Abstract
Blue tansy essential oil (BTEO) (Tanacetum annuum L.) was analyzed by GC-MS and GC-FID using two different capillary column stationary phases. Sabinene (14.0%), camphor (13.6%), myrcene (8.0%), β-pinene (7.7%), and chamazulene (6.9%) were the main components using an SE52 column (non-polar). On a polar CW20M phase column, sabinene (15.1%), camphor (14.4%), α-phellandrene (7.9%), β-pinene (7.7%), and myrcene (6.9%) were the most abundant compounds. To assess the oil for potential applications in integrated pest management strategies, behavioral bioassays were conducted to test for repellency against yellow fever mosquito Aedes aegypti, and for attractant activity for Mediterranean fruit fly Ceratitis capitata. Results showed that BTEO was not effective in repelling Ae. aegypti (minimum effective dosage [MED]: 0.625 ± 0.109 mg/cm2 compared with the standard insect repellent DEET (N,N-diethyl-3-methylbenzamide). In assays with male C. capitata, BTEO displayed mild attraction compared with two positive controls (essential oils from tea tree Melaleuca alternifolia and African ginger bush Tetradenia riparia). Additional studies are needed to identify the specific attractant chemicals in BTEO and to determine if they confer a synergistic effect when combined with other known attractants for C. capitata. To the best of our knowledge, this study represents the first investigation of BTEO for repellency against the mosquito vector Ae. aegypti and for attractancy to C. capitata, a major agricultural pest worldwide.
Keywords
Global warming is likely to impact human life in part through an increase in infectious diseases to humans and in part through a decrease in agricultural production. When the average temperature is higher, the metabolic rate of insects accelerates, which contributes to greater losses of agricultural products and a concomitant need for increased efficiency in food production. Additionally, there is a shift in insect numbers to new geographic locations. 1 The ease of human travel has led to globalization which comes with an increased risk of introduction of exotic pathogens and their invertebrate vectors to new locations. 2,3 This is the case with Aedes aegypti L. (Diptera: Culicidae) and other Aedes species, primary vectors of chikungunya, Zika, yellow fever, and dengue viruses, which affect the health and well-being of humans globally. 4 Moreover, climatic changes could have a significant impact on the development and distribution of agricultural insect pests in new areas. 5,6 Furthermore, many countries enforce subsequent quarantine regulations against agricultural pests such as invasive species of tephritid fruit fly; the consequent restrictions have a significant detrimental impact on the agricultural productivity to international markets. 5 -9 For example, the outbreak of Mediterranean fruit fly (medfly), Ceratitis capitata Wiedemann (Diptera: Tephritidae) in the Dominican Republic in March 2015 resulted in an export ban that cost the country an estimated $40 million. 10 Current prevention of mosquito-borne diseases and control of agricultural pests largely rely on the use of synthetic chemical pesticides. However, excessive exposure may lead to development of pesticide resistance as well as adverse effects on human health, the environment, and non-target (beneficial) organisms. 11,12 Therefore, there is a need for research to identify effective and safe alternative approaches and strategies for suppression of disease vectors and agricultural pests. In recent years, reduced-risk agents such as essential oils and natural (“green”) volatile compounds have gained increased recognition as key elements for improving integrated pest management strategies. 8,13 -27
Tanacetum annuum L. (synonyms: Balsamita annua L., B. multifida Clemente, and Chrysanthemum annuum [L.] Fiori) is a member of the giant family of Asteraceae (Compositae). Members of the genus Tanacetum have been used for medicinal purposes for decades due to their wide range of pharmacological and chemical properties as well as excellent medicinal values. 28,29 Various Tanacetum representatives have also shown a high insect repellent activity, such as against Tribolium castaneum, Ae. aegypti and Ixodes ricinus. 30 -33
In a continuing effort to identify and discover new semiochemicals (chemical signals) for control of insect pests suited to protect human populations against mosquito-borne diseases and for sustainable crop protection and food security, this present study was conducted to evaluate the potential of commercially available blue tansy essential oil (BTEO) for its repellency against Ae. aegypti with three hoursuman volunteers using arm-in-cage bioassays and for the attraction of C. capitata in short-range laboratory bioassays. To the best of our knowledge, this study represents the first investigation of BTEO for repellent and attractant activities.
Results and Discussion
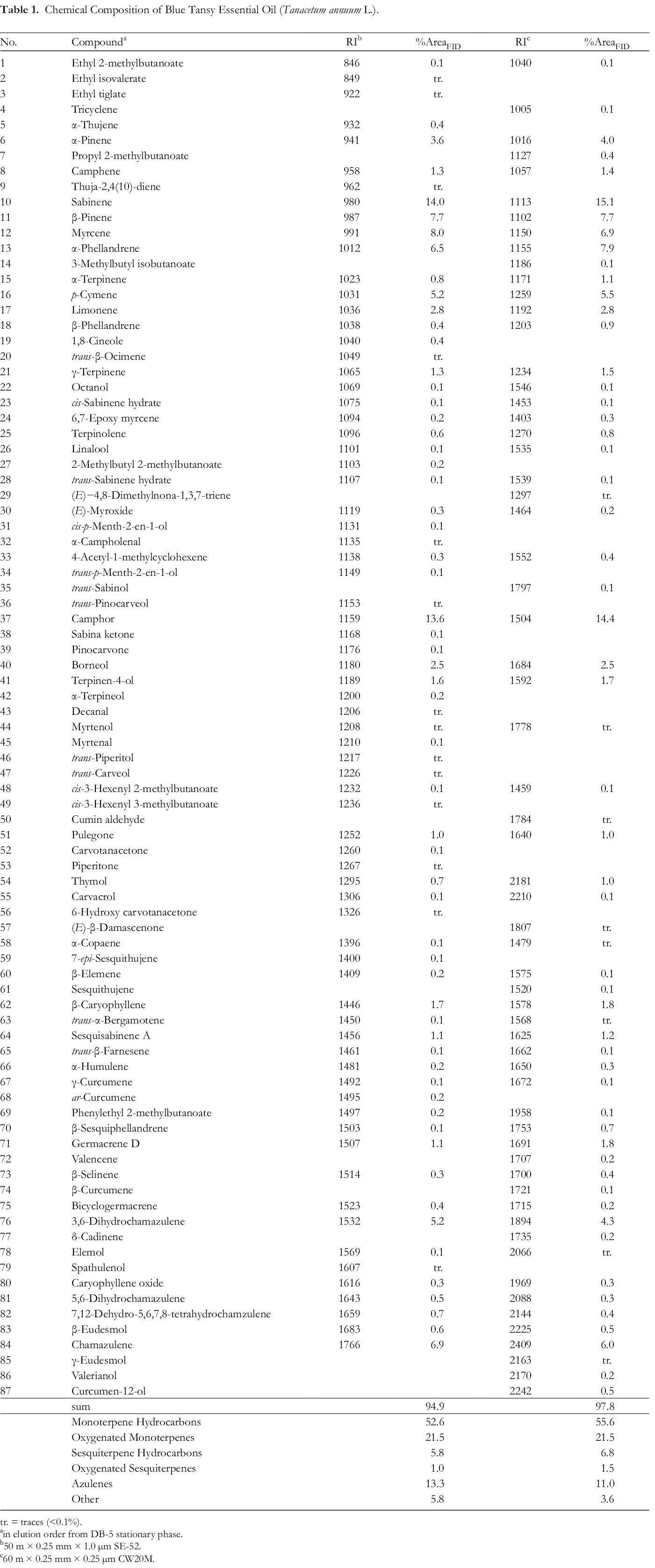
BTEO was analyzed simultaneously by GC-MS/GC-FID using two columns differing in polarity (see Experimental) to ensure a more comprehensive identification and detect coeluting constituents. The results of quantitative and qualitative oil analyses are given in Table 1. A total of 73 components were identified when analyzed on a non-polar column (SE52), accounting for 94.9% of the oil. The oil contained sabinene (14.0%), camphor (13.6%), myrcene (8.0%), β-pinene (7.7%), chamazulene (6.9%), and α-phellandrene (6.5%). Additional components in an amount above 5% were 3,6-dihydrochamazulene (5.2%) and p-cymene (5%). Separation on a polar column (CW20M) allowed for the identification of 62 compounds (97.8% of the oil). The most abundant constituents were sabinene (15.1%), camphor (14.4%), α-phellandrene (7.9%), β-pinene (7.7%), and myrcene (6.9%), followed by chamazulene with 6.0% and p-cymene (5.5%). There is a good correlation between compounds identified using the two methods. The results corroborate findings of some earlier publications 34,35 whereas they differ from an investigation describing BTEO derived from Morocco. 36 The authors of that study found a high content of sabinene (22.3%), which was probably due to environmental and climatic conditions. 37,38 Interestingly, our sample showed four azulene derivatives: Besides chamazulene and 3,6-dihydrochamazulene, small amounts of 5,6-dihydrochamazulene (0.5%/0.3%) and 7,12-dehydro-5,6,7,8-tetrahydrochamazulene (0.6%/0.5%) were identified, accounting for a total of 13.2% and 11.1%, respectively.
Chemical Composition of Blue Tansy Essential Oil (Tanacetum annuum L.).
tr. = traces (<0.1%).
ain elution order from DB-5 stationary phase.
b50 m × 0.25 mm × 1.0 µm SE-52.
c60 m × 0.25 mm × 0.25 µm CW20M.
The mosquito repellency assays with Ae. aegypti mosquitoes revealed BTEO to have a MED (see Experimental) for repellency of 0.625 ± 0.109 mg/cm2; however, this indicated weak repellency compared to the reference standard repellent, DEET (N,N-diethyl-3-methylbenzamide) (MED = 0.008 ± 0.002 mg/cm2). The repellency of BTEO was significantly less (F = 10.741; df = 4.10; P < .001) than the other three oils, Anthemis scorbicularis essential oil (AMEO), 39 Caryopteris × clandonensis essential oil (CCEO), 40 and Prangos platychlaena essential oil (PPEO), 41 but all these oils were less potent than DEET (Figure 1). Additionally, we have tested some of the predominant compounds in BTEO for their repellency. Sabinene and α-phellandrene were not repellent as high as a MED value of 0.187 ± 0 mg/cm2. Myrcene did not repel mosquitos at a concentration of 0.375 ± 0 mg/cm2. (−)-Camphor had slightly better repellency, with a MED of 0.375 ± 0 mg/cm2, than its (+)-enantiomer (0.500 ± 0.125 mg/cm2). In our prior study, we found that (−)-β-pinene showed mild repellency with a MED value of 0.140 ± 0.047 mg/cm2, while p-cymene was not repellent at the highest tested dose of 1.5 mg/cm2. 15,19

Minimum effective doses (MED mg/cm2 ± SE) for repellency values for essential oils tested on human volunteers against Ae. aegypti mosquitoes. Response observed with blue tansy essential oil (BTEO) was compared with that of known MED values, Anthemis scorbicularis essential oil (AMEO), 39 Caryopteris × clandonensis essential oil (CCEO), 40 Prangos platychlaena essential oil (PPEO), 41 and to the standard insect repellent DEET (N,N-diethyl-3-methylbenzamide). Means followed by the same letter are not significantly different based on the Tukey-Kramer method; α = 0.05, with human subjects, n = 3.
In short-range laboratory bioassays with male C. capitata, there were significant differences in mean behavioral response to five essential oil treatments (F = 10.087; df = 4,20; P < .001). Attraction to BTEO was significantly lower than that observed with two positive controls, that is, tea tree oil (TTO) Melaleuca alternifolia (Maiden and Betche) Cheel., 25 and ginger bush Tetradenia riparia (Hochst.) Codd essential oil (TREO) 26 (Figure 2). However, the level of attraction to BTEO was equivalent to that seen with two sources of mastic gum (Pistacia lentiscus L. var. chia) essential oil (MGEO-1 and 2). 27 It is reasonable to assume that there are chemical constituents common to BTEO and MGEO that confer the mild attraction of C. capitata. We recently analyzed the chemical composition of MGEO-1 and 2, reporting that α-pinene (52% to 56%), myrcene (19% to 20%), and β-pinene (3%) were the dominant components characteristic of wild-type MGEO. 27 Studies using electroantennography (EAG) to quantify olfactory responses confirmed that α-pinene and myrcene elicit weak EAG responses in male C. capitata. 42,43 Our research group previously found that myrcene was negatively correlated with attraction of male C. capitata in short-range bioassays; 21 however, no studies have included enantiomeric identification of these compounds. Therefore, individual enantiomers of α- and β-pinene need to be evaluated for attractancy of male C. capitata. In addition, BTEO should be combined with other known medfly attractants (eg, TTO and other essential oils high in α-copaene 21 to assess potential synergistic effects, as has been observed with kairomones for other insect pests. 44

Attraction (mean ± SE) of male Mediterranean fruit fly, Ceratitis capitata, to essential oils presented in short-range laboratory bioassays. Response observed with blue tansy essential oil (BTEO) was compared with response to known strong attractants, tea tree oil (TTO), 25 Tetradenia riparia essential oil (TREO) 26 and known mild attractants, two sources of mastic gum essential oils (MGEO-1 and MGEO-2). 27 Bars topped with the same letter are not significantly different (Tukey HSD mean separation, P < 0.05).
In conclusion, the chemical composition of BTEO differed from other blue oils, such as Matricaria chamomilla essential oils from different geographic locations, 45 by the presence of four azulene derivatives. Due to their high chamazulene 46 and low thujone content, blue oils are considered as potential skin care applications with a wound healing 47 and anti-inflammatory 48 effect. The reason for choosing this particular oil for our investigation was that it was characterized by a very high amount of chamazulene, which was screened prior to testing, and therefore would be even more beneficial for people suffering from skin disorders. The results should be transmissible to other commercially available BTEOs with similar high chamazulene contents. However, the repellent activity of the investigated BTEO was too weak for practical use as, for example, skin care products against Ae. aegypti. There was mild attraction of male C. capitata to BTEO; further studies are needed to identify the specific attractive components and to determine potential application for improvement of medfly lures.
Experimental
Essential Oil
Blue tansy essential oil (BTEO) was provided by Kaders Paul GmbH Import u. GroßHdl, Hamburg, Germany, Ref 170911.
Essential Oil Analysis
GC-FID and GC-MS analysis were carried out simultaneously in one instrument configuration using a Finnigan Thermo Quest Trace GC with two split/splitless injectors each connected to a GC capillary column of different polarity, an FID detector, a Finnigan Automass quadrupole mass spectrometer (Thermo Quest Finnigan S.A., France) and a CTC GC-PAL autosampler. One GC injector was connected to a 60 m × 0.25 mm × 0.25 µm Carbowax 20 M (polyethylene glycol) column (J&W Scientific, USA), the other injector was coupled to a 50 m × 0.25 mm x 1.0 µm SE-54 (5% diphenyl, 1% vinyl-, 94% dimethylpolysiloxane) fused silica column (CS Chromatographie Service, Germany). Both columns were joined at the end with a quartz Y connector and the combined effluents split with another Y connector to the FID and MS detector. A short (ca. 0.3 m × 0.1 mm) uncoated deactivated GC capillary served as a flow restriction transfer line between the MS ion source and the split connector and a ca. 1 m × 0.25 mm GC capillary was connected to the FID detector. Only one injector was operated and pressurized at a time at a normal flow rate while the other was left with only a very small gas flow to protect the unused column.
Helium 5.0 was used as a carrier gas at a constant flow rate of 1.5 mL/min; injector temperature was 230 °C; the FID detector temperature, as well as the GC-MS interface heating were 250 °C; the ion source was at 150 °C; EI mode at 70 eV; scan range 40‐500 amu. The following temperature program was applied: 46 °C for 1 minute to 100 °C at a rate of 5 °C/min; 100 to 230°C at 2 °C/min; 230 °C for 13.2 minutes. Essential oil samples were diluted 1:20 with dichloromethane and 1 µl of this solution was injected into the GC at a split ratio of 1:100.
Thermo Xcalibur 1.2 software was used for identifying the compounds by correlating mass spectra to databases of NIST 08, 49 Wiley 8th ed., 50 Adams library, 51 MassFinder terpenoids library 52 and our own library. Retention indices were determined with the use of the measured retention times of a series of n-alkanes that elute over the whole span of the chromatogram and calculated according to the method of van den Dool and Kratz 53 and confirmed using retention indices of data from the literature and those from reference compounds, if necessary. Quantification was performed using normalized peak area calculations of the FID chromatogram without (by first approximation) relative FID-response factors.
Repellency Bioassays
Female Ae. aegypti mosquitoes were obtained from a laboratory colony maintained at the Mosquito and Fly Research Unit at the Center for Medical, Agricultural and Veterinary Entomology, Gainesville, FL, USA. Repellency was determined by the MED(mg/cm2), which is the minimum threshold surface concentration necessary to prevent mosquitoes from biting through the treated surface. The rearing conditions and detailed methods of evaluation have been described in earlier studies. 14,15,19,54,55 BTEO was applied on the cloth at the highest concentration available and subsequent tests were with serial two-fold dilutions in acetone until the concentration failed to repel (greater than or equal to five bites in one minute) in a cage of 500 mosquitoes. The lowest concentration which passed was determined to be the MED for compound or plant extracts. The MED for each candidate was averaged from these tests of participants and reported as a mean MED ± SE.
The test was conducted by having each volunteer affix the treated cloth onto a plastic sleeve to cover a 32 cm2 window previously cut into the sleeve. Each of the volunteers wore this sleeve-cloth assembly above a nylon stocking that covered the arm with the hand of each volunteer protected by a glove. If ~1% or the volunteer receives five bites through the cloth over the sleeve during one minute test, the oil was considered to have failed, that is, was not repellent at that concentration. 14,15,19,54,55 Untreated (acetone and ethanol) solvent controls exhibited 0% repellency. The standard repellent DEET (N,N-diethyl-3-methylbenzamide, 97% Sigma Aldrich, St. Louis, MO, USA) diluted in ethanol was included in the bioassays and results were compared with our recent studies of AMEO, 39 CCEO, 40 and PPEO. 41 Compounds such as sabinene (CAS # 3387‐41-5), α-phellandrene (CAS# 99‐83-2), (−)-camphor (CAS# 464‐48-2), (+)-camphor (CAS# 464‐49-3), (−)-β-pinene (CAS# 18172‐67-3) and p-cymene (CAS# 99‐87-6) were purchased from Sigma-Aldrich Co. (St. Louis, MO). Analysis of variance (ANOVA), followed by mean separation with Tukey-Kramer method; α = 0.05, n = 3, was performed using MS Excel (Microsoft Office 2016 Professional).
Short-Range Attraction Bioassays
Sterile male C. capitata, 5‐10 days posteclosion, were used in this study. The source of insects, rearing protocols and experiment design for laboratory bioassays were as described previously. 25 -27 All observations were carried out at room temperature in small collapsible cages (20.3 × 20.3 × 20.3 cm) into which 50 flies were introduced 1 hour prior to the start of each experiment. Tests were initiated by introducing a Petri dish (53 mm diameter × 12 mm height) with a filter paper disk containing a sample of essential oil (10 µL of a 10% dilution in acetone). Each test used a separate cage to observe fly response to (i) BTEO, (ii) TTO, a known strong attractant, 25 obtained from Melaleuca alternifolia (Maiden and Betche) Cheel. (Essential Oil India-SAT Group, Kannauj, India), (iii) Tetradenia riparia, TREO, a good attractant, 26 and (iv) mastic gum (Pistacia lentiscus L. var. chia) essential oils, MGEO-1 and 2, a mild attractant. 27 Tests were concluded after 30 minutes, at which point results were recorded as the number of flies within a Petri dish, which was then converted to percentage of flies attracted. Tests were replicated five times, and the position of the cages was randomized between replicate runs. ANOVA, followed by mean separation with Tukey test (P < .05), was used to analyze results (Systat Software). 56
Footnotes
Acknowledgments
The authors are grateful to Kaders Paul GmbH Import u. GroßHdl, Hamburg, Germany, for providing tansy essential oil for this study. We thank Natasha M. Agramonte, Greg Allen (USDAARS, CMAVE, Gainesville, FL) and Micah Gill (USDA-ARS, Miami, FL) for bioassays; Monica Blanco, Amanda Perez-Castro and Sean Brown (USDA-ARS, Miami, FL) for technical assistance; Dr. Martina Hoeferl (University of Vienna, Austria) and Dr. Eugene K. Blythe (Auburn University, Auburn, AL, USA) for helpful suggestions; and journal reviewers/editors for critical reviews of an earlier version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Mention of trade names or commercial products in this publication is solely for the purpose of providing specific information and does not imply recommendation or endorsement by the U.S. Department of Agriculture. USDA is an equal opportunity provider and employer.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the United States Department of Agriculture (Project Number: 6038-22000-006-00-D) and the U.S. Department of Defense through the Armed Forces Pest Management Board.
