Abstract
Among the large number of plants that are part of the Ayurvedic system of medicine in India and Bangladesh, Zingiber officinale Roscoe (Zingiberaceae), or ginger in English, holds a special place and is often referred to as “Mahaushadha” (great medicine) and “Vishvabhesaja” (worldwide or universal herb) to signify its special status. The plant and particularly its rhizomes are used both in the raw and dry form for the relief of a multitude of disorders. Since a number of these disorders occur in patients infected with severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), it was of interest to perform in silico studies (molecular docking) to evaluate the binding affinities of a number of constituents of Zingiber officinale with the 3C-like protease or main protease (Mpro) of SARS-CoV-2, which plays an essential role in the cleavage of viral polyproteins and subsequent viral replication. Our studies indicated that 2 of the compounds present in ginger, namely, chlorogenic acid and hesperidin, had high binding affinities for Mpro with predicted binding energies of −7.5 and −8.3 kcal/mol. The two-dimensional and three-dimensional interactions also showed that, while chlorogenic acid interacts with one of the His41 amino acids of the catalytic dyad of Mpro, hesperidin interacts with the other amino acid Cys145, which can account for their predicted high binding energies and, therefore, possibly can inhibit Mpro activity. Taken together, our findings indicate that ginger, besides alleviating the symptoms induced by SARS-CoV-2, may also play a role in inhibiting the virus.
Introduction
Ayurveda is one of the traditional systems of medicine, dating from around 5000 years ago, and which still persists in the modern era because of its accuracy in mentioning the healing power of various herbs. The Sanskrit word Ayurveda means the “science of life” and the herbal information of Ayurveda can be found initially in the Vedas, the 4 sacred books being Rig, Sama, Yajur, and Atharva Veda. The oldest among them, the Rig Veda mentions 67 plants, while the Yajur and Atharva Vedas mention 81 and 293 medicinal plants, respectively. 1 The Vedic medicinal literature was further augmented by 2 of the most famous Ayurvedic practitioners, namely Charaka and Sushruta; Charaka compiled a list of medicinal plants for general use (Charaksamhita) and Sushruta compiled a list of plants, procedures, and instruments used in a surgery known as Sushruta Samhita. Together with Ashtang Sangraha, the 3 Ayurvedic treatises are collectively known as Brihattrayee or The Big Three. 2 The 3 main treatises were supplemented in later days by “nighantus” or pharmacopeias detailing various mono and polyherbal formulations.
The Ayurvedic literature is full of mentions of Zingiber officinale Roscoe (Zingiberaceae), known in English as ginger, as a medicinal plant of the highest importance, so much so that it is often referred to in Ayurveda as “Mahaushadha” (great medicine) and “Vishvabhesaja” (world or universal herb) to signify its special status. Although originating in India, various species of ginger are grown around the world at present. Sandeep 3 compiled a list of these various types of ginger and they are listed according to the original list of Sandeep 3 (Table 1). It can be seen from the list that all gingers are not various cultivars of Zingiber officinale, but may belong to even a separate genus. Although the different varieties offer important scope for study of this much-used and much-valued medicinal herb and culinary spice, unfortunately, there does not appear that any concerted effects have been made, even in India, to study the various phytoconstituents of different country-wide ginger species and correlate them with any reported pharmacological activities or ethnic uses. Such correlation could have brought forth the traditional uses of ginger along with modern findings and paved the route towards possible use of the plant and/or plant parts as novel therapeutic(s) for other diseases than the ones reported in the traditional medicinal literature.
Ginger Varieties Cultivated Throughout the World.

Ayurveda recognizes 2 types of ginger, fresh rhizomes, and dried rhizomes. The former is known as “adraka” or “ardraka,” the latter as “sunthee” or “sunthi.” The therapeutic properties are thought to be different in some aspects, but similar in other aspects. Overall, a dry ginger rhizome is considered better and more potent than fresh rhizomes. The Bhavprakash Nighantu (nighantu) written by the famous Ayurvedic physician Acharya Bhavprakash (Acharya being a term of honor for a learned person) mentions that sunthi is beneficial for rheumatoid arthritis, vomiting, colic, heart disease, filaria, piles, cough, respiratory disease, and gastrointestinal (GI) disorders. The nighantu further mentions taking slices of fresh ginger with salt (adraka-lavana yoga) before meals for treatment of dyspepsia and indigestion. According to the nighantu, adraka was indicated for skin diseases, anemia, dysuria, wound, ulcer, bleeding disorders (raktapitta or blood coming out of the various orifices of the body in the absence of external injury, but due to some internal cause), and burning sensations. 4
The present pandemic sweeping the world has been named coronavirus disease 2019 (COVID-19) because it is caused by the coronavirus designated severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) and was first reported from Wuhan in China at the tail end of December 2019. The virus has the ability to spread quickly and, in a little more than a year, has been the cause of 126,683,142 infections and 2,778,889 deaths throughout the world (as of March 27, 2021) [https://www.worldometers.info/coronavirus/]. The worst sufferer has been the United States of America with 30,853,032 cases and 561,142 deaths. According to the US Census Bureau, the US population numbered around 331 million at the start of the year 2020. Assuming no major changes in the US population, the numbers suggest that about 1 in 10 people has been infected with the virus in the United States of America, the most scientifically advanced country in the world. The disease, like its 2 predecessor coronavirus diseases SARS-CoV and the Middle East respiratory syndrome (MERS-CoV), cannot be cured with any drugs at present, although antivirals like remdesivir, lopinavir, and ritonavir have been tried with, at the most, limited success. COVID-19 symptomatic treatments include azithromycin, dexamethasone, colchicine, and bronchodilators, fibrinolytics, and anticoagulants. 5 Several vaccines have obtained emergency approvals and vaccinations have started, but the vaccination program and vaccines have faced problems. Most vaccines need 2 doses to be administered, which, considering the world population of around 8 billion, will necessitate at least 16 billion doses. Moreover, vaccines developed by Pfizer-BioNtech and Moderna need a temperature of −70 °C for storage, which is a difficult condition for developing countries. On top of it, the AstraZeneca vaccine developed by AstraZeneca-Oxford is yet to obtain US approval. It is too early to know about the long-term effects of vaccines, whether they need to be given yearly or at what intervals, and whether vaccines will be equally effective against the 3 present variants of SARS-CoV-2, namely the United Kingdom, and variants from South Africa and Brazil. A new mutant of SARS-CoV-2 and termed by World Health Organization as “virus of concern” with unknown properties has been reported from India on March 25, 2021 (www.bbc.com/news/world-asia-india-56507988), which has currently turned India into the epicenter of COVID-19 with hundreds of thousands of dead and millions more infected. As a result, patients infected with SARS-CoV-2 are essentially getting symptomatic treatment, which may necessitate a cocktail of drugs depending on the symptoms, and each drug has its own adverse effects.
COVID-19 symptoms can range from mild to severe. The symptoms may appear after 2 to 14 days following exposure to the virus. 6 Fever, cough, and shortness of breath (difficulty breathing) are the most common symptoms; other typical and atypical symptoms that have been observed during the initial phases of infection include tiredness, rhinorrhea, stuffy nose, myalgias, sore throat, headache, diarrhea, lack of appetite, abdominal pain, vomiting, loss of smell or taste, conjunctivitis, and skin rash.6,7 Progression to more severe disease may include pneumonia, respiratory failure, multiple organ dysfunction, severe cardiac impairment, and death. The binding of SARS-CoV-2 to its receptor angiotensin-converting enzyme 2 (ACE2) can bring the virus to cardiovascular cells and tissues leading to a host of cardiovascular disorders like heart failure, heart attack, blood clots, and arrhythmias, to name only a few.6,8
The various Ayurvedic uses of the Z. officinale rhizome (both dry and raw forms) and which relate to the symptoms of COVID-19 include enhancement of appetite, clearing the throat, alleviating cough, acting as a pain killer, antipyretic, asthmatic conditions, vomiting, stomach ache, heart disorders, lethargy and physical weakness, and headaches. 9 It is clearly evident that the traditional uses of ginger are proven to be correct, can go a long way in alleviating various symptoms associated with COVID-19. A substantial body of scientific evidence is accumulating that the traditional therapeutic uses of ginger are to a large extent, validated by modern scientific methods, and there are a number of reviews compiling the pharmacological and clinical data.9,10 Besides the traditional uses mentioned, other properties of ginger include anti-inflammatory (like in rheumatoid arthritis), hypoglycemic, cancer-preventive, treatment for breast and ovarian cancer, mosquito larvicidal, antioxidant, and antibacterial.11-17
The rhizome and its essential oil are rich in phytochemicals. Some of these present in essential oil and rhizomes include (−)-zingiberene, (+)-curcumene, (−)-β-sesquiphellandrene, β-bisabolene, eudesmol, γ-terpinene, alloaromadendrene, α-pinene, δ-cadinene, elemol, farnesal, E-β-farnesene, neril acetate, β-myrcene, cineol, citral, cineole, geranyl acetate, terpineol, borneol, geraniol, limonene, linalool, and 5-hydroxy-1-(4-hydroxy-3-methoxy phenyl) decan-3-one. 18 Detailed phytochemical-pharmacological reviews can be found in Gupta and Sharma, 19 Ashraf et al, 20 and Singh et al. 21
That ginger and some of its constituents also have antiviral properties have been reported in several studies. Aqueous extract of Z. officinale demonstrated inhibitory activity against the chikungunya virus. 22 It has been reported that the hot water extract of fresh, but not dried, ginger rhizomes inhibits human respiratory syncytial virus-induced plaque formation in Hep-2 and A549. 23 Gingerenone A, a constituent of ginger rhizome, reportedly suppressed the replication of 3 influenza A virus (IAV) subtypes (H1N1, H5N1, H9N2) in 4 different cell lines, namely Madin–Darby canine kidney, A549 (a human lung cancer cell line), 293T (a human embryonic kidney cell line), and DF1 (a chicken fibroblast cell line), as well as in the lungs of IAV-infected mice. 24
Considering that the COVID-19 situation is again getting critical, even after the introduction of new vaccines, and that phytochemicals have been shown in silico studies to have strong binding and possibly inhibitory activity against SARS-CoV-2,25-27 it was of interest to conduct molecular docking studies with chemicals from Z. officinale and the main protease or Mpro of SARS-CoV-2. Mpro, also known as a chymotrypsin-like cysteine protease (or 3CLpro), plays a vital role in SARS-CoV-2 gene expression and replication. As such, the protease Mpro is considered by scientists to be an attractive target for drug designing and in silico binding studies of potential drugs. 28
Methodology
Protease (Mpro)
We have used the pdb file (6LU7) of the main protease of SARS-CoV-2, 3C-like protease or Mpro, as published before. 29 In the pdb file (6LU7), the protease was bound to an inhibitor N3, which was removed from the pdb file prior to using the protease structure in our molecular docking studies. It is to be noted that the interacting residues of the inhibitor N3 with the protease amino acids included His41, Met49, Phe140, Leu141, Asn142, Gly143, His163, His164, Glu166, Leu167, Pro168, Gln189, Thr190, and Ala191. The catalytic site active residues of SARS-CoV-2 Mpro are His41 and Cys145. 29 Mpro exists as a dimer; the monomeric form of Mpro was used for molecular docking studies.
Compounds Used in Docking Studies
Thirty phytochemicals present in Z. officinale were studied based on random selections of the phytochemicals. Ligand molecules were downloaded from Pubchem in sdf format. 30 They were optimized with the force field type MMFF94 using OpenTable software and saved as pdbqt format. Besides the 30 phytochemicals of Z officinale, we have also used the experimental antiviral compound lopinavir as a control. Lopinavir is an antiviral drug currently under investigation in various clinical trials against COVID-19. 31 The drug has been used previously to treat severe acute respiratory syndrome (SARS) and the Middle East respiratory syndrome and found to be an effective inhibitor of viral main protease. 32
Molecular Docking
With different proteins and different ligands or even with the same protein but different ligands, various molecular docking programs have been reported to be used, depending possibly on the availability and greater preference of 1 program over another by the authors. For instance, in a study with acetylcholinesterase, ligands were docked within the crystallographic structure of the enzyme using Molegro Virtual Docker software. 33 Molegro Virtual Docker is able to predict the most likely conformation of a ligand's binding to a macromolecule and has also been used for molecular docking studies with human butyryl cholinesterase. 34
Binding studies were carried out through blind molecular docking using AutoDock Vina. 35 Blind molecular docking enables the researcher to search throughout the entire surface of the macromolecule for binding sites of the experimental ligands. We selected the blind molecular docking process (although the active sites for Mpro are so far known to be in domains 1 and 2), for that enabled us also to look for any possible binding of the ligands to domain 3 of Mpro, which is involved in the dimerization and catalytic activation of the protease.36,37 Blind docking was the approach used to identify potential inhibitors of SARS-CoV-2 Mpro through molecular docking, even in a recent study published in 2021. 38
Polar hydrogen atoms were added in this study because crystallographic structures usually lack hydrogen atoms. The addition of polar hydrogen atoms and removal of water molecules and N3 was achieved with PyMOL software. 39 The grid box in AutoDock Vina was generated aiming to cover up all the key residues for ligand binding of the main protease, where the center was at X: −6.35, Y: 8.36, Z: 41.26, and the dimensions of the grid box were, X: 38.84, Y: 38.77, and Z: 97.25 (unit of the dimensions, Å). We have used exhaustiveness “16” for better ligand and protein binding. The predicted binding affinity values are an average of 5 independent runs of the docking program. Docked poses of selected phytochemicals bound to SARS-CoV-2 main protease (Mpro) were obtained from PyMOL and displayed in Discovery Studio. 40
Lipinski’s Rule of 5
Lipinski's rule of 541,42 was followed to determine the drug-like properties of the constituents of Z. officinale in the present work. This rule proposes that molecules with poor permeation and oral absorption violate 2 or more of the following properties, namely molecular weight >500, ClogP > 5, more than 5 hydrogen bond (HB) donors, more than 10 acceptor groups, and molar refractivity outside a range of 40 to 130.
Biological Activity Prediction
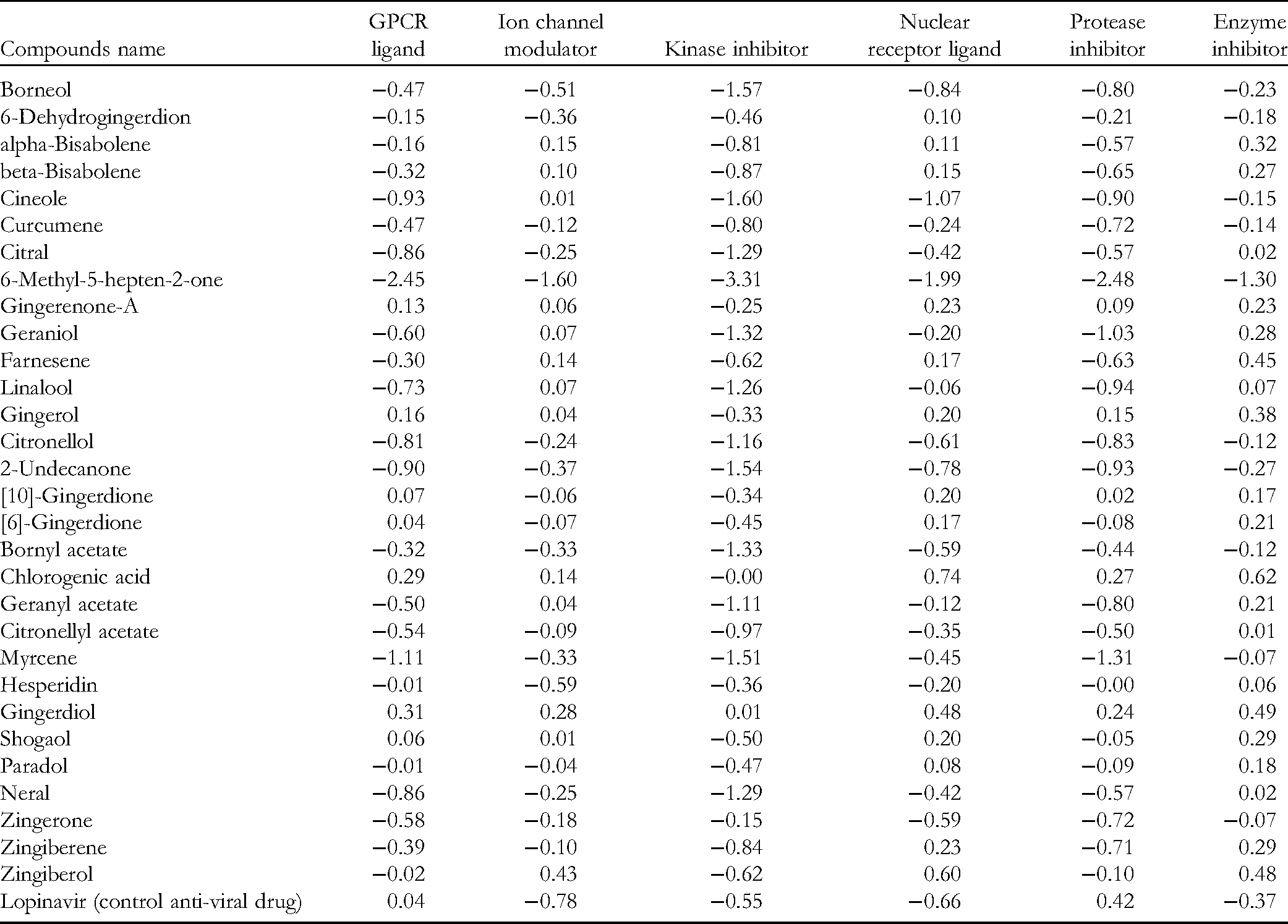
The phytochemicals were assessed for potential bioactivity by calculating their activity scores as G-protein coupled receptor ligands, ion channel modulators, kinase inhibitors, nuclear receptor inhibitors, protease inhibitors, and enzyme inhibitors. The various parameters were evaluated with the aid of the software Molinspiration (www.molinspiration.com, Nova Ulica, Slovensky Grob, Slovak Republic). 43
Results
The structures of the 30 phytochemicals studied are shown in Figure 1. Their predicted binding energies, along with that of the antiviral drug lopinavir (used as a control compound), are shown in Table 2. The least binding energies (ie higher binding affinities) were demonstrated by 2 compounds, namely chlorogenic acid (CA) and hesperidin, with predicted binding energies at − 7.5 and − 8.3 kcal/mol, respectively. The control antiviral drug lopinavir gave predicted binding energy of −8.2 kcal/mol. From the binding energy viewpoint, the flavanone glycoside hesperidin demonstrated a greater affinity for Mpro than the cinnamate ester CA. However, lopinavir showed a better binding affinity than CA. Other studies have also found that lopinavir exhibited inhibitory effects against Mpro. 44

Structure of Zingiber officinale phytochemicals in the present study.
Predicted Binding Energies of Zingiber officinale Phytochemicals to Mpro of SARS-CoV-2.

Abbreviations: Mpro, main protease; SARS-CoV-2, severe acute respiratory syndrome coronavirus 2.
The physicochemical properties of the Z. officinale constituents and lopinavir are shown in Table 3. Lipinski’ rule of 5 states that molecules which are poorly absorbed by the intestinal wall (ie oral bioavailability is not good) should present 2 or more of these characteristics: molecular weight more than 500, lipophilicity (logP > 5), HB donor groups (expressed as the sum of OHs and NHs groups) more than 5, more than 10 HB acceptor groups (expressed as the sum of O and N atoms), and molar refractivity outside a range of 40 to 130.41,42 These are considered violations of Lipinski's rule of 5, which is considered a ‘rule of thumb’ and does not totally eliminate a candidate drug to be effective if some rules are violated.
Physicochemical Properties of Zingiber officinale Phytochemicals and the Antiviral Drug Lopinavir.

Table 3 shows that the hesperidin violated 3 of the 5 rules of Lipinski; CA violated 1 and lopinavir 2, which may imply that lopinavir, is a better drug than hesperidin. It should be noted, however, that common drugs, like atorvastatin and montelukast violate Lipinski's rules in more than 1 way, but are considered effective drugs. 41 A study found that in 2007, out of the top-selling 60 drugs, 7 did not fulfill the rules. 41 Moreover, if a drug cannot be given orally for any reason, there are parenteral means to overcome the problem. Another negative factor for lopinavir is that it needs to be co-administered with ritonavir to obtain the desired anti-COVID-19 effects. Lopinavir–ritonavir should not be used in patients with renal impairment, hepatic impairment, hemophilia, pancreatitis, immune reconstitution inflammatory syndrome, and osteonecrosis among others, according to the European Medicines Agency (Kaletra, INN-lopinavir, ritonavir—European Medicines Agency, Annex 1, Summary of Product Characteristics).
Biological activity calculations of the Z .officinale constituents, with the help of Molinspiration software, are shown in Table 4. The pharmacokinetic data of CA and hesperidin are shown in Table 5, and, as predicted from Lipinski's rules, violation number in Table 2, have low GI absorption. Low GI absorption of drugs can be increased through crystalline solid, amorphous, and lipid formulations, as reviewed by Gupta et al. 45 The authors also mention the emerging strategy of self-emulsifying drug delivery systems (SEDDSs), which can include self-emulsifying, self-microemulsifying, and self-nanoemulsifying drug delivery systems to enhance the bioavailability of drugs with low GI absorption.
Biological Activity Prediction of Selected Phytochemicals of Zingiber officinale.
Pharmacokinetic Data of Chlorogenic Acid, Hesperidin, and Lopinavir.

The two-dimensional (2D) and three-dimensional (3D) interactions of CA, hesperidin, and gingerenone-A with Mpro are shown in Figures 2 to 4, respectively. Only these 3 compounds were chosen to be presented, CA, and hesperidin because of their high binding affinities for Mpro, and gingerenone-A for its moderate affinity for M-pro (predicted binding energy at −6.4 kcal/mol). Mpro of SARS-CoV-2 is catalytically active as a dimer. The monomeric unit consists of 3 domains; domain I consists of amino acid residues 8 to 101, domain II amino acid residues 102 to 184, and domain III amino acid residues 201 to 306, as reported by Zhang et al. 46 Domain III does not directly participate in interacting with the substrate; it is involved in regulating dimerization of Mpro, 36 and dimerization is necessary for the protease to be catalytically active. 37 A catalytic dyad is formed in Mpro of SARS-CoV-2 by Cys145 and His41.

2D and 3D interactions of chlorogenic acid with Mpro.

2D and 3D interactions of hesperidin with Mpro.

2D and 3D interactions of gingerenone-A with Mpro.
Figure 2 shows that the chlorogenic acid interacts with amino acid residues Thr25, Thr26, His41, Leu141, Gly143, His163, and Glu166 of Mpro. The results indicate that chlorogenic acid interacts with both domains 1 and 2 amino acids, including 1 of the 2 amino acids forming the catalytic dyad, His41. The interacting amino acids of Mpro with the irreversible inhibitor N3 29 are His41, Met49, Phe140, Leu141, Asn142, Gly143, His163, His164, Glu166, Leu167, Pro168, Gln189, Thr190, and Ala191. Thus, of the 14 amino acids of Mpro interacting with N3, 5 interact with CA, including the important catalytic dyad amino acid, His41. The hydrophobic bonds that CA forms with both domains 1 and 2 amino acids of Mpro possibly contribute to the predicted high binding affinity.
Unlike CA, hesperidin was found to interact only with domain 2 amino acid residues of Mpro. These amino acids included Phe140, Asn142, Cys145 (1 of the 2 amino acids forming the catalytic dyad), His163, and Met165. The combination of hydrophobic bonding with Phe140 and Met 165, along with interaction with Cys145, would create a favorable situation of blocking the entry of any other compound to the catalytic site and possibly accounts for the predicted higher binding affinity observed with hesperidin. Gingerenone-A interacted with Thr24, Met49, Gly143, Met165, and Glu166. This phytochemical did not interact with the 2 amino acids His41 and Cys145 of the Mpro catalytic dyad, but did interact in common with the inhibitor N3 interacting amino acids, Met49, Gly143, and Glu166.
Discussion
The results indicate that 2 constituents of Z. officinale (among the 30 screened), namely chlorogenic acid (CA) and hesperidin, demonstrated high predicted binding affinities and bind to 1 of the 2 catalytic dyad amino acids of Mpro. The antiviral effects of CA and hesperidin have been previously reported. Lonicera japonica Thunb., which is rich in CA, is used in China to treat influenza virus, parainfluenza virus, and respiratory syncytial virus. 47 Anti-hepatitis B virus activity of CA has also been shown in vitro and in vivo experiments by Wang et al. 48 CA also inhibited the replication and viability of enterovirus 71 in vitro. 49 During exploratory molecular docking studies for active compounds in traditional Mongolian medicine for anti-COVID-19 drugs, CA was found to inhibit effectively the binding of S protein of SARS-CoV-2 with its receptor ACE2. 50 In silico studies have shown that CA is a potential inhibitor of COVID-19 with multiple targets including ACE receptor. 51
Hesperidin has been shown to demonstrate antiinfluenza virus A activity, enhancing cell-autonomous immunity. 52 Against the hepatitis B virus, hesperidin was only moderately effective. 53 Hesperidin has been mentioned as a prophylaxis and treatment for COVID-19. The compound has been shown to block the entry of SARS-CoV-2 through its ACE2 receptors. The compound can boost host cellular immunity and its anti-inflammatory action can inhibit the SARS-CoV-2-induced cytokine storm. When co-administered with diosmin and heparin, the combination can protect against venous thromboembolism, which in turn may prevent disease progression. 54 In various in silico studies, hesperidin has been shown to have low predicted binding energies to both Mpro and spike protein (S) of SARS-CoV-2, lower than that of lopinavir, ritonavir, and indinavir. 55 Our results for hesperidin and lopinavir binding energies to Mpro are in agreement with the above study. Gingerenone-A, because of its ability to inhibit JAK2 (Janus kinase 2), which kinase is a crucial factor in IAV replication, has been mentioned as a potential IAV therapeutic. 24 In molecular docking simulation using Hex software, gingerenone-A showed best binding affinities to Mpro and S protein compared to other phytochemicals of Z .officinale. 56
Our results suggest that CA and hesperidin can prove to be good therapeutics for COVID-19. However, as discussed earlier, to be an effective drug, there remains a question of absorption and bioavailability. The bioavailability of CA in rats has been shown to depend on the gut microflora. 57 However, CA has also been reported to be quickly absorbed from rat stomach in its intact form. 58 A further study has reported that CA is absorbed and hydrolyzed in the small intestine and its bioavailability in rats depends largely on its uptake into the gut mucosa. 59
In mice, it has been reported that sulfates of hesperidin can be absorbed by the duodenum and ileum, deglycosylated metabolites could be absorbed by cecum, and phenolic acids absorbed by the cecum and colon. 60 In Sprague–Dawley rats, Nectoux et al 61 reported that following a single oral administration of hesperidin (hesperetin-7-O-rutinoside), glucuronized hesperidin (but not hesperidin) was detected in the circulating bloodstream; it was indicated that hesperidin or hesperetin was possibly methylated and demethylated during the intestinal membrane transport process; the authors concluded that hesperidin can enter into the circulating bloodstream in its conjugated forms.
We would like to point out that dozens of papers involving mostly in silico studies either have been or are being conducted for finding a plant-based compound with the potential to be either a lead compound or an approved marketable therapeutic against COVID-19. Thus far, to the best of our knowledge, there are no reports of such a therapeutic being discovered, despite numerous leads from in silico studies on phytochemicals. However, we would like to draw attention to the fact that compounds derived from marine-based invertebrates are also being investigated for their potential role in the inhibition of Mpro. In an in silico study conducted with 227 terpene natural products isolated from Red Sea invertebrates, erylosides B was identified as a more potent inhibitor of Mpro than lopinavir. 62 Another study has identified 17 potential Mpro inhibitors in molecular docking analysis of natural substances of marine origin. 63 Natural-like products have also been studied in silico for inhibition of Mpro. 64
Hesperidin (a flavanone glycoside) is not the only flavonoid group compound to be shown in silico studies as a potential Mpro inhibitor. Flavonoids, especially rutin, have been shown to be one of the most potent inhibitors of Mpro.65,66 In silico and in vitro studies have also shown the potential of the dietary flavonoid quercetin as an inhibitor of various targets in SARS-CoV-2, including Mpro. 67 Various flavonoids with therapeutic potential against COVID-19 have been reviewed. 68 In another study on the identification of potential SARS-CoV-2 inhibitors from spices, salvianolic acid from Salvia officinalis (sage) and curcumin from Curcuma longa (turmeric) have been shown to be highly effective inhibitors of Mpro. 69
Conclusion
Two compounds present in Z. officinale rhizomes, namely CA and hesperidin, demonstrated predicted low binding energies (ie high binding affinities) with Mpro. The 2D and 3D figures show that both compounds interact with at least 1 amino acid of the catalytic dyad of Mpro, making them possible inhibitors, and as such, of therapeutic value for inhibition of SARS-CoV-2 viral replication. The limitation of this study is the absence of wet lab studies that are studied in vivo on the virucidal activity of the compounds or use as therapeutics for COVID-19, the last required clinical trials. On the other hand, the strong point of the present study is to demonstrate the value of Ayurveda in systematic choice of plants, which can be at the preliminary stage, selected for in silico studies of interactions between the plant's constituents and different targets of SARS-CoV-2, like Mpro or the S protein. Two other points need to be emphasized here. First, ginger or the rhizomes of Z.officinale itself can be used in the raw or dry form to alleviate many of the symptoms exhibited by COVID-19 patients (these uses of ginger can be found in Ayurvedic texts, as well as confirmed in modern pharmacological studies). The second point is that ginger has been experimentally seen to be beneficial in diabetes, hypertension, and cardiovascular disorders.70-72 It has been found that COVID-19 patients who have diabetes or hypertension as comorbidity are more prone to develop a more severe course and disease progression. Elderly patients with these comorbidities are increasingly likely to enter the intensive care units of hospitals and have a higher mortality rate. 73 In a meta-analysis, which included 1576 COVID-19 patients, the prevalence of diabetes, hypertension, and cardiovascular disorders were, respectively, 9.7%, 21.1%, and 8.4%. 74 Taken together, Z. officinale and its constituents may be beneficial to inhibit SARS-CoV-2 replication, alleviate COVID-19 symptoms, and also have a beneficial effect on COVID-19 patients-associated comorbidities.
Footnotes
Acknowledgments
This work was funded solely by the authors. MLP thanks to Project CICECO-Aveiro Institute of Materials, UIDB/50011/2020 and UIDP/50011/2020, national funds by the Portuguese Foundation for Science and Technology/MCTES.
Author Contributions
RJ, MR, VN, MLP, CW, PW conceptualized and guided the work. MR wrote the first draft of the manuscript. AKP, AH, KJ, TAB, and TM did the in silico studies and cross-checked the references. All authors took part in editing and correcting the first draft. The final draft was approved by all authors.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
