Abstract
The development of new antimicrobials has always been a research hotspot. In this study, β-pinene-based derivatives were synthesized, and their antimicrobial activity was evaluated. The purpose was to develop some novel, promising new fungicides. Three β-pinene derivatives containing bis-hydronopyl were prepared, and their antifungal and antibacterial activities were evaluated against 6 plant pathogenic fungi and 4 bacterial species; a preliminary structure-activity relationship is discussed. The results indicated that the derivatives containing the blend of alkyl group and bis-hydronopyl had potent inhibitory activities against plant fungal pathogens and bacteria. Among these molecules, bis-hydronopyl dimethyl ammonium bromide showed excellent effects on
Plant pathogenic fungi have become a serious threat to global crop production and food security, which has resulted in huge losses to human beings. 1 -3 In addition, pathogenic bacteria are the cause of many human diseases. 4 In the past few decades, the large-scale use of chemical fungicides and antibiotics has been the principal tool to eliminate these disasters. 5 However, the widespread application of chemical fungicides and antibiotics often leads to drug resistance of fungi and bacteria. Drug-resistant bacterial and fungal strains are causing a serious threat to global human health, and so it is very urgent to look for new alternatives. 6,7 Therefore, the development of safe and eco-friendly alternatives can not only control the increase of microorganisms but also reduce the environmental risks. 8
β-Pinene is a natural compound with antibacterial activity, which can be used to participate in many chemical reactions.
9
-13
A large number of β-pinene derivatives can be synthesized by chemical modification, and some of these derivatives have been proved to have increased antibacterial activities.
14
-19
For example, Gavrilov et al
20
synthesized 3 series of β-pinene-based derivatives, and the reaction mixes of the sulfoxides had high activity against
Herein, a series of quaternary ammonium salts were synthesized from β-pinene in order to obtain derivatives with potent antimicrobial activity. This study can be used as a guide for the development of new and efficient botanical antimicrobial agents.
Materials and Methods
Synthetic Procedures and Structural Characterization of the Title Compounds
Materials and structural characterization techniques
Pure chemicals were purchased from Jiangxi Jingke Scientific Instrument Co., Ltd., China. Fourier transform infrared (FT-IR) spectroscopy was performed using a Nicolet IS10 FT-IR spectrometer (America). Melting points (m.p.) were determined with a WRS-2 melting point apparatus (Shanghai Precision & Scientific Instrument Co., Ltd., Shanghai, China) and are uncorrected. Proton nuclear magnetic resonance (NMR) was performed on a Bruker 400 spectrometer with tetramethylsilane and deuterated chloroform (CDCl3) used as internal control and solvent, respectively. Mass spectrometry (MS) was carried out on a Bruker mass SL spectrometer (Bruker, Germany).
The synthesis of quaternary ammonium salts
(1
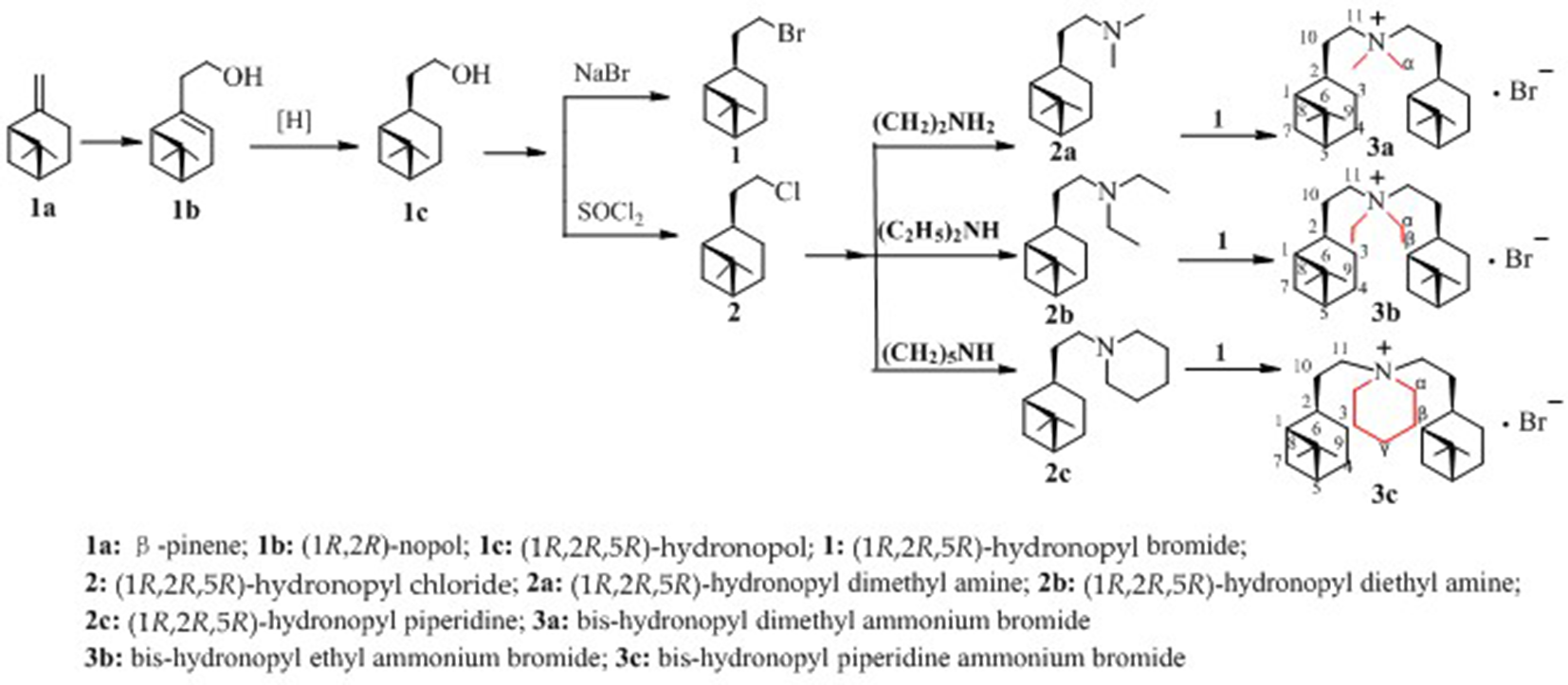
Synthetic routes of bis-hydronopyl quaternary ammonium salts (3a-c).
(1
(1
(1
Antimicrobial Tests
Fungal pathogens and bacteria were obtained from East China Woody Fragrance and Flavor Engineering Research Center of National Forestry and Grassland Administration. The microorganisms included 6 typical phytopathogenic fungi (
Determination of mycelial growth inhibition of fungal pathogens
The antifungal activities of the samples were tested using the mycelial growth inhibition (MGI). The compounds were added to sterilized water to generate respective stock solutions (2.0 × 104 mg/L), which were further diluted to 200, 100, 50, 25, and 12.5 µg/mL with potato dextrose agar, respectively. Then, a 5-mm disc containing mycelia was transferred to the center of the medicated media and incubated at 28 °C. 29,30 Each experiment was conducted in triplicate. The diameter of the fungal colony was measured using a cross method and averaged. Sterilized water was used as the no-treatment control and chlorothalonil (98%) as the positive control. MGI was calculated using Eq. (1).

where

Determination of effective concentration
The antimicrobial activities of
Determination of minimum inhibitory concentration of bacteria
The antimicrobial properties of
Results and Discussion
Chemistry
The FT-IR spectrum of
Biological Activity
Activity against phytopathogenic fungi
The antifungal activities of the β-pinene-based derivatives against six plant pathogens are shown in Tables 1
-4. For all the tested samples, the inhibitory rates improved with the increase in sample concentration. The EC50 values of the β-pinene-based derivatives against the 6 typical phytopathogenic fungi were less than 30 µg/mL. Compound
Antifungal Activities of 3a Against Plant Pathogenic Fungi.

Abbreviations: EC50, half-maximal effective concentration; MGI, mycelial growth inhibition.
Antifungal Activities of 3b Against Plant Pathogenic Fungi.
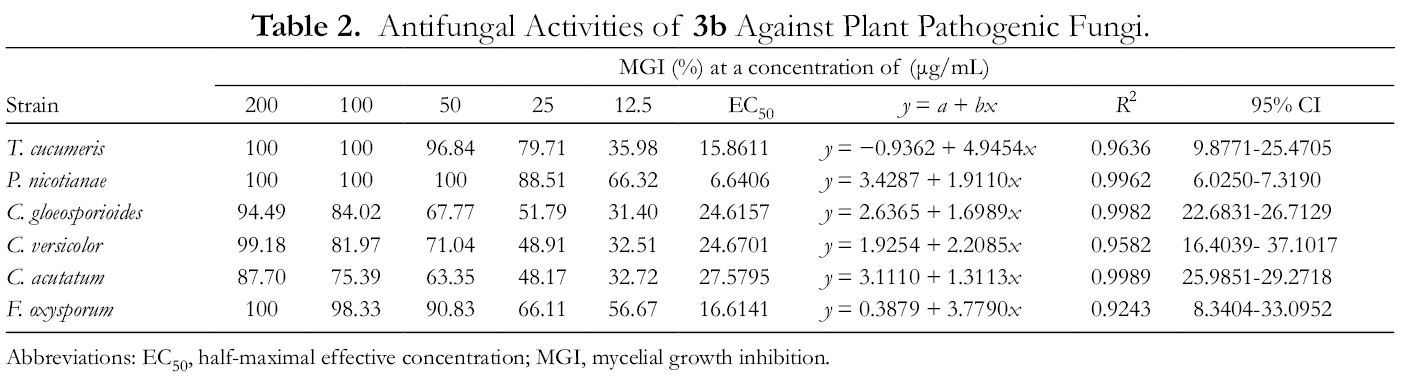
Abbreviations: EC50, half-maximal effective concentration; MGI, mycelial growth inhibition.
Antifungal Activities of
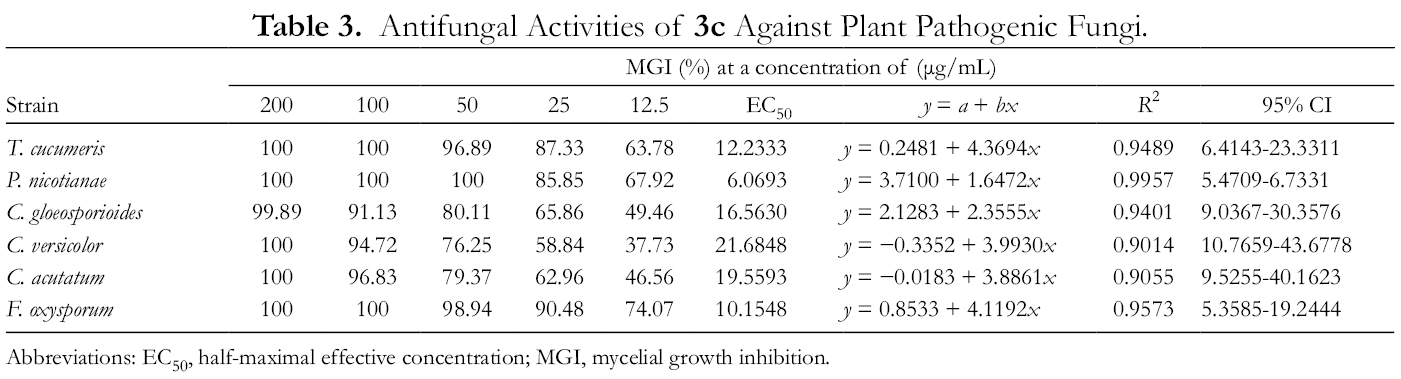
Abbreviations: EC50, half-maximal effective concentration; MGI, mycelial growth inhibition.
Antifungal Activities of Chlorothalonil Against Plant Pathogenic Fungi.

Abbreviations: EC50, half-maximal effective concentration; MGI, mycelial growth inhibition.
Of all the compounds tested,

Effect of bis-hydronopyl dimethyl ammonium bromide (3a) on mycelial morphology in
Although it was hard to give a comprehensive structure-activity relationship to these derivatives, we could find some interesting hints from the antifungal experiment. For
Antibacterial activity
Screening of the activities of bis-hydronopyl quaternary ammonium salts against bacteria was performed using Gram-negative (

MIC values of bis-hydronopyl quaternary ammonium salts against
Compared with the positive control kanamycin sulfate,
According to the recent literature, the introduction of piperidine can make the compounds have antioxidant, bacteriostatic, and antimalarial activities.
43
-45
Subba Poojari et al found that some novel piperidine derivatives had obvious inhibitory activity against
Conclusions
Derivatives of β-pinene (
Supplemental Material
Supplementary Material 1 - Supplemental material for β-Pinene Derived Products With Enhanced In Vitro Antimicrobial Activity
Supplemental material, Supplementary Material 1, for β-Pinene Derived Products With Enhanced In Vitro Antimicrobial Activity by Xuezhen Feng, Zhuanquan Xiao, Yuling Yang, Shangxing Chen, Shengliang Liao, Hai Luo, Lu He, Zongde Wang and Guorong Fan in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship,and/or publication of this article: This work was supported by the Key Projects of Key R&D Program of Jiangxi Province (20192ACB60011).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
