Abstract
A new phenolic glycoside, 7-(4-O-β-D-glucopyranosyl, 3-methoxyphenyl)-6,8-dihydrofuro[3′,4′:7,8]naphtho[1,2-d][1,3]dioxol-10(7H)-one, named malconenoside A (
Chaihu is a commonly used traditional Chinese medicine, which was first recorded in the Classic of the Materia Medica (Shen Nong Ben Cao Jing), as top grade. The root of Bupleurum chinense DC. and B. scorzonerifolium Willd. are the major and legal sources of Chaihu recorded in the Pharmacopoeia of People's Republic of China as a key player of many famous prescriptions, such as Xiao Chaihu Tang, Buzhongyiqi Tang, and Xiaoyao Powder. Bupleurum malconense (Zhu ye chai hu) is 1 source of regional supplies of Chaihu in Sichuan province. There are more than 20 Bupleurum species that can be used for medicine, and hundreds of compounds have been identified from them, such as triterpenoid saponins, lignans, and caffeic acid derivatives.1-7 The medicinal part collected for local medicine is the herb, which is quite different from the root for chaihu described by the Chinese Pharmacopoeia. Bupleurum malconense Shan et Y. Li it used to reduce fever and soothe the liver.1,7,8 In this study, we isolated 1 new phenolic lactone (Figure 1) and, herein, we describe its isolation and structure identification.

Chemical structure of compound
Results and Discussion
Compound

COSY ( ) and key HMBC (
) and key HMBC (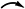 ) and ROESY (
) and ROESY ( ) correlations of
) correlations of
1H (600 MHz) and 13C NMR (150 MHz) Data of

The absolute configuration of

Comparison between the experimental spectrum of
Considering that Bupleurum species are used for the treatment of cancer,4 evaluation against human cancer cells (HepG2, BGC-823, and A549) was conducted to determine any potential beneficial effects of

Cytotoxic effects of
IC50 Values for

HepG2: human liver cancer; BGC-823: human gastric cancer; A549: human lung cancer; HUVEC: normal human umbilical vein endothelial cells.
*P < 0.05 (vs 5-FU group).
Experimental
General
Column chromatography was performed on silica gel (200 − 300 mesh; Qingdao Marine Chemical Inc., China), on C-18 silica gel (40 − 60 μm; Daiso Co., Japan), MCI gel CHP 20P (75 − 150 μm, Mitsubishi Chemical Industries, Tokyo, Japan) and on Sephadex LH-20 (Amersham Pharmacia, Sweden). UV spectra were recorded on a Shimadzu UV-2401PC spectrometer. Semi-preparative HPLC was carried out using an Agilent 1200 liquid chromatograph; the column used was a 250 mm × 9.4 mm, i.d., 5 μm, Zorbax SB-C18 and a 250 mm × 4.6 mm, i.d., 5 μm, Daicel Chiralpak IC. NMR spectra were recorded on a Bruker AV-600 spectrometer, with TMS as an internal standard. EI-MS and HR-EI-MS were recorded using an AutoSpec Premier P776 spectrometer, and ESI-MS and HR-ESI-MS with an API QSTAR Pulsar 1 spectrometer.
Plant Material
B. malconense Shan et Y. Li was collected from Wenchuan (Sichuan Province, China) in September 2013 and authenticated by Prof. Ying-Fang Wei, Chengdu University of TCM. A voucher specimen (cd20130920024) was deposited at Chengdu University of TCM, Chengdu, China.
Extraction and Isolation
The dried and powdered aerial part of B. malconense (10 kg) was soaked in 70% EtOH for 48 h, then percolated. The final extract was evaporated to dryness and extracted with EtOAc. This extract was divided into 7 parts Fr. 1–Fr. 7 by silica gel column chromatography by eluting with a gradient of CHCl3/MeOH (100:1-1:1). Fr. 3 (51 g) was fractionated on a MCI gel CHP 20P column eluting with a gradient of aqueous MeOH (20:80-100:0) to provide eleven portions, Fr. 3.1 − Fr. 3.11. Fr. 3.2 (8.4 g) was separated by Sephadex LH-20 (MeOH) chromatography, followed by passage through a RP-18 column (MeOH/H2O, 40:60-50:50), and by semi-preparative HPLC (MeOH/H2O, 25:75) to give compound
Malconenoside A (1)
Yellowish gum.
[α]D20–13.6 (c 0.022, MeOH); CD (MeOH) Δε202–15.72, Δε237 + 5.84, Δε291 + 2.38.
UV (MeOH) λmax (logε) 285 (2.44), 200 (3.53) nm
1H and 13C NMR: Table 1.
ESIMS m/z 537 [M + Na]+.
HRESIMS m/z 537.1374 [M + Na]+ (calcd for C26H26NaO11, 537.1373).
Acid Hydrolysis of Compound 1
Compound
ECD Calculations
Molecular Merck force field (MMFF) and DFT/TDDFT calculations were performed with the Gaussian09 program package.9 Conflex conformational search generated low-energy conformers within a 10 kcal/mol energy, which was finished by software CONFLEX 7. The predominant conformers were optimized by DFT calculation at the B3LYP/6 to 31G (d, p) level with the PCM in MeOH. ECD calculations were conducted at the B3LYP/6 to 31G (d, p) level with the PCM in MeOH. For comparision of the calculated curves and experimental CD spectra, the program SpecDis 1.62 was used.
Cell Viability
All cell lines were purchased from the Cell Bank of China Science Academy (Shanghai, China) and maintained in Dulbecco's modified Eagle's medium (DMEM) supplemented with 10% fetal bovine serum and 100 U/mL penicillin-streptomycin, and incubated at 37 °C in an atmosphere of 5% CO2. Cell viability was evaluated by a CCK8 assay kit (Dojindo Laboratories, Tokyo, Japan), used according to the manufacturer's instructions. Exponentially growing cells were seeded at 3 to 8 × 103 cells per well in 96-well culture plates for 24 h. Cells were exposed to increasing concentrations (0–80 μM) of
Conclusion
In this paper, one new phenolic glycoside, malconenoside A, was isolated from Bupleurum malconense. It was shown that
Supplemental Material
sj-docx-1-npx-10.1177_1934578X211037410 - Supplemental material for Malconenoside A, a New Phenolic Glycoside from Bupleurum malconense
Supplemental material, sj-docx-1-npx-10.1177_1934578X211037410 for Malconenoside A, a New Phenolic Glycoside from Bupleurum malconense by Jie Yan, Qin Luo, Fei Long and Mei-Lian Zhao in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the National Natural Science Foundation of China (81503200), Application basic research project of the Science and Technology Department of Sichuan Province (2019YJ0333), China Postdoctoral Science Foundation (2020M673566XB), the fund of scientific research innovation team construction in Sichuan Provincial University (18TD0017), and Scientific research promotion plan of “Xinglin Scholars” (QNXZ2018015, CXTD2018016).
Ethical Approval
Not applicable, because this article does not contain any studies with human or animal subjects.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.
Trial Registration
Not applicable, because this article does not contain any clinical trials.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
