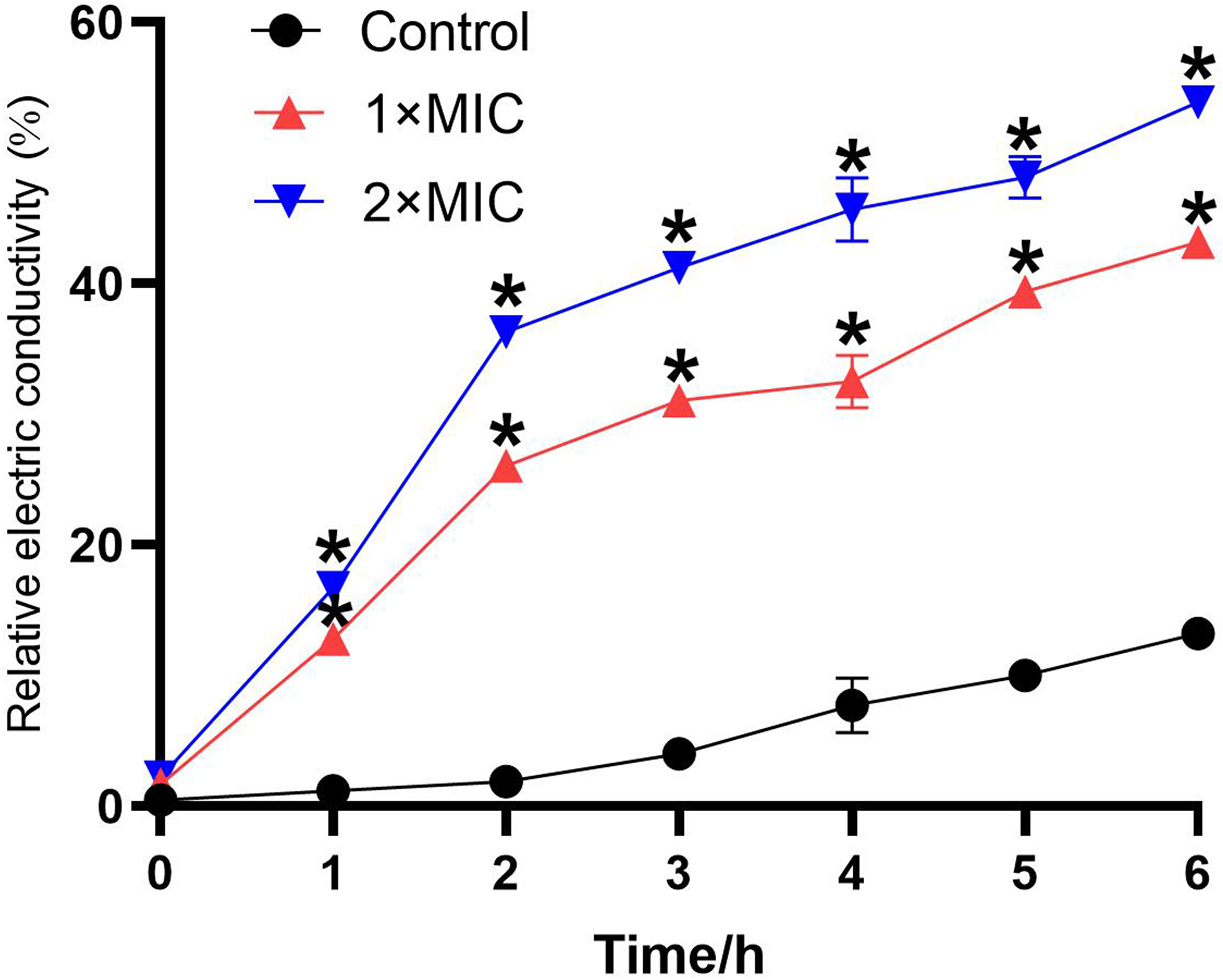
Effect of LCEO on the permeability of A. baumannii. *P < 0.05 vs the control groups.
The Effect of LCEO on Integrity of the Cell Membrane
Nucleic acid and protein of macromolecules in the cell have maximum absorption peaks at 260 nm and 280 nm, respectively. Figure 5 shows the leakage of nucleic acid and proteins of the bacteria treated with different concentrations of LCEO (1 × MIC, 2 × MIC). The OD260 value of the control group showed few changes during the 6 hours of incubation, as shown in Figure 5(A). However, the value for the treatment groups continued to increase as the culture time increased, and the increase for the bacteria treated with LCEO at 2 × MIC was higher than that for those treated with LCEO at 1 × MIC. A similar situation was exhibited for the leakage of the proteins (Figure 5(B)).

Effects of LCEO on the integrity of the cell membrane of A. baumannii. (A) OD260, (B) OD280. *P < 0.05 vs the control groups.
The Effect of LCEO on Protein Synthesis of the Bacteria
SDS-PAGE profiles of bacterial proteins from A. baumannii treated with LCEO are shown in Figure 6. The size of the tested proteins ranged from 10 kDa to 180 kDa. Compared to the control group, the protein bands of the bacteria which had been treated with LCEO were much fainter. As the concentration of essential oil increased and the duration of treatment increased, the effect of LCEO was more obvious. From 55 kDa to 100 kDa, the bands of the treated groups showed stronger intensities than those of the control group.

SDS-PAGE of bacterial cells treated with LCEO. Lane M:Marker; Lane 1&4:Untreated bacteria; Lanes 2&3:LCEO-treated bacteria at 1 × MIC and 2 × MIC, respectively, after 3 hours; Lane 5&6:LCEO-treated bacteria at 1 × MIC and 2 × MIC, respectively, after 6 hours.
Discussion
This study explored the antibacterial activity and its mechanism of LCEO against A. baumannii. Previous studies reported that LCEO had effective antibacterial activity against MRSA, the MIC and MBC values for which were 0.5 mg/mL and 1.0 mg/mL respectively, and the bacterial population treated with LCEO at MIC concentration declined by 99.99% in 2 hours. 7 In this study, the MIC and the growth curves of A. baumannii indicated that LCEO had a significant inhibitory activity and could almost completely inhibit the growth and reproduction of A. baumannii, which showed that LCEO had a potent antibacterial effect on A. baumannii. However, the MIC value of LCEO against A. baumannii was higher than that against MRSA, thus LCEO had better activity against Gram-positive bacteria than Gram-negative bacteria, which was consistent with the results of previous research. 15 That Gram-negative bacteria show a higher degree of resistance against LCEO might be attributed to the outer membrane of the bacteria, which have a hydrophilic lipopolysaccharide structure. 16 Owing to this structural property, they could prevent hydrophobic compounds from penetrating into the target cell membrane. This may explain why Gram-negative bacteria are relatively resistant to hydrophobic antibiotics.
The SEM images clearly showed the damage to the morphology of A. baumannii. The morphological changes may result from the leakage of the intracellular substances. Also, the leakage of the contents such as proteins and nucleic acid could impact the metabolic activities of the bacteria, which could lead to cell death. A similar effect on MRSA treated with LCEO was reported in previous work; the surfaces of MRSA treated with LCEO were rough and uneven, which was consistent with the results of this experiment. 7
AKP is an enzyme that exists between the cell wall and cell membrane and is undetectable when the bacterial structure is complete. Once either the cell wall or cell membrane is damaged, AKP leaks out of the cell because of the increasing permeability. 17 The results showed that the AKP concentration in the bacterial culture solution increased in the first 2 hours, then remained stable, which revealed the damage caused by LCEO on the cell wall of the target bacteria. The cell membrane is an important barrier which can block extracellular material entering the cell, and maintain the relative stability of the intracellular environment. 18 When the cell membrane of bacteria is destroyed, the internal electrolytes leak into the culture medium, which causes the conductivity of the culture medium to increase. 19 In this study, an obvious increase in conductivity was observed, which indicated that LCEO could damage the bacterial cell membrane structure, leading to the leakage of components and a change in the permeability. The effect of LCEO was significant at the initial stage, which was consistent with the results of AKP leakage.
As the previous work reported, nucleic acid and protein play important roles in bacterial metabolism for they dominate the genetic information and cellular structure. 20 We found an obvious increase in the optical density at 260 nm and 280 nm in the cultures that were treated with LCEO (1 × MIC and 2 × MIC), which indicated that it can disrupt the integrity of the membrane, contributing to the leakage of nucleic acid and proteins through the membrane. Proteins are necessary substances of life, and when the activity of protein synthesis is inhibited, normal physiological metabolism of bacteria is affected, leading to cell death. 21 In our study, 2 new bands (approximately 85 kDa, and 100 kDa) appeared in the lane of the treated bacteria, but the band at approximately 85 kDa disappeared with increasing treatment time. Moreover, the protein bands of the bacteria got much fainter after being treated with LCEO, and the effect was more significant with the extension of time. The results indicated that LCEO might have an effect on cellular proteins either by interfering with synthesis by bacteria or by disrupting them, or result in the leakage of proteins from bacterial cells. The reason for the new protein bands probably is that the bacteria are stimulated by the external environment, and their own protein synthesis increases to perform corresponding biological functions.
Conclusion
This study reports the antibacterial activity and mechanism of LCEO against A. baumannii. The essential oil showed significant antibacterial activity against A. baumannii. The results indicated that LCEO could damage the cell wall and cell membrane, which leads to leakage of AKP, nucleic acid and proteins, and an increase in electric conductivity. In addition, the effect of LCEO on cell morphology was confirmed by the results of the SEM experiment. The SEM images showed many of the treated bacteria as irregularly deformed and shrunken. In conclusion, this study showed that LCEO is a potential inhibitor of A. baumannii. As the major component of the essential oil, citral might be responsible for the antibacterial activities. However, because the essential oil of Litsea cubeba has many kinds of chemical constituents, it seems impossible that there is only one component that has an antibacterial effect or only one antibacterial mechanism. Therefore, further study, such as separating new chemical compounds, and in vivo antibacterial activity are necessary to investigate fully the antibacterial mechanism of the essential oil against A. baumannii.
Material and Methods
Bacterial Strains and Chemicals
LCEO, extracted from the fruit of LC by steam distillation by the manufacturer, was purchased from a local agency (Nanning, China). The chemical composition of the oil was provided by the manufacturer (Table 1). The LCEO microemulsion system was constructed on a pseudoternary phase diagram at 25 ℃. The composition was LCEO: ethanol: Tween 80 = 1:2:4 (w/w/w). LCEO (0.1 g), ethanol (0.2 g) and Tween 80 (0.4 g) were mixed well, then diluted with water to 3 ml. The physical stability of the microemulsion was determined according to the method of Al-Adham et al., 22 with some modification. After centrifugation at 5000 × g for 10 minutes at 20 °C, no phase separation was observed in the sample. Then, the same sample was stored at 28 °C for 1 month, and no phase separation was observed.
Chemical Composition of LCEO.

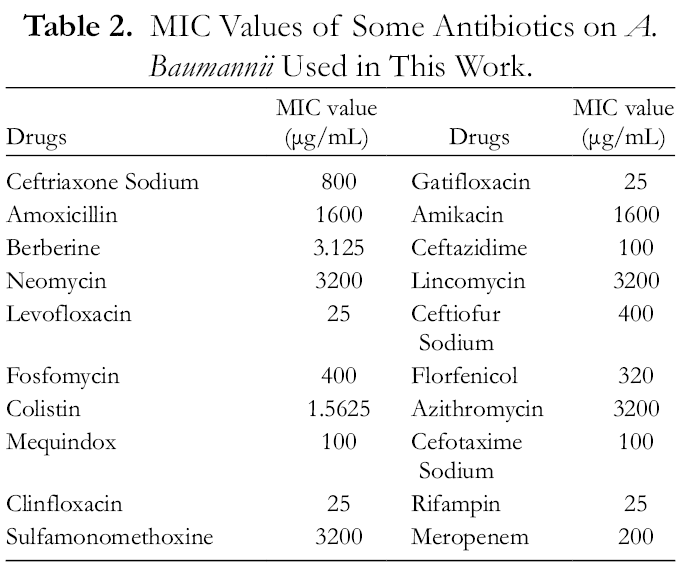
The Acinetobacter baumannii used for this work was provided by The First People’s Hospital of Nanning, China. The strain was highly resistant to common antibiotics such as amoxicillin, amikacin, neomycin, sulfamonomethoxine, lincomycin, and azithromycin (Table 2, provided by The First People’s Hospital of Nanning).
MIC Values of Some Antibiotics on A. Baumannii Used in This Work.
The strain was stored in tryptic soy broth (TSB) containing 30% glycerol and stored in the refrigerator at −80 °C. One mL A. baumannii was taken from the refrigerator, inoculated into TSB, and cultured, with shaking, at 220 rpm for 24 hours at 37 °C to activate the strain.
Determination of MIC and MBC
The minimum inhibitory concentration (MIC) was determined using a microdilution experiment based on the M7-A8 method from the Clinical and Laboratory Standards Institute. Two-fold serial dilutions of samples were prepared in sterile Mueller-Hinton broth ranging from 0.033 to 16.667 mg/mL. Afterward, the 50 µL solution was mixed with 50 µL bacterial suspension (approximately 106 CFU/mL) in 96-well plates. A negative control (without bacteria solution) and a positive control (without drug) were also performed to ensure the growth of the microorganism and the sterility of the medium during the experiment. The groups with Tween 80 and absolute ethanol were marked as blank controls. The microdilution plates were incubated at 37 °C for 18 hours, MIC was defined as the concentration of the lowest serial dilution of the EO which completely inhibited bacterial growth after the incubation period. All assays were performed in triplicate, and the results were expressed in mg/mL.
Minimal bactericidal concentration (MBC) was determined from the microdilution plates used for the MIC assay. Fifty µL solution was taken from each well without visible growth and transferred to tryptic soy agar (TSA) plates and incubated at 37 °C for 24 hours. MBC was defined as the lowest concentration of the EO which completely inhibited bacterial growth on the plates. All assays were performed in triplicate, and the results were also expressed in mg/mL.
Determination of Bacterial Growth Curves of A. Baumannii
The growth curves of A. baumannii were determined using an ultraviolet spectrophotometric method. 23 Firstly, bacteria cultured to the logarithmic growth phase were inoculated in LB medium, and adjusted to approximately 105 CFU/mL. Then, LCEO was added to the medium at a final concentration of 1 × MIC and 2 × MIC, and an equal volume of absolute ethanol was used as a control. Afterward, the bacteria were cultured at 37 °C with shaking at 220 rpm for 24 hours. Finally, the absorbance at 600 nm was determined by ultraviolet spectrophotometry every 2 hours.
SEM Analysis of Morphological Changes
The effect of LCEO on the morphology of A. baumannii was observed by SEM according to the method of Bajpai, 24 with some modification. Overnight cultures of both control and treated samples were centrifuged at 4000 × g for 10 minutes and washed with 0.1M phosphate buffer solution (PBS, PH = 7.4), 3 times. Then, the cells were transferred into glutaraldehyde (2.5%) at 4 °C for 4 hours to fix the samples. After washing 3 times with PBS, the cells were dehydrated in sequentially graded ethanol (50% to 100%). Finally, ethanol was replaced by 100% tertiary butyl alcohol. Samples were sputter-coated with gold in an ion coater for 2 minutes, and observed by SEM.
Cell Wall Damage Assay
The effect of LCEO on the cell wall was determined by the leakage of alkaline phosphatase. 25 LCEO was added to the target bacterial suspensions (approximately 106 CFU/mL) to obtain final concentrations of 0, 1 × MIC and 2 × MIC, and then incubated at 37 °C with shaking at 220 rpm. The bacterial sample was taken at the specific time and centrifuged at 3500 × g for 10 minutes. The supernatant was used to determine AKP activity using an AKP kit (Nanjing JianCheng Technology Co. Ltd., Nanjing, China), according to the manufacturer’s instructions.
Cell Membrane Permeability
The effect of LCEO on cell membrane permeability against A. baumannii was determined according to the method of Kong by measuring the relative electrical conductivity. 26 The bacterial strain was cultured at 37 °C for 6 hours, then centrifuged at 4000 × g for 10 minutes, followed by washing with 5% glucose solution (w/v) until the electrical conductivity came close to that of 5% glucose solution, at which point the bacterial solution was isotonic. The conductivity of the bacterial isotonic solution treated in boiling water for 5 minutes was marked as L0. The conductivities after essential oil had been added to 5% glucose at different concentrations (1 × MIC, 2 × MIC) were marked as L1. Essential oil at different concentrations (1 × MIC, 2 × MIC) was added to the bacterial isotonic solution and cultured at 37 °C for 6 hours, and the conductivity of the treated bacteria was measured and marked as L2; an equal volume of absolute ethanol was added as control. The cell membrane permeability was calculated using the following formula:

Integrity of the Cell Membrane
The integrity of the cell membrane could be determined by examining the release of the cell constituents. 27 The bacterial strain was cultured to the logarithmic growth phase and centrifuged at 4000 × g for 10 minutes, then washed with sterile PBS 3 times and re-suspended in sterile PBS. The bacteria were cultured in different concentrations of LCEO (1 × MIC, 2 × MIC) at 37 °C with shaking. Then, 2 ml of each sample was centrifuged at 8000 × g for 5 minutes, and the amount of DNA and RNA released from the cytoplasm to the supernatant was measured by the detection of absorbance at 260 nm; an equal volume of absolute ethanol was used as control. Correction was made for absorption of the suspension with the same PBS containing the same concentration of the oil after 2 minutes of contact with the tested strain. The untreated cells were corrected with PBS only. The leakage of the protein was measured using the same method at 280 nm. Data were taken for each time-point, in triplicate.
SDS-PAGE of Whole-Cell Proteins
SDS-PAGE of the bacterial protein was performed according to the method of Li, 28 with some modification. The bacterial strain was incubated at 37 °C with shaking for 6 hours, then different concentrations of LCEO (1 × MIC, 2 × MIC) were added to the bacterial solution and the mixture cultured continuously; an equal volume of absolute ethanol was used as control. The cells were collected by centrifugation at 4000 × g for 10 minutes, washed with sterile PBS 3 times, re-suspended in the same buffer, and then the solutions were adjusted to the same cell density (OD600). An equal volume suspension was collected by centrifuging at 4000 × g for 10 minutes; the pellets were re-suspended in sample dilution buffer. The samples were placed on ice for 10 minutes, then heated in boiling water for 10 minutes. Finally, the samples were subjected to SDS-PAGE using a vertical electrophoresis apparatus. The SDS-PAGE was performed with a 5% stacking gel and a 12% separating gel, followed by Coomassie brilliant blue staining.
Statistical Analysis
All the assays were carried out in triplicate. The data were recorded as mean ± standard. A general analysis of variance (ANOVA) and Duncan’s multiple range tests were performed. Values of P < 0.05 were considered to be statistically different.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (31760746) Science and Technology Major Project of Guangxi (China) (AA17204057), Key Research and Development Plan of Guangxi (AB19245037) and Innovation Project of Guangxi Graduate Education 725 (YCSW2020007).
