Abstract
This work analyzed the chemical compositions and evaluated the antioxidant and cytotoxic activities of essential oils (EO) of Eugenia patrisii (Epat), Eugenia stipitata (Esti), Myrcia splendens (Mspl), Myrcia sylvatica (Msyl), Psidium guajava (Pgua), and Psidium guineense (Pgui-1 and Pgui-2) from the Brazilian Amazon. Sesquiterpenoids were found in high concentrations in the oils of E. patrisii and M. splendens, which were rich in E-caryophyllene (32.0% and 45.8%); E. stipitata and M. sylvatica, which displayed germacrene D (11.8%) and germacrene B (24.5%); and P. guajava that showed epi-β-bisabolol (16.1%) as the main compound. However, P. guineense samples (Pgui-1 and Pgui-2) were rich in monoterpenoids such as limonene (Pgui-1: 30.2%; Pgui-2 30.4%) and α-pinene (Pgui-1: 22.5%; Pgui-2: 17.7%). The samples showed a weak and moderate antioxidant activities in the DPPH assay, displaying inhibition rates from 11.5% to 38.6% (at 10 mg/mL). All samples were cytotoxic against human cancer cells by the MTT method. Epat oil showed higher activity against melanoma (SKMEL-19, IC505.8 µg/mL), gastric (AGP01, IC503.2 µg/mL), and colon (HCT116, IC506.7 µg/mL). Meanwhile, the samples Pgua and Pgui were more active against breast cancer cells (MCF7, IC5012.4 µg/mL and 11.6 µg/mL, respectively).
Cancer represents a significant public health problem and affects around 18 million people worldwide, and caused more than 9.6 million deaths in 2018. 1 In Brazil, the greatest number of deaths are related to cancers of the breast, stomach, colon, and rectum. 2 Together they represent about 23% of the total of more than 224 thousand deaths caused by cancer. 2
The cause of uncontrolled multiplication of the healthy cells is mainly related to oxidative stress, a state generated by an imbalance of reactive oxygen species (ROS) released during mitochondrial respiration. There is a higher concentration of ROS compared to cellular antioxidants. 3
The loss of redox homeostasis by cells can trigger destructive processes in biomolecules that are influenced by superoxide (O2 -), hydroxyl (OH●), and hydrogen peroxide (H2O2), such as the breakdown of nucleic acids (DNA and RNA), enzymatic inactivation, polysaccharide depolymerization, lipid peroxidation, in addition to also causing oxidation of carbohydrates and proteins. 3,4
Despite advances and improvements in diagnosis and monitoring, overall cancer survival rates remain elevated. There has been an increase in targeted molecular therapies to develop better clinical outcomes for cancer patients. Unfortunately, these strategies did not provide substantial improvement due to the development of resistance to therapies. In this context, phytochemicals have received considerable attention due to their diverse pharmacological properties, including cytotoxicity, with minimal or no toxicity in normal cells as well as providing cancer chemopreventive effects. 5
Therefore, the anticancer potential of essential oils of Myrtaceae from Brazil has been widely studied, aiming to mitigate the resistance development to multiple drugs and side effects of antitumor drugs currently used. 6 Essential oils extracted from Eugenia egensis DC., Eugenia flavescens DC., Eugenia patrisii Vahl, Eugenia polystachya Rich., Eugenia uniflora L., and Psidium myrtoides O. Berg showed cytotoxic activity against human cancer cell lines. 6,7
Myrtaceae are generally woody shrubs or tree species, mostly perennial, and commonly distributed in the tropics and subtropics. 8 Among the 132 Myrtaceae genera, Eugenia, Myrcia, and Psidium are the largest in the American and African continents, with more than 1400 occurring in different biomes. 6,9,10
Eugenia and Psidium fruits are consumed in nature or used in desserts, juices, creams, and ice creams. They are also used in traditional Brazilian medicine, such as Psidium guineense Sw. against inflammatory diseases and Eugenia stipitata McVaugh to treat intestinal, urinary disorders, and relieve cold symptoms. 11,12 On the other hand, Myrcia species are commonly used as phytopharmaceuticals to prepare teas to treat diabetes, hypertension, diarrhea, and compresses form to treat inflammation and skin infections. 13,14
Essential oils have been shown to possess a wide range of anticancer properties and mechanisms. Moreover, the ability of essential oils to demonstrate anticancer effects through action on various pathways and cellular mechanisms has aroused interest in searching for possible phytopharmaceuticals against cancer. Thus, more studies are necessary to expand the present knowledge of these mechanisms to promote cell-specific and individualized cancer therapy. 15
Due to the great phytotherapeutic use of Eugenia, Psidium, and Myrcia species and the use of their essential oils in traditional medicine, this study is aimed to analyze the chemical composition and evaluate the antioxidant and cytotoxic activities of essential oils of some Myrtaceae species from the Brazilian Amazon.
Results and Discussion
Yield and Chemical Composition of Essential Oil
The descending order of leaf essential oil yields of Myrtaceae samples are Myrcia sylvatica (G. Mey.) DC. (0.7%), Psidium guineense (Pgui-1: 0.5%; Pgui-2: 0.6%), Eugenia patrisii (0.4%), Psidium guajava L. (0.4%), Eugenia stipitata (0.2%), and Myrcia splendens (0.1%). In all samples, 173 compounds were identified by GC-FID and GC-MS, mainly terpenoids representing an average of 94.18% of total composition in oils (Table 1). Eugenia and Myrcia species showed a predominance of sesquiterpene hydrocarbons: E. patrisii (85.7%), M. splendens (76.5%), M. sylvatica (71.5%), and E. stipitata (61.7%). Sesquiterpene hydrocarbons (43.6%) and oxygenated sesquiterpenoids (44.7%) were the major terpenoid classes in P. guajava oil. Meanwhile, the two P. guineense oil samples showed monoterpene hydrocarbons as predominant (Pgui-1: 58.1%; Pgui-2: 54.3%).
Leaf Essential Oil Yield (%) and Constituents (%) of Myrtaceae Species.
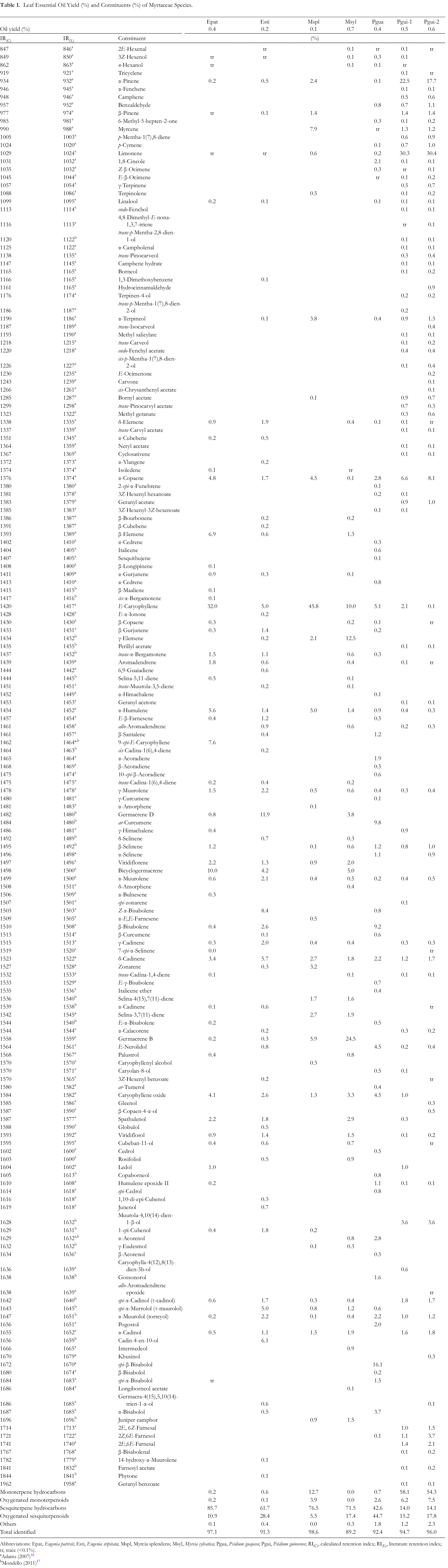
Abbreviations: Epat, Eugenia patrisii; Esti, Eugenia stipitata; Mspl, Myrcia splendens; Msyl, Myrcia sylvatica; Pgua, Psidium guajava; Pgui, Psidium guineense; RI(C), calculated retention index; RI(L), literature retention index; tr, trace (<0.1%).
aAdams (2007) 16
bMondello (2011) 17
Among Eugenia species, E. patrisii presented E-caryophyllene (32.0%) and bicyclogermacrene (10.0%) as major compounds, whereas E. stipitata showed germacrene D (11.8%) and Z-α-bisabolene (8.38%). It is known that E. patrisii leaves oils were characterized by the presence of cyclic sesquiterpenes with cadinane and caryophyllane skeletons, including trans-cadin-1,4-diene (16.5%), trans-muurola-3,5-diene (13.3%), and E-caryophyllene (11.1%). 18 However, E. patrisii aerial parts (leaves and fine stems) had a mixture of acyclic and cyclic oxygenated sesquiterpenes as 2E,6E-farnesol (34.5%), 2E,6Z-farnesol (23.2%) and caryophylla-4(12), 8(13)-dien-5β-ol (15.6%) as major constituents. 6,19
Eugenia stipitata leaves essential oil collected in Portugal was characterized by E-caryophyllene (22.7%) and caryophyllene oxide (15.4%). 20 Likewise, the EO of the leaves from the Brazilian Northeast was characterized by guaiol (13.7%) and E-caryophyllene (11.3%). 21 The variability in the chemical compositions of Eugenia spp. may be related to the environmental differences of the collection sites since this is a factor that can influence chemotypes varieties. 6,19
The oils of Myrcia samples also displayed sesquiterpenoids as main constituents: M. splendens presented E-caryophyllene (45.8%), M. sylvatica showed an abundance of germacrene B (24.5%) and γ-elemene (12.5%). M. splendens EOs extracted from its leaves exhibited different chemical profiles. 22 The EO from M. splendens collected in Amazonian Ecuador was characterized by E-nerolidol (67.8%) and α-bisabolol (17.5%). 22 Similarity, α-bisabolol (83.8%) and α-bisabolene (80%) were identified as primary compound in 2 samples collected in the southern and southeastern regions of Brazil, respectively. 23,24 On the other hand, its EO from the aerial parts (leaves and stems) collected in three regions of Brazilian amazon (I-III) showed different chemical profile characterized by selin-11-en-4α-ol (24.7%), caryophyllene oxide (16.6%), and spathulenol (13.8%) (sample I); cis-calamenene (30.1%), spathulenol (18.7%) and α-calacorene (11.5%) (sample II); and spathulenol (40.2%) and β-bisabolene (14.7%) (sample III). 25 M. sylvatica EOs presented a chemical composition influenced by seasonal factors. 9 The monthly monitoring of EO composition during 1 year of a specimen from northern Brazil showed variability in its major constituents β-selinene (6.2 to 10.5%) and 1-epi-cubenol (5.9 to 9.8%). 9
Psidium guajava (Pgua) showed the sesquiterpenes epi-β-bisabolol (16.1%), ar-curcumene (9.8%), β-bisabolene (9.2%), E-caryophyllene (5.1%), and caryophyllene oxide (4.5%) as the main compounds. While P. guineense samples (Pgui-1 and Pgui-2) showed as the main compound the monoterpenes limonene (Pgui-1: 30.2% and Pgui-2 30.4%) and α-pinene (Pgui-1: 22.5% and Pgui-2: 17.7%).
The chemical composition of 22 genotypes of P. guajava from Brazil were evaluated and showed an abundance of E-caryophyllene (5.1% to 32.3%), caryophyllene oxide (1.8% to 20.9%), α-humulene (1.7% to 19.9%), β-bisabolol (2.2% to 19.4%), E-nerolidol (2.1% to 13.7%), β-selinene (0.5% to 12.8%), hinesol (3.2% to 12.4%) epi-α-cadinol (1.1% to 12.0%), α-selinene (0.5% to 11.2%), and limonene (0.1% to 11.0%). These quantitative changes were attributed to environmental factors. 26 Leaves of P. guineense EO from the Brazilian Midwest showed high amounts of spathulenol (80.7%). 11 However, P. guineense from Brazilian Amazon displayed β-bisabolol (17.4%), limonene (6.8%), and epi-α-bisabolol (6.7%) as main constituents. 27
Antioxidant Activity
DPPH method is based on reducing the alcoholic DPPH solution in the presence of hydrogen donating antioxidants through the formation of the non-radical form (DPPH-H). As the EO reduces the DPPH radical, the solution changes color from purple to yellow (arising of diphenylpicrylhydrazine). The reduction capability of the DPPH radical is determined by the decrease in its absorbance at 517 nm. 28 All Myrtaceae samples showed antioxidant activity against the DPPH method as shown in Table 2.
Antioxidant Capacity of the Oils of Myrtaceae Species.
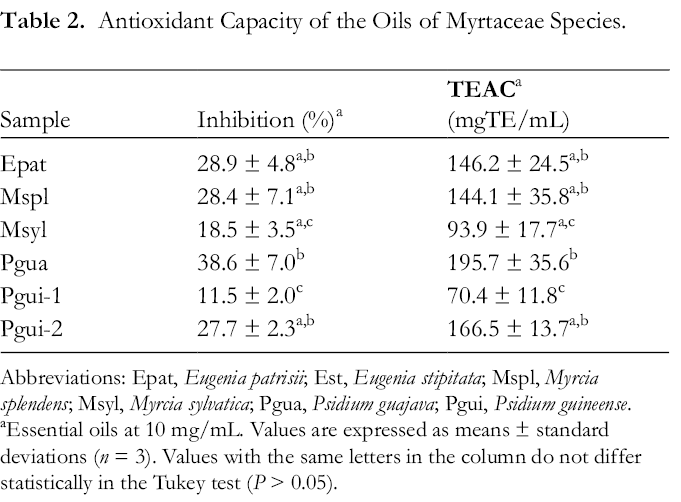
Abbreviations: Epat, Eugenia patrisii; Est, Eugenia stipitata; Mspl, Myrcia splendens; Msyl, Myrcia sylvatica; Pgua, Psidium guajava; Pgui, Psidium guineense.
aEssential oils at 10 mg/mL. Values are expressed as means ± standard deviations (n = 3). Values with the same letters in the column do not differ statistically in the Tukey test (P > 0.05).
Psidium guajava essential oil (Pgua) showed the highest antioxidant activity, inhibiting 38.4% of the DPPH radical, this oil was rich in the sesquiterpenes epi-β-bisabolol (16.1%) and ar-curcumene (9.8%). Total antioxidant capacity was expressed as Trolox equivalent (TE), and Pgua sample showed a value of 195.7 mgTE/mL, only five times smaller than the Trolox Standard. On the other hand, the least active sample was Pgui-1, rich in limonene (30.2%) and α-pinene (22.5%), which presented an inhibition rate of 11.5% of DPPH radical (70.4 mgTE/mL). However, the Pgui-2 samples were also rich in limonene (30.4%) and α-pinene (17.7%) and presented inhibition of 27.7% (166.49 TE/mL), 6 times less active against DPPH compared to Trolox. The sample Pgua presents the highest content of oxygenated sesquiterpenes (44.7%), which can be attributed to its significant antioxidant activity, as this compound’s class generally exert good free radical scavenging activities. 29
Psidium guajava oil from Malaysia displayed DPPH radical scavenging with an IC50 value of 460.4 µg/mL and a high phenolic compound (495.9 mgGAE/g), activity related to the concentration of phenols that the species presents. 28 Psidium guineense rich in spathulenol (80.7%), presented inhibition of DPPH radical with IC50 of 63.08 µg/mL, being lass active of 2,6-di-tert-butyl-4-methylphenol standard (BHT, IC50 16.7 µg/mL). 11
The E. patrisii essential oil (Epat), rich in E-caryophyllene (32.0%) and bicyclogermacrene (10.0%), presented 28.9% of DPPH radical scavenging (146.2 mgTE/mL), about six times less activity than Trolox standard. Another E. patrisii sample from Brazilian Amazon, rich in 2E,6E-farnesol (34.5%), showed DPPH inhibition of 40.9%, about 10 times less active than Trolox standard (111.2 mgTE/mL). 19
Myrcia splendens essential oil (Mspl), rich in E-caryophyllene (45.8%) presented DPPH radical scavenging of 28.4%, about seven times less than Trolox (144.1 mgTE/mL). On the other hand, M. sylvatica oil sample (Msyl), rich in the sesquiterpene hydrocarbons germacrene B (24.5%),
Oils from Myrcia species have been reported as antioxidants. For example, M. splendens demonstrated antioxidant activity with an IC50 of 43.5 µg/mL, rich in the E-nerolidol sesquiterpene (67.81%). 13 Moreover, EO of Myrcia oblongata DC., rich in caryophyllene oxide (22.0%) and trans-verbenol (11.9%), was identified as an antioxidant (IC50 2.8 µg/mL) compared to the synthetic antioxidant BHT (IC50 1.4 µg/mL). 30
Cytotoxic Activity
The cytotoxic activity of the seven Myrtaceae essential oil samples were evaluated by the MTT method and showed antiproliferative activity against the 4 hoursuman cancer cell lines, breast (MCF7), melanoma (SKMEL-19), gastric (AGP01), colon (HCT116), and a non-malignant human lung fibroblast cell (MRC5), see Table 3. It has been suggested IC50 values below 100 µg/mL show relevant activity. 31
Cytotoxic Activity of the Myrtaceae Species Essential Oils on Cell Lines.
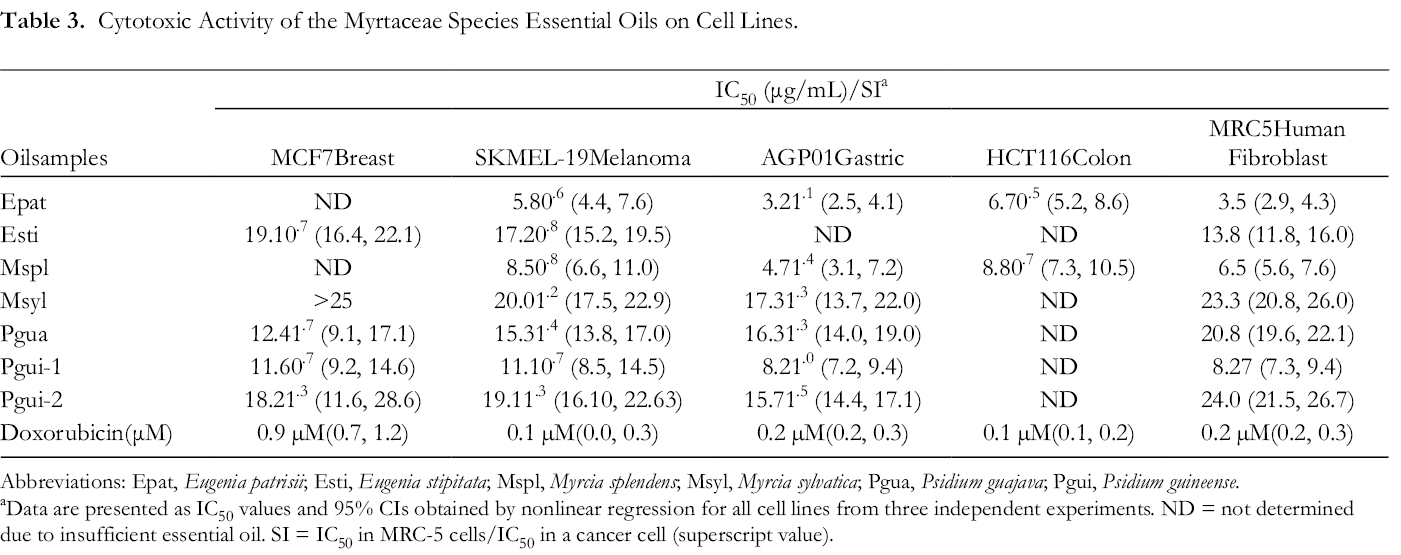
Abbreviations: Epat, Eugenia patrisii; Esti, Eugenia stipitata; Mspl, Myrcia splendens; Msyl, Myrcia sylvatica; Pgua, Psidium guajava; Pgui, Psidium guineense.
aData are presented as IC50 values and 95% CIs obtained by nonlinear regression for all cell lines from three independent experiments. ND = not determined due to insufficient essential oil. SI = IC50 in MRC-5 cells/IC50 in a cancer cell (superscript value).
Among the samples tested, the oil of E. patrisii (Epat) showed the highest activity against melanoma (SKMEL-19) and colon (HCT116) cells with IC50 values of 5.8 and 6.7 µg/mL, respectively. Likewise, gastric ascites (AGP-01) was the most sensitive human cancer cell line against Epat oil and showing an IC50 value of 3.2 µg/mL and SI of 1.1. The SI value reveals the differential activity; therefore, the higher the SI value is, the more selective it is. On the other hand, an SI value <2 suggests general toxicity to compounds. 32 In contrast to our findings in this study, E. patrisii EO from Brazilian Amazon, rich in (2E,6Z)-farnesol (23.2%) and (2E,6E)-farnesol (34.5%), sesquiterpene alcohols that have shown anti-neoplastic effects in various human cancers, 33 did not show activity against gastric ascites cancer cells. 19
Eugenia stipitata essential oil (Esti) displayed cytotoxic activity against melanoma (IC50 17.2 µg/mL), and breast (IC50 19.1 µg/mL) human cancer cells; and the normal human fibroblast (IC50 13.8 µg/mL). As far as we are aware, there have been no reports of cytotoxic activity of essential oils of E. stipitata reported in the literature.
Myrcia splendens (Mspl) showed good cytotoxic activity against gastric (IC50 4.7 µg/mL), melanoma (IC50 8.6 µg/mL), and colon (IC50 8.8 µg/mL) human cancer cells. However, Mspl oil was more selective against AGP01 (SI 1.4). Myrcia sylvatica oil (Msyl) did not show activity against breast cells, but displayed activity against melanoma (IC50 20.0 µg/mL) and gastric (IC50 17.3 µg/mL) cancer cells. Previous studies have reported the cytotoxic activity of M. splendens EO. A sample rich in E-nerolidol (67.8%), α-bisabolol (17.5%), and E-caryophyllene (4.2%) displayed cytotoxic potential against breast (MCF7, IC50 5.6 µg/mL) and normal human keratinocytes (HaCaT, IC50 21.6 µg/mL). 13 However, there have been no reports about the cytotoxic activity of M. sylvatica EO.
Psidium guajava (Pgua) showed activity against melanoma (IC50 15.3 µg/mL), gastric (IC50 16.3 µg/mL), breast (IC50 12.4 µg/mL) human cancer cells with a SI of 1.7 against MCF7 cells. Likewise, P. guineense samples (Pgui-1 and Pgui-2) displayed activity against breast (IC50 12.6 and 18.2 µg/mL), melanoma (IC50 11.1 and 19.1 µg/mL), gastric (IC50 8.2 and 15.7 µg/mL), and a normal human fibroblast (IC50 8.27 and 24.0 µg/mL). Pgui-1 sample was more active than Pgui-2 against all cell lines. Pgui-1 and Pgui-2 sample have similar chemical compositions of their essential oils, except for the concentration of E-caryophyllene (Pgui-1 2.1%; Pgui-2 0.1%).
Literature data have already demonstrated the feasibility of applying P. guajava OE in the pharmacological sector due to its variability of organic compounds, as well as its efficiency in preliminary tests of anti-inflammatory activity, indicating possible viability in combating gastric ulcers. 26,34 In addition, significant cytotoxic activity was reported by the P. guineense essential oil against ovarian cancer cells (OVCAR-3) with GI50 of 0.9 µg/m. 35
Moreover, literature data indicate that E-caryophyllene, a compound detected in all samples of Myrtaceae essential oils in this study, has a potent anticancer activity, affecting the growth and proliferation of different types of cancer cells. 36 For example, E-caryophyllene showed cytotoxic active against MCF7 cells (LC50 190 µM). 37 In addition, this compound has shown synergistic enhancement with other essential oil components. 38 Thus, a synergistic effect between E-caryophyllene with various substances found in these essential oils likely contribute to the cytotoxic activities and may explain the different activities of Pgui-1 compared to Pgui-2.
Conclusions
The essential oil of Myrtaceae samples showed significant antioxidant activities in comparison to Trolox, and notable cytotoxic activity against the lung, colon, stomach, and melanoma cells, with a real prospect to their subsequent phytotherapeutic development. However, due to the presence of different chemical profiles requires their standardization to consider development of new anticancer drugs.
Materials and Methods
Plant Material
The leaves of seven Myrtaceae specimens were collected in Pará state (PA), Brazil, during the dry season (August-December). The collection site, voucher number, geographic coordinates for these specimens are listed in Table 4. After identification, plant vouchers were deposited in the Herbaria of Museu Paraense Emílio Goeldi (MG) and Embrapa Amazônia Oriental (IAN) at the city of Belém (PA), Brazil. The leaves were dried for 2 days at room temperature, ground and then submitted to essential oil extraction.
Collection Site, Voucher Number, and Coordinates for the Myrtaceae Specimens.

Abbreviation: NR, not registered.
aThe botanical identification was made by comparison with authentic samples.
Extraction and Chemical Composition Analysis of the Essential Oil
The plant material was air-dried (2 days) at room temperature after collection. Then, it was ground and submitted to hydrodistillation using a Clevenger-type apparatus (3 hours). 39 The oils obtained were dried over anhydrous sodium sulfate and total oil yields were expressed as mL/100 g of the dried material. 40 The chemical composition analysis was performed by GC-MS, using a Shimadzu instrument Model QP 2010 ultra (Shimadzu, Tokyo, Japan), equipped with a Rtx-5MS (30 m × 0.25 mm; 0.25 µm film thickness) fused silica capillary column (Restek, Bellefonte, USA). Helium was used as carrier gas adjusted to 1.0 mL/min at 57.5 KPa; split injection (split ratio 1:20) of 1 µL of hexane solution (oil 5 µL : hexane 500 µL); injector and interface temperature were 250 °C; oven temperature programmed was 60 to 240 °C (3 °C/min), followed by an isotherm of 10 minutes. EIMS (Electron Impact Mass Spectrometry): electron energy, 70 eV; ion source temperature was 200 °C. The mass spectra were obtained by automatic scanning every 0.3 s, with mass fragments in the range of 35‐400 m/z. The compounds present in the samples were identified by comparison of their mass spectrum and retention index calculated for all volatile components using a linear equation of Van Den Dool and Kratz 41 with data present in commercial libraries of FFNSC 2 17 and Adams. 16 Retention indices were calculated using n-alkane standard solutions (C8–C40, Sigma-Aldrich, St. Louis, MO, USA), in the same chromatographic conditions. The GC-FID analysis was carried out on a Shimadzu QP-2010 instrument, equipped with FID detector, in the same conditions, except that hydrogen was used as the carrier gas. The percentage composition of the oil samples was computed from the GC-FID peak areas.
DPPH Radical Scavenging Assay
A stock solution of DPPH radical (0.5 mM) in ethanol was prepared. After dilution in ethanol (60 µM approx.), the solution measured an initial absorbance of 0.62 ± 0.02 in 517 nm at room temperature. The reaction mixture was composed by 1000 µL of DPPH solution, 5 µL of the oil sample, 900 µL of Tris-HCl buffer (100 mM, pH 7.4), 45 µL of ethanol, and 50 µL of Tween 20 solution (0.5%, w/w). For each sample, an ethanol blank was also measured. The absorbance was measured at the beginning of the reaction (time zero), each 5 minutes during the first 20 minutes and then at continuous intervals of 30 minutes up to constant absorbance.
Standard curves were prepared using Trolox (6-hydroxy-2,5,7,8-tetramethylchroman-2-carboxylic acid) (1.0–10.0 mM). The DPPH inhibition percentage calculated the radical scavenging activity of each sample according to the Equation, Inhibition = 100[(A-B)/A], where A and B are the blank and sample absorbance values in the end reaction. The total antioxidant activity was expressed as milligrams of Trolox, calculated utilizing the Equation, TE(mg/)g = [(A-B)/(A-C)]*[25/1000]*[250.29/1000]*[1000/10]*D, where A, B, and C are the blank, sample, and Trolox absorbance values in the end reaction, and D is the dilution factor. All experiments were triplicated. 42
Cell Viability Assay (MTT Assay)
The cell viability was determined by reduction of the yellow dye 3-(4,5-dimethyl-2-thiazol)−2,5-diphenyl-2H-tetrazolium bromide (MTT) to a blue formazan product as previously described by Mosmann (1983). 43 Oils (0.4, 25 µg/mL) were tested for cytotoxic activity against the cancer cell lines HCT116 (colon), AGP01 (malignant gastric ascites), MCF7 (breast), and SKMEL-19 (melanoma), as well as, against the normal non-malignant human lung fibroblast (MRC-5) cells. Cell lines were provided by Núcleo de Pesquisa e Desenvolvimento de Medicamentos (Drugs Research and Development Center) of Universidade Federal do Ceará. All cell lines were maintained in DMEM (Dulbecco’s Modified Eagle Medium) supplemented with 10% fetal bovine serum, 2 mM glutamine, 100 U/mL penicillin, 100 µg/mL streptomycin, at 37 °C and with 5% CO2. The DMSO final concentration in the culture medium was kept constant (<0.1%). The oils were incubated with the cells for 72 hours, and the negative control received the same amount of DMSO (0.001% in the highest concentration). Doxorubicin was used as positive control. 44,45 SI (selectivity index) values were measured using the ratio between IC50 of the essential oil against MRC-5 and IC50 of the same oil essential against each cancer cell line.
Statistical Analyses
The antioxidant experiments were performed in triplicate and their data analyzed are expressed as mean ± SD. Statistical differences were evaluated by Tukey’s test (P < 0.05) using the software GraphPad Prism 6.0. The IC50’s values of MTT assay were calculated by nonlinear regression using GraphPad 6.0 software (Intuitive Software for Science, San Diego, CA). 42
Footnotes
Acknowledgments
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
