Abstract
Fractionation of the leaf extract from Murraya exotica led to the successful isolation of 12 compounds (
Introduction
Tumor necrosis factor (TNF)-related apoptosis-inducing ligand (TRAIL) is a member of the TNF superfamily that can induce apoptosis in cancer cells after complexing with death receptors 1 . TRAIL has attracted great interest as an anticancer target because it selectively induces apoptosis in cancer cells while sparing normal cells 2 . Unfortunately, over time, some cancers become resistant to TRAIL-apoptotic pathways such as gastric cancer, breast cancer, lung cancer, prostate cancer, and colon cancer 3 . We have already reported some compounds with TRAIL-resistance-overcoming activity1,4; however, we are still searching for naturally derived compounds with TRAIL-resistance-overcoming activity.
Murraya exotica, an evergreen shrub in the Rutaceae family, has ethnomedical uses described in the Chinese Pharmacopeia, such as anti-stomachache, anti-rheumatic arthralgia, anti-toothache, anti-swelling, and analgesic 5 . M. exotica is reported to contain a variety of secondary metabolites, including alkaloids 6 , coumarins 7 , flavonoids 8 , and dipeptides 9 , having anti-implantation 10 , antioxidant 11 , anti-inflammatory 12 , and anti-proliferative activities 13 . However, the TRAIL-resistance-overcoming activity of M. exotica has not been explored.
Our screening results indicated that M. exotica extract has TRAIL-resistance-overcoming activity. In this study, we isolated 12 compounds (
Result and Discussion
Isolation and Structure Elucidation
Although there was a report of antimalarial and antipyretic activity of the methanolic extract of M. exotica
14
, the TRAIL-resistance-overcoming activity of M. exotica has not been studied. Plant extracts in our library were screened for TRAIL-resistance-overcoming activity and the M. exotica extract was identified as a hit sample (40% inhibition at 50 µg/mL). M. exotica leaves were extracted thrice with methanol (MeOH), passed through a Diaion HP-20 column, and partitioned with hexane, ethyl acetate (EtOAc), and butanol (BuOH) to afford their respective layers. The EtOAc layer was fractionated by column chromatography and preparative high-performance liquid chromatography (HPLC) together with the TRAIL-resistance-overcoming activity test to yield compounds
The isolated compounds were identified as xanthinosin (
Compounds
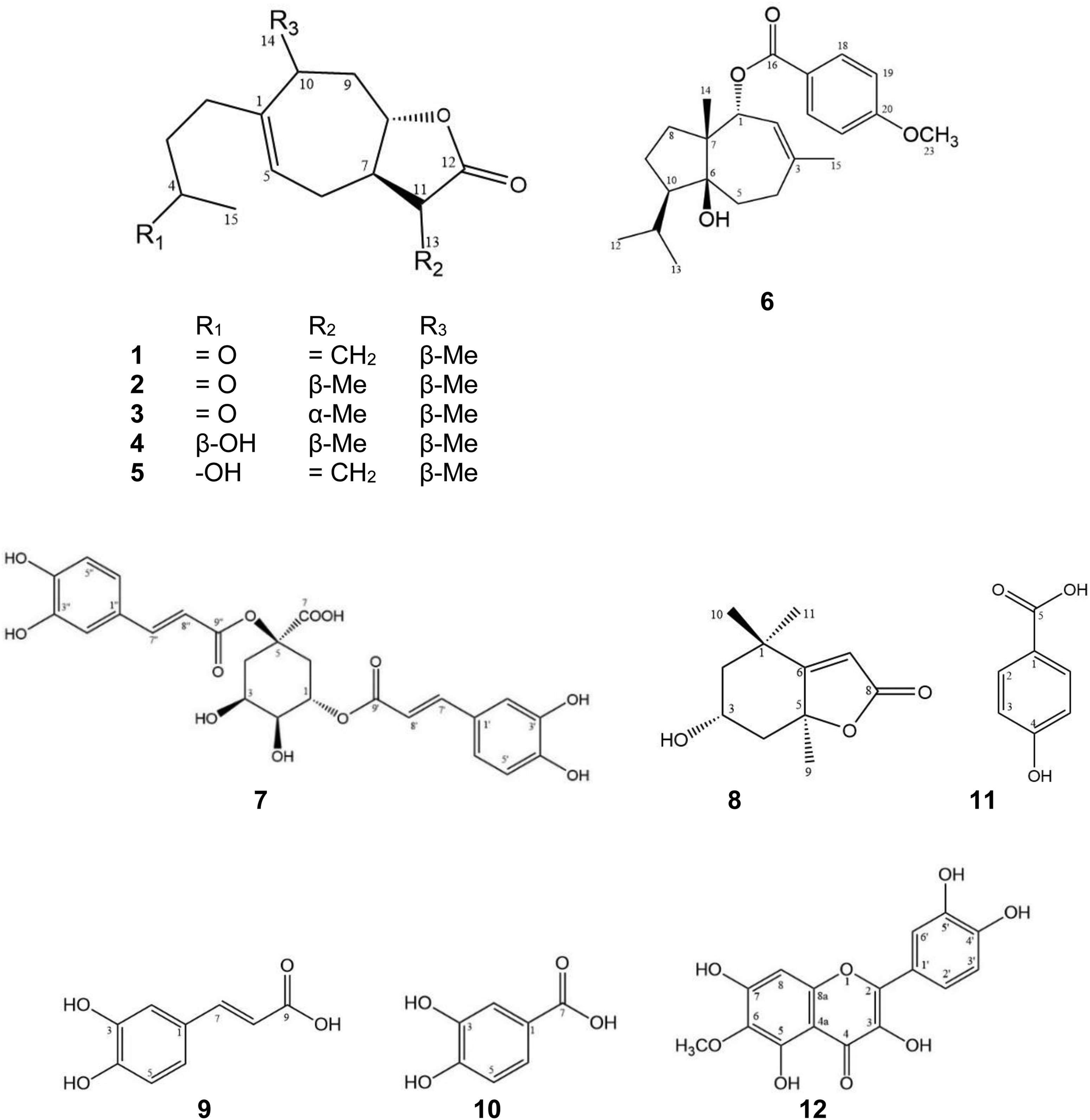
Structures of compounds
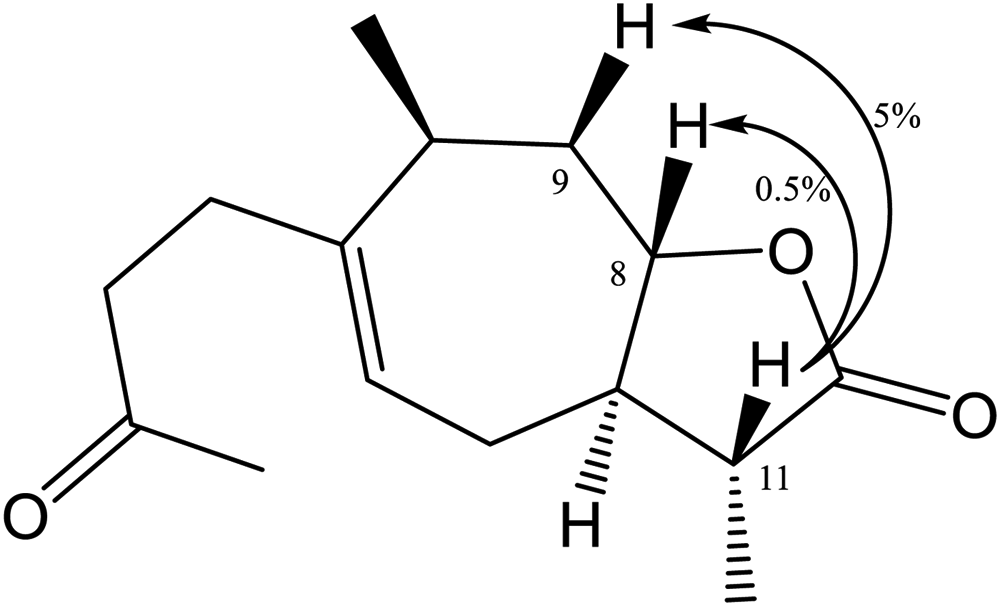
Key NOE difference in 11β, 13-dihydroxanthinosin (
TRAIL-Resistance-Overcoming Activity of Isolated Compounds
All the isolated compounds (

TRAIL-resistance-overcoming activity of isolated compounds (
Although
Effect of Compounds 2, 7, and 8 on the Expression of Apoptotic Proteins
The related apoptotic proteins were analyzed after overnight treatment of AGS cells with

Western blot analysis of cleaved caspase-3, procaspase 8, cleaved caspase-8, DR5, CHOP, p53, Bcl-2, FLIP, and cleaved caspase-9 proteins in AGS cells after 24 h treatment using
In the intrinsic pathway, the released cleaved caspase-8 activates Bid and transforms it into truncated Bid (tBid). Then, tBid translocates to the mitochondria and releases cytochrome c, which binds to Apaf-1 and forms apoptosome that subsequently activates the proteolytic cleaved caspase-9
27
. The released cleaved caspase-8 and −9 activate the transformation of procaspase-3 to active cleaved-caspase-3 in the final step. (-)Loliolide (
Conclusion
Six natural sesquiterpenes (
Experimental
General Experimental Procedures
The analytical instruments and materials used in this study included a P-2200 polarimeter (JASCO) for optical rotations; an ECZ-600 spectrometer for NMR spectroscopy (solvent chemical shifts were used as the internal standard); a PU 980 (JASCO) pump and UV 970 (JASCO) for HPLC; an LCMS system including LCMS 2020 (Shimadzu) for ESI-MS; silica gel 60 F254 (0.25 mm, Merck) and silica gel 60 RP-18 F254S (0.25 mm, Merck) for analytical thin layer chromatography; silica gel 60 N (Kanto Chemical Co., Inc.) for column chromatography; and COSMOSIL AR-II (10.0 × 250 mm, Nacalai Tesque), Inertsil-ODS-3 (10.0 × 250 mm, GL sciences), and YMC-SIL06 (10 × 250 mm, YMC Co., Ltd) for preparative HPLC.
Plant Material
Leaves of M. exotica (430 g) were collected from Bangladesh and deposited at the Department of Natural Products Chemistry, Graduate School of Pharmaceutical Sciences, Chiba University, Japan, under voucher specimen No. KKB301.
Extraction and Isolation
The dry leaf powder of M. exotica was macerated with MeOH (3 × 800 mL) at room temperature. The crude extracts (18.3 g) were chromatographed on a Diaion HP-20 (5 × 15 cm) to afford MeOH (9.1 g) and acetone extracts (3.8 g). Each extract was partitioned into n-hexane, EtOAc, BuOH, and water layers. The MeOH-EtOAc layer (2.4 g) was separated on a sequence of silica gel 60N and ODS columns to yield fractions 4A-4E. Fraction 4B (24.5 mg) was eluted on an Inertsil ODS-3 (10 × 250 mm) column using 60% MeOH as the mobile phase to afford
The structures of the isolated compounds were elucidated based on 1H- and 13C-NMR, heteronuclear multiple quantum coherence and heteronuclear multiple bond coherence spectral analysis, and ESI-MS data, as well as by comparison with published data. The physical and spectroscopic data of isolated compounds
Bioassay
The TRAIL-resistance-overcoming activity of the isolated compounds was determined according to our previously published method
4
. The results in the bar graph are expressed as mean ± SD together with significance tests. Western blot analysis of compounds
Supporting Information
NMR spectra and TRAIL-resistance-overcoming activity of isolated compounds (PDF).
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the KAKENHI (grant number 20H03394, 20K16024).
