Abstract
1-Deoxy-D-xylulose 5-phosphate reductoisomerase (DXR) is a key enzyme in the methylerythritol phosphate pathway for terpenoid biosynthesis. Furthermore, it is an ideal target for the screening of novel antibiotics because it is present in causative organisms, but absent from humans. To identify more lipophilic DXR inhibitors from natural resources, we tested the DXR inhibitory activity of five proanthocyanidins in this study. The results indicated that all these compounds specifically restrained the activity of DXR, with procyanid B2 exhibiting a relatively low effect against DXR (IC50 ∼ 305 μM) and procyanid C1 displaying moderate activity (IC50 75.1 μM). The other three compounds cinnamtannin A2, cinnamtannin B1, and cinnamtannin D1 (IC50 ∼ 89.3, 105.0, and 97.8 μM, respectively) showed DXR inhibitory effects that were slightly weaker than that of procyanid C1. In addition, based on the initial characterization, the structure–activity relationship of this series of compounds against DXR is discussed.
Keywords
Introduction
1-Deoxy-D-xylulose 5-phosphate reductoisomerase (DXR), the key enzyme of the methylerythritol phosphate (MEP) pathway for terpenoid biosynthesis,
1
mediates the first committed step of this pathway, namely, the transformation of 1-deoxy-D-xylulose 5-phosphate (DXP) (

The first committed step of the methylerythritol phosphate (MEP) pathway for terpenoid biosynthesis and its inhibitors.
Proanthocyanidins are one of the richest sources of polyphenols in plants. 18 Previous studies have shown that such compounds possess many types of biological activities, including antioxidant, anticancer, antidiabetic, anti-obesity, and cardioprotective.19–22 In addition, they display a significant antibacterial activity. 23 In the present study, we have described functional bioassays of five proanthocyanidin compounds, procyanid B2 (PB2), procyanid C1 (PC1), cinnamtannin A2 (CA2), cinnamtannin B1 (CB1), and cinnamtannin D1 (CD1) (Figure 2), performed to investigate their inhibitory activity against DXR. Based on these results, we have discussed the structure–activity relationships of these compounds.

Chemical structures of the proanthocyanidin compounds.
Materials and Methods
Materials
Proanthocyanidins PB2, PC1, CA2, CB1, and CD1 were isolated and elucidated from the bark of Cinnamomum cassia (C. cassia) and C. tamala using a previously described method, 22 and stock solutions of each compound (10 mg/mL) were prepared in either 40% aqueous DMSO or 0.01% Triton X-100 just before use. Triton X-100 and 2,4-dinitrophenylhydrozine (DNPH) were purchased from Sinopharm Chemical Reagent Co., Ltd (Beijing, China), NADPH from Gen-View Scientific Inc. (Tallahassee, FL, USA), fosmidomycin from Toronto Research Chemicals, Inc. (North York, ON, Canada), and calf intestinal alkaline phosphatase (CIAP) from Takara (Dalian, China). DXP was synthesized using a previously reported procedure. 24 E. coli DXR was prepared according to a previously described method. 25 HPLC-grade methanol was purchased from Sigma-Aldrich Chemical Co. (Shanghai, China). All other chemicals were of analytical grade.
Determination of the Inhibitory Activity of the Proanthocyanidins Against E coli DXR
The inhibitory activity of the proanthocyanidins against E. coli DXR was assessed using a pre-column derivatization HPLC method published previously utilizing DNPH as a derivatizing reagent.
17
The reaction mixture comprised 50 mM Tris-HCl (pH 7.0), 5 mM MgCl2, 0.5 mM NADPH, 2% (w/v) DMSO or 0.01% Triton X-100, and 4 μg/mL DXR in a final volume of 100 μL. Proanthocyanidins (PB2, PC1, CA2, CB1, and CD1) were added to the mixtures, respectively before the addition of DXP (final conc. 1 mM) to initiate the reaction. In the control assay, fosmidomycin (
Characterization of CIAP Inhibition of the Proanthocyanidins
The HPLC assay described above was also employed to measure the inhibitory activity of the proanthocyanidins against CIAP. The reaction mixture consisted of 10 μL CIAP buffer and 2 U CIAP in a final volume of 100 μL. Stock solutions of the proanthocyanidins were added individually to each reaction mixture to final concentrations of 0.1 and 1.0 mM before the addition of DXP (final conc. 1 mM) to initiate the reaction. The mixtures were then incubated at 37°C for 120 min before the CIAP was inactivated in boiling water for 3 min. Subsequently, the reaction mixtures were derivatized with DNPH and analyzed using the HPLC procedure described in section “Determination of the Inhibitory Activity of the Proanthocyanidins Against E. coli DXR.”
Measurement of Particle Size by Dynamic Light Scattering (DLS)
Particle sizes of the proanthocyanidins in Tris-HCl buffer (50 mM, pH 7.0) containing 2.0% DMSO (v/v) were determined at room temperature in the absence and presence of DXR (final conc: 4 μg/mL) using a DLS analyzer (NICOMP-380, Particle Sizing Systems Inc., Santa Barbara, CA, USA). The detection time was set at 10 min. The concentrations of proanthocyanidins used were about twice their IC50 values. A solution of 2.0% aqueous DMSO was used as a reference, and baicalein as a positive control.
Results
Evaluation of Pre-Column Derivatization HPLC Method for its Applicability in This Study
Considering the strong UV absorption of the proanthocyanidins at 340 nm, which could definitely interfere with the quantification of DXR activity using a typical UV protocol, 26 we decided to determine DXR inhibitory activity by the pre-column HPLC method described in section “Determination of the Inhibitory Activity of the Proanthocyanidins Against E. coli DXR.” 17 To evaluate the applicability of the HPLC method in this study, we first chromatographed reaction mixtures {50 mM Tris-HCl buffer (pH 7.4), 5 mM MgCl2, 2% (w/v) DMSO} containing each of the 5 proanthocyanidins after the mixtures had been hydrolyzed with CIAP and derivatized with DNPH to elucidate whether the addition of these compounds would disturb the analysis of DXP. The HPLC results depicted in Figure 3 show that after CIAP and DNPH treatment, only DXP displayed a peak at a retention time of ∼8.7 min (DX-DNPH); no peak at retention times ranging from 8 to 9.5 min for any of the proanthocyanidins was detectable. This could have occurred because the proanthocyanidins might have been completely decomposed under the relatively harsh reaction conditions for dephosphorylation (pH 9.5, 120 min) and DNPH derivatization (about 1% perchloric acid, 45 min). Fortunately, the decomposed proanthocyanidins did not interfere with the determination of DXP. Thus, we can conclude that the pre-column derivatization HPLC procedure is suitable for the measurement of the DXR inhibitory activity of the proanthocyanidins.

Evaluation of interference of the proanthocyanidins on the quantification of DXP by the pre-column derivatization HPLC method. The HPLC analyses were carried out using the procedure described in “Materials and Methods,” section “Determination of the Inhibitory Activity of the Proanthocyanidins Against E. coli DXR.” Line 1, DNPH only; Line 2, DNPH-derivatized DXP (1 mM) after being treated with CIAP; Lines 3 to 7, DXR assay mixtures containing either PB2, PC1, CA2, CB1, or CD1 (200 μM) after being treated with CIAP and DNPH.
Effects of the Proanthocyanidins on the Activity of CIAP
In the DXR inhibition experiments, CIAP was employed to dephosphorylate residual DXP prior to DNPH derivatization; consequently, whether the procyanidins possess any inhibitory activity against CIAP is crucial to assess accurately their DXR inhibitory activity. Therefore, we examined the effects of the proanthocyanidins on the activity of CIAP accordingly. The acquired data indicate that, even at concentrations up to 1 mM, none of the proanthocyanidins suppressed CIAP activity (Figure 4).

Effects of the proanthocyanidins on the activity of CIAP. The reaction mixtures comprised 10 μL CIAP buffer and 2 U CIAP in a final volume of 100 μL; DXP was added (final conc. 1 mM) to start the reaction. The reaction mixtures were incubated at 37°C for 120 min, then derivatized with DNPH, and analyzed using the HPLC method. For assays of the proanthocyanidins, the compounds were added individually before the addition of DXP (2 concentrations were employed for each compound).
DXR Inhibitory Activity of the Proanthocyanidins With 2% DMSO as a Co-Solvent
Based on the data mentioned above, we determined the DXR inhibitory activity of these proanthocyanidins using 2% DMSO as a co-solvent. At a final concentration of 100 μM, all the compounds exhibited an unequivocal DXR inhibitory activity; among these, compound PC1 showed the strongest inhibitory activity, with an inhibition rate of 64.2% against DXR. CA2, CD1, and CB1 exhibited slightly weaker inhibition of DXR (54.9%, 50.6%, and 46.9%, respectively) than PC1. The inhibitory activity of PB2, however, was significantly lower than that of the above compounds, with an inhibition rate of only 9.3%. When the final concentration of the proanthocyanidins was increased to 200 μM, PC1 almost completely hindered the activity of DXR; compounds CA2, CD1, and CB1, however, reduced the DXR activity by more than 80%. PB2 showed only a 26.7% inhibition of DXR at this concentration, but at a concentration as high as 900 μM, it exhibited almost complete inhibition of DXR activity, which was approximately 4.5 times as high as that of PC1. The concentration-inhibition rate curves of these compounds are depicted in Figure 5, from which it can be clearly observed that these proanthocyanidins display a significant concentration-dependent inhibitory activity against DXR. The IC50 values are shown in Table 1. As a positive control, the inhibition curve of fosmidomycin against DXR activity was also determined using the pre-column derivatization HPLC method (shown as an inset in Figure 5A). Its IC50 was calculated and is listed in Table 1.
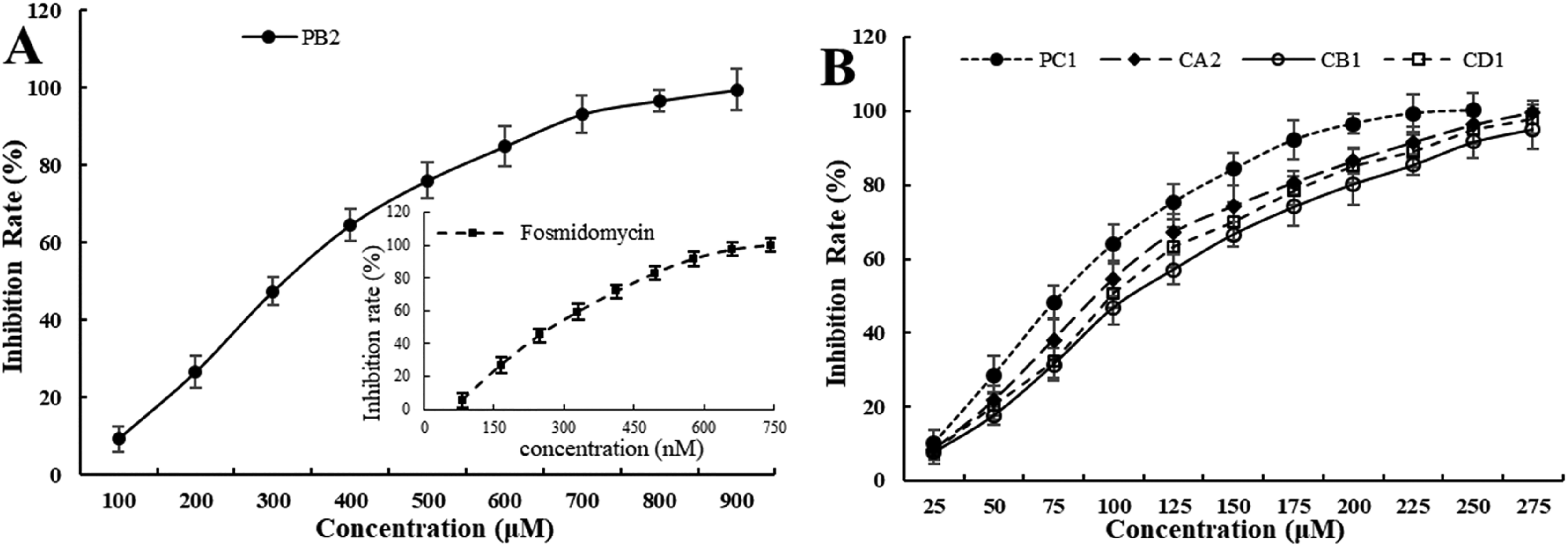
Inhibition curves of the proanthocyanidins against E. coli DXR. (A) Inhibition curve of PB2 (inset: inhibition curve of fosmidomycin); and (B) inhibition curves of PC1, CA2, CB1, and CD1.
IC50 Values of Fosmidomycin and the Proanthocyanidins Against E coli 1-Deoxy-D-Xylulose 5-Phosphate Reductoisomerase (DXR).

*The value shown in parenthesis is a measure of the inhibitory activity of fosmidomycin against E. coli DXR obtained from the literature. 27
DXR Inhibitory Activity of the Proanthocyanidins Using 0.01% Triton X-100 as a Surfactant
A significant drawback in the search for enzyme inhibitors is the disproportionate number of false hits associated with drug candidates that produce colloidal aggregates, especially when phenolic compounds are screened. In fact, some flavonoid compounds, such as myricetin, baicalein, and quercetin, have been reported to suppress significantly DXR activity (IC50 < 5 μM) using DMSO as a surfactant. Their inhibitory activity against DXR, however, substantially decreased when the nonionic detergent Triton X-100 was used to enable the flavonoids to be soluble. These compounds are, therefore, referred to as nonspecific inhibitors of DXR. 28 In effect, Triton X-100 is a frequently used dispersing reagent to differentiate between specific and nonspecific inhibition.28–30 Hence, we further determined the DXR inhibitory activity of the proanthocyanidins employing 0.01% Triton X-100 as a surfactant to verify whether they were authentic inhibitors of DXR. First, the effect of the detergent on DXR was measured, and the results showed that it did not affect the activity of DXR at 0.1%. Subsequently, two concentrations for each of the five compounds were employed to assess their inhibitory activity against DXR, with one concentration being around its IC50 value and the other being approximately half of that concentration. The results are presented in Table 2. None of the proanthocyanidins showed a noticeable DXR inhibitory effect either in the absence or presence of the surfactant at both the concentrations. We also analyzed DXR inhibition of the flavonoid compound baicalein, which has been characterized as a nonspecific inhibitor of DXR 28 ; the data clearly indicated that the compound exhibits strong DXR inhibition in the absence of Triton X-100, but 6 to 7 times lower activity in the presence of 0.01% of the detergent. Based on these data and according to the criteria set up by Shoichet and co-authors, 31 we deduced that the proanthocyanidins tested in this study might be specific inhibitors of the enzyme, DXR.
Influence of 0.01% Triton X-100 (Triton) on the Inhibition of E. coli 1-Deoxy-D-Xylulose 5-Phosphate Reductoisomerase (DXR) Using five Proanthocyanidins and Baicalein as a Positive Control.
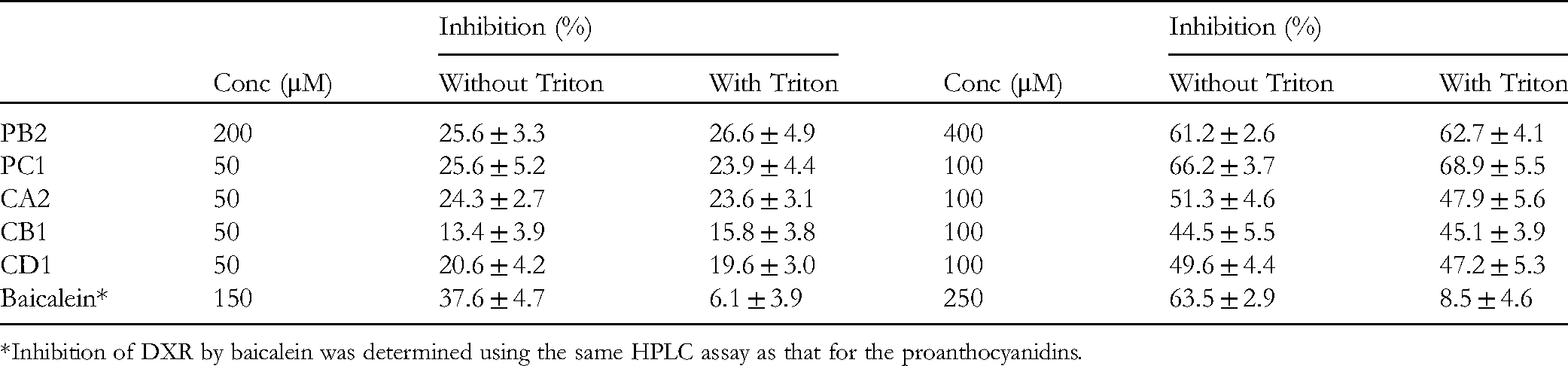
*Inhibition of DXR by baicalein was determined using the same HPLC assay as that for the proanthocyanidins.
Analysis of Particle Size by DLS
To confirm further that the proanthocyanidins indeed suppress DXR activity in a specific manner, we analyzed whether the presence of these compounds could induce aggregation of DXR by measuring the particle size of the assay mixtures using the DLS method.31,32 The proanthocyanidins were diluted to final concentrations of around twice their IC50 values in 50 mM Tris-HCl buffer (pH 7.0) with or without the addition of DXR (4 μg/mL). The results presented in Table 3 indicate that no obvious particles were detectable in all the determinations concerning the proanthocyanidins; however, a well-defined aggregate was observed in the baicalein (positive control) plus DXR mixture. These data strongly support the conclusion drawn from the Triton X-100 assay that proanthocyanidins specifically suppress the activity of DXR.
DLS Analysis of the Mixtures of Proanthocyanidins With/Without DXR in 50 mM Tris-HCl Buffer (pH 7.0).

In a final dilution of DMSO of 2% (v/v); Blank: 2% DMSO (v/v).
N/A: not available.
Used as a positive control.
Measurement of Inhibition Kinetics of PC1 and CD1 Against DXR
To uncover the modes of inhibition of the proanthocyanidins against DXR, PC1, and CD1 were chosen to investigate their inhibition kinetics employing fixed inhibitor concentrations and at different DXP/NADPH concentrations using the HPLC protocol described above. Lineweaver-Burk graphical charts were acquired by plotting the reciprocal of the reaction velocity of the compounds against the reciprocal of the concentration of DXP/NADPH. The results (Figure 6) revealed that both the compounds show uncompetitive inhibition of DXR against NADPH and also acted as noncompetitive inhibitors of DXR against DXP.

Lineweaver–Burk plots of DXR with respect to NADPH/DXP in the absence and presence of the inhibitors. (A) NADPH was used as a variable substrate (final conc. 0.05-0.275 mM) and either PC1 (A-1) or CD1 (A-2) as an inhibitor; (B) DXP was used as a variable substrate (final conc. 0.075-0.3375 mM) and either PC1 (B-1) or CD1 (B-2) as an inhibitor. Reaction mixtures comprised 50 mM Tris-HCl (pH 7.4), 5 mM MgCl2, 2 mM VC, 0 to 30 μM compound PC1 or CD1, and 2 μg/mL DXR in a final volume of 100 μL. The mixtures were incubated for 10 min at 37°C before being treated with CIAP, derivatized with DNPH, and enzyme kinetics determined using HPLC.
Discussion
Proanthocyanidins are polyphenols found in medicinal and edible materials, as well as in many medicinal herbs. Moreover, they are one of the most abundant sources of polyphenolic compounds of plant origin. Many types of bioactivities and broad benefits of this class of components, including their high antimicrobial effects, have been reported on human health.19,23 It has been generally accepted that the bioactivity of proanthocyanidins is closely associated with their unique chemical scaffold, which is characterized by highly hydroxylated moieties constructed from flavan-3-ol blocks. 33 As for the antibacterial activity of this category of compounds, very limited research has revealed that proanthocyanidins inhibit the growth of microorganisms by deteriorating the integrity and permeability of the bacterial cell wall and membrane, crippling protein biosynthesis, and binding to DNA.34,35 This antimicrobial mechanism of action is quite similar to that of the simple phenolic compounds eugenol and carvacrol isolated from essential oils.36,37
Previous studies related to the screening of lipophilic DXR inhibitors have shown that the catechol moiety of a compound might be important for DXR inhibition because its vicinal diol can chelate bivalent metal ions present in the active site of the proteins, preventing the substrate DXP from binding to DXR.12,38,39 As a typical structural feature, proanthocyanidins have at least two catechol moieties in their scaffold (Figure 2) and thus may possess DXR inhibitory activity. Based on this finding and our previous investigations on the inhibitory effects of plant essential oils, catechins, and theaflavins against DXR,13–17 we extended our search for DXR inhibitors to proanthocyanidins. The results suggest that the five polyphenolic compounds used in this study were weak but specific inhibitors of DXR. Further determination of the inhibition kinetics of compounds PC1 and CD1 indicated that both the compounds are uncompetitive inhibitors of DXR with respect to NADPH and noncompetitive inhibitors of the protein with respect to DXP. The modes of action of the two compounds are quite similar to those of theaflavins reported earlier. 16 Perusal of the structure of the compounds allows us to recognize that the DXR inhibitory effect of the proanthocyanidins clearly corresponds to the number of catechol moieties in their scaffolds, namely the polymerization degree (n) of each component. It seems that the presence of three catechol moieties leads to the most potent DXR inhibitory activity, as demonstrated in the cases of compounds PC1, CD1, and CB1; one catechol moiety less, that is, two moieties, could substantially reduce the inhibitory effect (compound PB2), whereas a single catechol moiety could only slightly increase the activity (compound CA2). Similarly, previous studies have uncovered the bioactivities of these types of compounds, such as antioxidant, anticancer, antidiabetic, anti-obesity, and cardioprotective effects, which are closely related to their polymerization degree. 19 Further the comparison of the structures of PC1, CD1, and CB1 gives the impression that the location of the three catechol moieties may also influence their DXR inhibitory activity; when these moieties are on the same side of the molecule (compound PC1) the activity could be better, but the separation of them could slightly decrease their activity (compounds CD1 and CB1).
Conclusion
The inhibitory effect on DXR of five proanthocyanidin compounds was evaluated, and their structure–activity relationship was discussed. The obtained results might be useful to elucidate the antimicrobial mode of action of these compounds; in addition, they should be useful in expanding the application of these compounds as antibacterial agents.
Footnotes
Acknowledgments
This work was supported by the National Natural Science Foundation of China (grant nos. 31800666, 31770848, 31971208) and the Natural Science Basic Research Plan in Shaanxi Province of China (grant no. 2018JM3019).
Author Contributions
XH determined DXR inhibitory activities of the proanthocyanidins; BRZ and LLW prepared the proanthocyanidins; WYG and HL designed the experiments concerning DXR; YML and QJ designed the isolation and elucidation of the proanthocyanidins; WYG, HL, YML, and QJ finished the data analysis and prepared the manuscript. All the authors of the paper have approved the final article and agreed to the submission.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
