Abstract
Anxiety is a state that becomesa disorder when a person experiences disproportionate levels of anxiety on a regular basis. This disproportion is also accompanied by excessive nervousness and fear. This study aimed to determine the protective effect of prunin using different anxiety models. Three preclinical anxiety models, elevated plus maze, light/dark, and social interaction, were employed in the study. Albino mice were selected and treated with pruninand other drugs for 7 days to determine their anti-anxiety effect. Thereafter, their behavior was examined using the plus maze, light-dark chamber, and other stimulatory parameters, such asimmobility, sniffing, and crawling during experimentation. Two doses (50 mg/kg and 100 mg/kg, p.o.) of prunin were administered to two separate mice groups. Further, fluoxetine (10 mg/kg; p.o.) was administered to one of the mice groups for 7 days. Thereafter, the levels of neurotransmitters, such as serotonin and GABA, in brain samples were determined. Based on the results, prunin significantly improved the behavior and mobility of animals in all three anxiety models. Further, prunin modulated the release of serotonin and GABA, demonstrating the mechanistic approach it employs to interact with cellular receptors to mimic neurotransmission. The mRNA expression levels of tph2 (5-HT synthesizing enzyme) and slc6a4 (5-HT transporter) were also found to be downregulated in both prunin- and fluoxetine-treated mice brains. Collectively, our findings suggest that prunin could be administered to treat anxiety in mice. However, further studies should be carried out to explore its potential for clinical application.
Anxiety is an unpredictable dynamic process that changes the physiological functioning of the brain to an abnormal state. This state, if left untreated, may lead to other psychological disorders. 1 The drugs used/prescribed for anxiety-like disorders, benzodiazepines or selective neuromodulators, provide either symptomatic relief or increasecalmness in patients; however, patients treated with these drugs may experience withdrawal effects if the drugs are used long term. 2 Traditional sources, such as most herbs (mainly aromatic plantsources), are the best treatments for anxiety; however, they are less preferred relative to synthetic compounds becauseoftheir purity and standardization. 3 Many plant constituents, either in a natural/semi-synthetic/synthetic form, have been traditionally used and are claimed to exert therapeutic effects against anxiety; however, biological screening/drug discovery,as well as pre-clinical findings are required to establish their action to provide scope for further discovery. 3 Prunin is a flavone glycoside found in many pigmented fruit plants, including citrus fruits and tomatoes. The aglycone ofpruninis called naringenin. The enzyme, alpha-L-rhamnosidase, breaks naringin into prunin and rhamnose, and glucosidase breaks prunin into glucose and naringenin. Prunin is reported to exert different pharmacological activities, includingantioxidant, 4,5 anti-inflammatory, 6 deoxyribonucleic acid-binding affinity, 7 -9 antiviral, 10 hypocholesterolemic, 11 cardio protective, 12 and anti-atherosclerotic effects. 4 -6,13 In fact, a prior study revealed that prunin may affect the release of monoamines in the brain and is effective for anxiety or neurological difficulties. 4 As a result, pruninhas been selected for use in studies to demonstrate its efficacy for the treatment of anxiety. Three well-known anxiety models were employed in the present study to evaluate the anti-anxiety effect of prunin. Furthermore, the pharmacological use of this compound and the molecular mechanism it employs to ameliorate anxiety were explored.
Results
Acute Toxicity
Based on the results of acute toxicitystudies, prunin did not cause mortality and toxicity inmice. As a result,doses of 50 and and 100 mg/kg, p.o.,were selected for use in this study.
Elevated Plus Maze (EPM) Model
Effects of pruning on the time spent by animals in each arm and on latency in the EPM model
Fluoxetine-treated mice spent more time in the open arm than the closed arm (P < .01). Similarly, mice treated with prunin showed a dose-dependent improvement in the time spent in the open arm (157.6 ± 2.55 s) compared withcontrol mice (56.6 ± 2.45 s). Prunin-treated mice tended to spend less time in the closed arm (94.6 ± 2.5 s) relative to control mice (140.1 ± 5.38 s), ultimately indicating a reduction in their anxiety.
A decrease in the duration of latency (49.4 ± 2.10 s) was observed in fluoxetine-treated mice compared to control mice (93.1 ± 1.23 s). The 50 mg/kg prunin dose caused an insignificant decrease in latency on the first day (66.2 ± 1.32 s); however, a significant decrease (P < .05) was observed on days 3 and 7. The higher prunin dose (100 mg/kg) caused a significant decrease in latency (48.30 ± 1.60 s.) (P < .01) compared to control treatment, ultimately resulting in a decrease in anxiety (Figures 1 -3).

Effectof prunin (PR) on the time spent by animals in the closed arm in the elevated plus maze model.

Effectof prunin (PR) on the time spent by animals in the open arm in the elevated plus maze model.
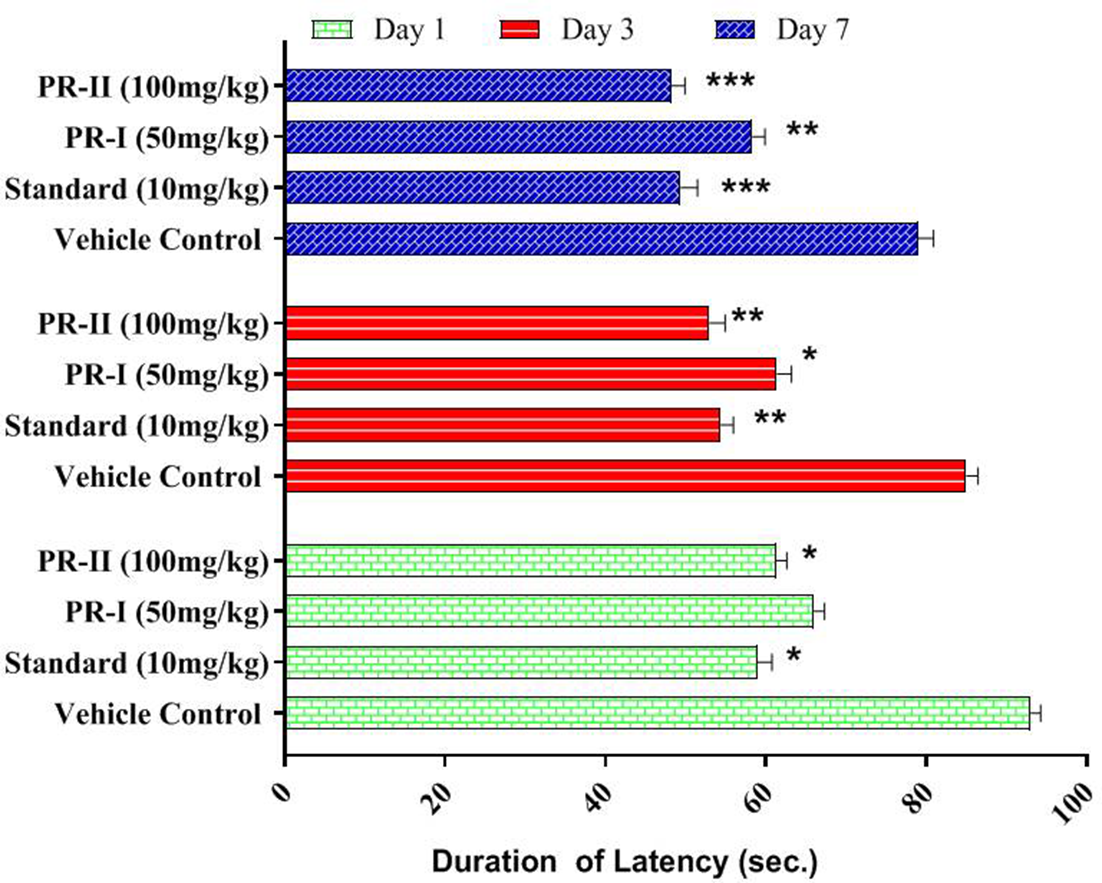
Effect of prunin (PR) on the duration of latency exhibited by animals in the elevated plus maze model.
Compared to the control group (93.1 ± 1.23), mice treated with 50 mg/kg and 100 mg/kg of prunin began to display a decrease in latency on different days of the experiment. The effect of the latter was exerted on the first day (48.30 ± 1.60), while that of the former was observed after the third day.
Effect ofpruninon the numberof entries made by animals into each arm in the EPM model
Fluoxetine-treated mice showed an increase in the number of entries made into the open arm (5.70 ± 0.81) and less preference with entries counted in the closed arm (3.11 ± 0.32) compared to control mice (open arm—1.42 ± 0.34, closed arm—6.80 ± 0.33). Further, fluoxetine-treated mice made frequent entries into the open arm and were restricted to the closed arm. Initially, prunin-treated mice made fewer entries into the open arm (3.11 ± 0.32) at the low dose (50 mg/kg); however, on days 1, 3, and 7 of the trial, the number of entries made by mice treated with the higher dose (5.97 ± 0.40) increased relative to that made by control mice (Figures 4 and 5).

Effect of prunin (PR) on the number of entries made by animals into the open arm in the elevated plus maze model.

Effect of prunin (PR) on the number of entries made by animals into the closed arm in the elevated plus maze model.
Effect of Pruninon Mice in theLight/Dark Model
Effect of pruninon miceimmobility
Fluoxetine-treated mice spent more time (275.2 ± 3.45 s) in the light compartment than control mice (232.2 ± 1.01 s). Treatment with 50 and 100 mg/kg prunin caused a gradual increase in the time spent by mice in the light compartment (248.1 ± 1.50, 268.1 ± 2.13 s) compared to control treatment (Figure 6). Moreover, mice treated with the high dose of prunin (100 mg/g, p. o.) preferred to enter and spend less time in the dark chamber on day 7 (280.2 ± 3.08 s) compared to control mice (309.7 ± 4.60 s) (Figure 7). The duration of immobility for control mice was 71.1 ± 1.9 s; however, immobility was found to be further reduced in fluoxetine-treated mice (41.0 ± 1.39 s). Prunin caused less immobility in mice administered 100 mg/kg, p.o. (52.1 ± 2.2 s); this was significant (P < .01) compared to that observed in control mice (Figure 8).

Effectof prunin (PR) on the time spent by animals in the light compartment in the light/dark model.

Effect of prunin (PR) on the time spent by animals in the dark compartment in the light/dark model.

Effect of prunin (PR) on the duration of immobility exhibited by animals in the light/dark model.
Effect of prunin on the number of crossings
Fluoxetine-treated mice performed more crossings (19.6 ± 0.9) (P < .1) than mice in the control group (7.2 ± 1.1). However, prunin (100 mg/g, p.o.) was found to cause a change in the number of crossings on days 1, 3, and 7. In addition, prunin treatment significantly (P < .01) increased the number of crossings compared with the control group (Figure 9).

Effect of prunin (PR) on the number of crossings performed by animals in the light/dark model.
Effect of pruninon the social interaction behavior exhibited by mice
Based on our findings, there were changes in the social interaction behavior of mice. First, fluoxetine-treated mice participated in more social interaction than control mice. However, mice treated with 100 mg/kg of prunin showed a significant increase in their “sniffing and crawling and climbing” abilities. Such findings indicate that they participated in more social interaction than control mice (Table 1).
Effect of Pruning on the Social Interaction Behavior Exhibited by Mice.

no. of animals (n) = 6, *P < .05, **P < .01, ***P < .001 (one-way ANOVA followed by Dunnett’s ‘t’ test). Values are expressed as mean ± SEM.
Effectof prunin on the levels of serotonin and GABA in the brain tissue
Compared to control mice, the level of serotonin was altered in the brain tissue of mice treated with fluoxetine. However, a higher level of serotonin was found in the brain of mice treated with prunin; the level of serotonin was highest when 100 mg/kg was administered, which demonstrates that prunin exhibits a dose-dependent effect.
The level of GABA was decreased in control mice and slightly improved in those treated with fluoxetine. However, the mice treated with prunin had a significantly higher level of GABA than the controls (Table 2).
Effect of Prunin on the Level of Serotonin in the Brain of Mice.

no. of animals (n) = 6, *P < .05, **P < .01, ***P < .001 (one-way ANOVA followed by Dunnett’s ‘t’ test). Values are expressed as mean ± SEM.
Effect of pruninon 5HT expression
The concentration of cerebral 5-HT and the gene expression of tph2 and slc6a4 were decreased with the appearance of anxiety-like behaviors in control mice. However, their levels were significantly elevated by treatment with prunin.(Figure 10)

mRNA expression levels of slc6a4 and TPH2 in the dorsal raphe nucleus. (
Discussion
The current study revealed the potential anti-anxiety effectof pruninin mice. In fact, the release of serotonin and GABA was found to be modulated in the brain tissue of mice treated with prunin, ultimately confirming its effectiveness, which has been demonstrated in previous studies. 14,15 Herein, anxiety was exhibited by control mice in the different anxiety models. However, for mice treated with fluoxetine, their status was found to improve, which aligns with earlier studies. 16,17 Prunin (50 and 100 mg/kg,p.o.) was also found to improve the anxiety behavior of mice. In fact, the state of anxiety was demonstrated to be improved in mice due to increases in the percentage of time spent in the open arm and the number of entries made into the open arm. 13,18,19 Furthermore, mice treated with prunin showed similar results to those treated with drugs that are proven to treat anxiety. 20 In the light-dark model, mice with anxiety preferred to remain in the dark; however, prunin-treated mice spent more time in the light box and made frequent entries into this chamber. Such a finding suggests that prunin exerts an anxiolytic effect. In the same model, prunin was found to improve mice immobility, which caused them to spend less time in the dark chamber, or decrease their immobility. 18,19,21 The effectiveness of prunin was also demonstrated by its modulation of neuronal release in the brain of mice. 22 -24
The social interaction model was developed to examine thesocial interaction behavior of mice and the anxiolytic behavior induced by social phobia, social failure/abnormality, and emotional immaturity. 13,18,19 The present study revealed that mice with anxiety participated in less social interaction, thereby aligning with previous studies. However, after prunin treatment, mice tended to be more active (ie, climbing and crawling under the condition of unfamiliar light).
In the social interaction test, mice were sensitive to both anxiogenic and anxiolytic effects, and a better alternative for anxiolytic compound assessment; this model is ideal for the treatment of social phobia, social failure/impairment, and emotional immaturity. Anxiety is a condition that is yet to be fully understood because of the changes in signaling molecules, especially neurotransmitters. 25 Preliminary studies have shown that GABAergic and serotonergic neurotransmission are involved in the etiology, expression, and treatment of anxiety. 3,21 The release of these neurotransmitters and stress are the new-age factors that create many complications in neuronal diseases, such as anxiety. 14 -17,26 The findings herein also reveal the variations in GABA and seroton in release at the synapse in mice. 27 Prunin is hypothesized to modulate the expression of cerebral serotonin, which is related to anxiety-like behavior in mice, and stimulates tryptophan, a precursor of serotonin and the main brain neurotransmitter involved in different mechanisms of anxiety. 28 Prunin promotes the availability of postsynaptic serotonin by binding to the 5-HT1A receptors that help to reduce anxiety in mice. 13,29 During an episode of anxiety, the tph2 gene is over-expressed in the 5-HTergic neurons of the mid-brain. 30 In addition, tph2 plays an important role in sleep, arousal, learning and memory, motor coordination, pain regulation, and ingestive behaviors. 31 In the present study, the tph2 gene was observed to be overexpressed 30,31 in the control animals. Slc6a4 is a serotonin transporter involved in 5-HT reuptake from the synaptic cleft to the presynaptic neuron. 32 -34 Because the 5-HT synthesizing enzyme (tph2) and transporter (slc6a4) genes were downregulated, the resultant 5-HT levels in anxiety-induced brains were decreased and further modulated by treatment with prunin.
Conclusion
In the present study, we found that prunin could exhibit a therapeutic effect against anxiety-like disorder. However, more experiments are still required to identify the molecular mechanism and expression levels of the 5-HT synthesizing enzyme (tph2) and transporter (slc6a4) genes, which are required to achieve anti-anxiety effects. Clinical trials are also required to confirm the effect of pruninon humans with anxiety disorders. Nevertheless, the findings presented here could serve as a basis for further investigation to determine the ideal use of prunin for the treatment of psychological disorders.
Materials and Methods
Preparation of Drugs and Chemicals
Fluoxetine is an active pharmaceutical ingredient procured from Qingdao Navid Biotechnology Co., Ltd. (Shandong, China). Prunin was purchased from Sigma Aldrich pvt Ltd. Beijing, China. Other chemicals, such as pyrogallol, quercetin, and serotonin, were purchased from Himedia laboratories (Mumbai). Other chemicals used in this study were of analytical grade.
Animals
Albino mice of both sexes (weight 25‐30 g) were obtained from registered breeders, housed under controlled conditions, and fed a standard pellet diet and water. The animal study was approved by the Animal Ethics Committee and the governing body (IEC-2019/05) for conducting animal experimentation.
Acute Toxicity Studies
The acute toxicity of prunin was determined according to the OECD423 guidelines up and down method. 18,19 Thereafter, doses of 50 and 100 mg/kg,p.o. of prunin were selected to observe pharmacological activity.
Experimental Models
Mice were randomly divided into four groups (six animals per group). In the normal control group, mice were administered normal saline (vehicle control) as per the experimental protocol. 11 Mice in the fluoxetine group (standard) were treated with Fluoxetine (10 mg/kg; p.o.) for 7 days. To prepare the prunin suspension before oral dosing, prunin was dissolved in 0.2% carboxymethyl cellulose in water. Two doses of prunin, 50 mg/kg,p. o. and 100 mg/kg, p. o., were administered to two groups (PR-I and PR-II).
Elevated Plus Maze Model
The test procedure and the scoring methods for the elevated plus maze test were performed as described by Kumar. 18 Briefly, the experiment was conducted between 9 a.m. and 4 p.m. to enable adaptation to the new environmental conditions. The trial began when an animal was placed on the central platform of the maze, facing the open arm. The following parameters were then measured: number of entries into the open and closed arm, time spent in the open and closed arm, and latency. 13,18,19
Light/Dark Model
The light-dark chamber was an open-top wooden box consisting of light and dark chambers. Mice were tested 60 minutes after drug (both fluoxetine and prunin, separately) administration and were placed in the middle of the arena of the light-dark chamber. The time spent on the light and darkside, number of crossings performed by mice, and the length of immobility were recorded after 10 minutes in each trial. 13,18,19,35 -37
Social Interaction Behavior of Mice
Mice were evaluated via social interaction tests using the protocol reported by Wang. 19 Briefly, mice were examined in a Perspex open-topped box (51 × 51 cm, length; 20 cm, height), with 17 ×17 cm marked are as on the floor. Mice were tested 1 hour prior to drug treatment (fluoxetine and prunin, administered separately). Thereafter, two mice were placed inside the box and their behavior was observed for 10 minutes. Mice were evaluated under two conditions: high light, unfamiliar arena (HU); or high light, familiar arena (HF). The social interaction behavior of animals was judged by the time they took to sniff a partner, crawl under or climb over the partner, or exhibit aggressive behavior. Five pairs of animals were selected to receive each dose. 13,18,19
Brain Tissue Extraction for the 5-HT Assay
After the animals were dissected, samples of their brain tissue were preserved. The brain tissue homogenate was prepared with 0.1 ml HCl-n-butanol (0.85 ml 37% HCl in 1 Ln-butanol) using a homogenizer. 13,18,19,35 The level of serotonin was determined using a previously reported method. 38 Further, sample readings were acquired at 360‐470 nm byspectroscopy.
Gamma Amino Butyricacid (GABA) Estimation
The whole brain of each animal from each group was preserved separately to estimate the level of GABA. Briefly, the whole-brain samples were homogenized with 5 ml of 0.01 M hydrochloric acid. Further sample processing and estimation were performed as previously described. 38 Sample absorbance was measured at 570 nm by spectrophotometry and standard GABA was used to determine the level of GABA in samples. 18,20
RNA Extraction and Real-time PCR
Total RNA was extracted using TRIzol reagent (Thermo Fisher Scientific Inc., Waltham, Massachusetts, USA), according to the manufacturer’s instructions. cDNA was synthesized using a Moloney murine leukemia virus reverse transcriptase kit (Promega Co., Fitchburg, Wisconsin, USA). Approximately 1.88 ng of cDNA was amplified using an Agilent Brilliant III Ultra-Fast qPCR SYBR Green Master Mix kit (Agilent Technologies, Santa Clara, CA, USA). The specific primers are listed in Table 3. The experiment was performed according to the method described by Pei-Tan Hsueh. 26 The PCR amplification profile included 40 cycles, followed by 95 °C for 5 s, and 60 °C for 10 s. At the end of each reaction, the raw data were automatically analyzed and an amplification plot as well as a Cq (quantification cycle) value were generated (Agilent Aria Mx 1.0). 39 Each gene (independent samples, n = 3) was first normalized to the reference gene using the following equation: ΔCq = Cqtarget gene−Cqtbp reference gene. To compare prunin-treated mice to anxiety-induced mice, the fold differences in gene expression were calculated using the following equation: ΔΔCq = (Cqtarget gene in the anxiety-induced group−Cqtbp reference gene in the anxiety-induced group)–(Cqtarget gene in the prunin-treated –Cqtbp reference gene in the prunin-treated); the relative fold was derived using 0.5ΔΔCq. 14
Primers for Tph2 and Slc6a4.

Statistical Analysis
All data were presented as Mean ± SEM and were analyzed using one-way analysis of variance (ANOVA) followed by Dunnett’s ‘t’ test [*P < .05, **P < .01, ***P < .001]. P values less than .05 were considered to indicate statistical significance.
Supporting Information
Significance of the Study
Prunindisplayed anti-anxiety effects in mice by modulating the release of serotonin and GABA at the synapses in the brain.
Future Findings
Based on the results obtained in the present study, exploring the molecular interaction between prunin and the signaling molecules in the brain may serve as the basis for future discoveries. Moreover, the findings obtained herein could prove beneficial in preclinical models and can be further applied to clinical models.
Footnotes
Acknowledgments
The authors would like to thank the participating institutes for supporting this study by providing there search facility and institutional funding required to carry out this study.
Statement of Human and Animal Rights
All experimental procedures involving animals were conducted in accordance with the institutional animal care review board guidelines of Central Hospital Affiliated to Shandong First Medical University, Jinan, 250013, China, and approved by the Administration Committee of Experimental Animals, Jinan,Shandong Province, China.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
