Abstract
Iron toxicity in iron-overloaded conditions, including high iron diet and blood transfusion, causes deleterious effects on vital organs. There currently are a number of chemical chelators in clinics to reduce iron concentration, for example, deferoxamine and deferiprone, but these produce diverse side effects. Hence, the need for a safe and effective iron chelator is demanded. To evaluate rigorously the potential of berberine on iron chelation and its anti-oxidant effect, 30 mice were divided into 5 groups of 6. Except for the control group, other groups received iron sucrose 5 times a week for 4 successive weeks as an i.p injection. Afterward, either berberine or deferoxamine was injected for 1 month. The mice were then euthanized and liver, kidney and lungs were carefully removed for biochemical and pathological analysis. In comparison with the iron group with an extraordinary amount of iron deposits, berberine (20 mg/kg/day) dramatically reduced iron sedimentation in all tissues (P < 0.01). Moreover, berberine lowered clinical symptoms of iron overdose, including inflammation, fibrosis and tissue degeneration. In terms of the activity of antioxidant enzymes, catalase and superoxide dismutase, iron overdose greatly reduced their activity compared to the control group. Berberine progressively increased their activity in comparison with the controls by lowering oxidative conditions (P < 0.05). Iron overdose similarly increased lipid peroxidation by increasing the level of malondialdehyde. Berberine promptly suppressed lipid peroxidation in an efficient manner and reduced the level of malondialdehyde, a marker of lipid peroxidation in the tissues. Accordingly, berberine, as a natural antioxidant compound, could adequately serve as a substitute for chemical chelators with fewer side effects and comparable effectiveness.
Iron is an essential element involved in the processes of oxygen transport, electron transport in the cellular respiration chain, and DNA synthesis. Excess iron and its accumulation are toxic to living organisms and lead to damage to tissues such as the liver, lung and kidneys. 1 -3 In some diseases, such as inherited hemochromatosis, thalassemia and hemolytic anemia, patients need blood transfusions, which can lead to iron overload. In addition, foods with excessive amounts of iron, like meat and fish, increase iron in the body. 1,4 The human body has the ability to excrete one milligram of iron per day, while a unit of blood enters 250 milligrams of iron into the body of patients. In the absence of iron chelators, 5 grams of excess iron accumulate in the body of these patients annually. 5 Because the human body is unable to excrete excess iron, iron overload eventually leads to serious injuries and death of these patients. Indeed, our body does not regulate iron storage and disposal, except through selective absorption in the intestine. 6 Imbalance in iron uptake and excretion leads to iron overload and consequent increase in unbound iron to transferrin in the plasma. This introduces the production of hydroxyl radicals and the emergence of reactive oxygen species (ROS). Studies have shown that oxidative stress leads to damage to large organs, especially the cardiovascular system and liver tissue. 7,8 ROS and reactive nitrogen species (RNS) are produced during cellular metabolism in mitochondrial respiration, as well as in the presence of oxidizing agents, heavy metals and hypoxia. In the event of an imbalance, oxidative stress can damage biomolecules such as DNA, proteins, and lipid membranes. 9,10
Nowadays, synthetic iron chelators such as deferoxamine (DFO; Figure 1(A)) and deferiprone are used to chelate iron. 11 These drugs exert many side effects, including patient irritation, and systemic allergic reactions, like rash, urticaria, anaphylactic response and angioedema. 12,13 This highlights the need for research on new chelators. Therefore, today, researchers have focused their attention on organic compounds. Studies have shown that flavonoids, coumarins, chromones, anthocyanins and chalcones possess the chelating power of heavy metals. 14,15 Thus, they can chelate intermediates such as iron in diseases like Parkinson’s, Huntington’s, Alzheimer’s and thalassemia. Berberine (Figure 1(B)) is an alkaloid found in the roots, stems and bark of plants such as Berberis (Berberis vulgaris). Therapeutically, berberine has long been used as an antibacterial compound in traditional medicine, especially in China to treat gastrointestinal infections and diarrhea. 16,17 It is additionally utilized orally to decrease blood sugar and treat diabetes, lower blood fats and pressure. Berberine typically exerts no adverse side effects, but in some cases gastrointestinal upset has been observed with symptoms like diarrhea, bloating, constipation and stomach pain. 18,19 In 2006, Shirwaikar et al. examined the antioxidant and chelating activity of berberine on lipids and antioxidant enzymes in vitro and showed that it possesses a superior ability to chelate iron, as well as reduce the effect of oxidants. 20 In addition, in 2017, Polu and colleagues, by examining the antioxidant effects of Tinospora cordifolia, which contains an extensive amount of berberine, showed that this plant exerts good antioxidant effects and demonstrates an excellent ability to chelate iron in vitro. 21 Zhao and colleagues also demonstrated that berberine, with its strong antioxidant power, possesses neuroprotective properties. 22 Therefore, according to these studies, which represent the chelating and antioxidant activity of berberine, in this study, we investigated the iron chelating and antioxidant potency of berberine in the liver, lung and kidney tissues of iron-overloaded mice in vivo as a suitable alternative to chemical chelators.
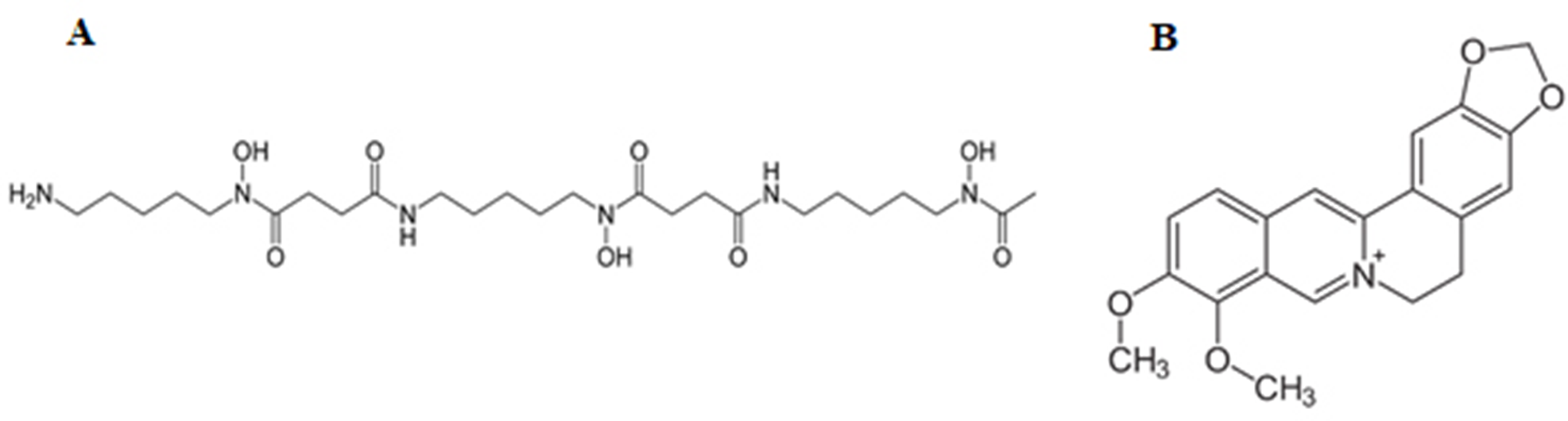
The chemical structures of deferoxamine (
Results
Total Non-Heme Iron in the Tissues
Figure 2(A) shows the amount of total non-heme iron in the liver, lung and kidney tissues. The amount of total non-heme iron in group I (kidney: 757.1 ± 37.0, liver: 2452 ± 2.3, lung: 328.5 ± 4.3 µg/g wet weight) increased significantly compared to group C (kidney: 189.6 ± 0.04; liver: 539.2 ± 34.0; lung: 108.3 ± 0.02 µg/g wet weight) and there was a significant difference, F(4, 5) =359.5, P = 0.001 in kidneys, F(4, 5) =548.4, P = 0.001 in lungs and F(4, 5) =703.5, P = 0.001 in the liver. In group B (kidney: 492.1 ± 10.36, liver: 1048 ± 77.9, lung: 210.9 ± 4.4 µg/g wet weight) and group DI, iron decreased significantly compared to group I. No significant difference was observed between groups B and DI in the liver. The quantity of non-heme iron in kidney of group B decreased significantly compared to DI, which indicates that berberine is more capable of removing iron than DFO in the kidneys.

(A) The total amount of non-heme iron in the tissues; B: the levels of MDA (µM), SOD (U/mL) and CAT activity (U/mL) in the tissues. C: the mice received normal saline; D: the mice received normal saline and then DFO; I: iron-overloaded mice receiving iron sucrose; DI: Iron-overloaded mice receiving DFO; B: Iron-overloaded mice receiving 20 mg/kg/day berberine. (Means ± SD) *<I>P < 0.003, **<I>P < 0.002, ***<I>P < 0.001 compared to control group.
The Level of Lipid Peroxidation
In the results of MDA and lipid peroxidation, iron injection to mice caused a sharp increase in MDA (kidney: 29.4 ± 0.8, liver: 32.6 ± 1.4, lung: 23 ± 2.5 µM; Figure 2(B)) and demonstrated a significant difference with the control group (kidney: 12.6 ± 1.2, liver: 13.1 ± 0.28, lung: 11.3 ± 0.9 µM), F(4, 5) =185.6, P = 0.001 in kidneys, F(4, 5) =27.5, P = 0.001 in lungs and F(4, 5) =197, P = 0.001 in the liver with DFO (Group DI), the MDA level decreased significantly (kidney: 10.2 ± 0.7, liver: 8.7 ± 0.3, lung: 15.15 ± 2.3 µM), but berberine provided a more superior efficiency in reducing MDA than DFO in the liver and kidney tissues (kidney: 10.8 ± 1.4, liver: 9.2 ± 0.6, lung: 7.6 ± 0.6 µM). There was no significant difference between DFO and berberine in reducing the amount of MDA in the lungs.
Activity Rate of Antioxidant Enzymes
As shown in Figure 2-B, increase in iron concentration caused a severe decrease in CAT activity (kidney: 1.31 ± 0.06, liver: 0.91 ± 0.09, lung: 1.07 ± 0.25 U/mL). With the use of berberine (kidney: 3.56 ± 0.63, liver: 2.98 ± 0.38, lung: 2.35 ± 0.13 U/mL), CAT activity was tremendously increased, even more than group D (kidney: 2.95 ± 0.09, liver: 1.58 ± 0.12, lung: 2.08 ± 0.06 U/mL), F(4, 5) =18.92, P = 0.003 in kidneys, F(4, 5) =35.32, P = 0.001 in lungs and F(4, 5) =33.03, P = 0.001 in the liver.
The activity of SOD antioxidant enzyme also decreased significantly with increasing iron concentration (kidney: 21.1 ± 2, liver: 17.6 ± 1.7, lung: 16.6 ± 1.4 U/mL), compared to the control group (kidney: 21.1 ± 2, liver: 17.6 ± 1.72, lung: 16.6 ± 1.4 U/mL). However, administration of berberine (kidney: 39.7 ± 2.9, liver: 56.6 ± 1.9, lung: 35.4 ± 1.8 U/mL) or DFO (kidney: 39.6 ± 1.5, liver: 59.8 ± 0.2, lung: 23.1 ± 1.8 U/mL) remarkably improved SOD activity (Figure 2(B)). Berberine had better efficacy in reducing oxidative conditions in the liver than DFO , F(4, 5) =27, P = 0.001 in kidneys, F(4, 5) =32.3, P = 0.001 in lungs and F(4, 5) =175.2, P = 0.001 in the liver.
Histopathological and Morphological Changes
The results of pathological H&E staining of tissues showed extensive damage due to iron accumulation. Iron sedimentation in the liver caused necrosis, fibrosis, and cirrhosis with inflammation and pigment accumulation (Figure 3). Berberine significantly reduced tissue damage in the liver, but inflammation and pigment buildup were still visible, dissimilar to the control group. Inflammation and accumulation of pigment in the liver could also be seen in the DI group. Excess iron causes necrosis, inflammation, and pigment accumulation in the kidneys and the adipose tissue around them, accompanied by glomerular injuries. However, the use of either berberine or DFO markedly reduced the amount of damage in kidneys. As expected, iron accumulation and the resulting damage in the lungs were lower than other tissues. However, inflammation and pigmentation, along with tissue fibrosis, were also seen in the lungs. Despite the gradual reduction in clinical manifestations with berberine use, inflammation and pigmentation in the lungs were still observed compared to the control group.
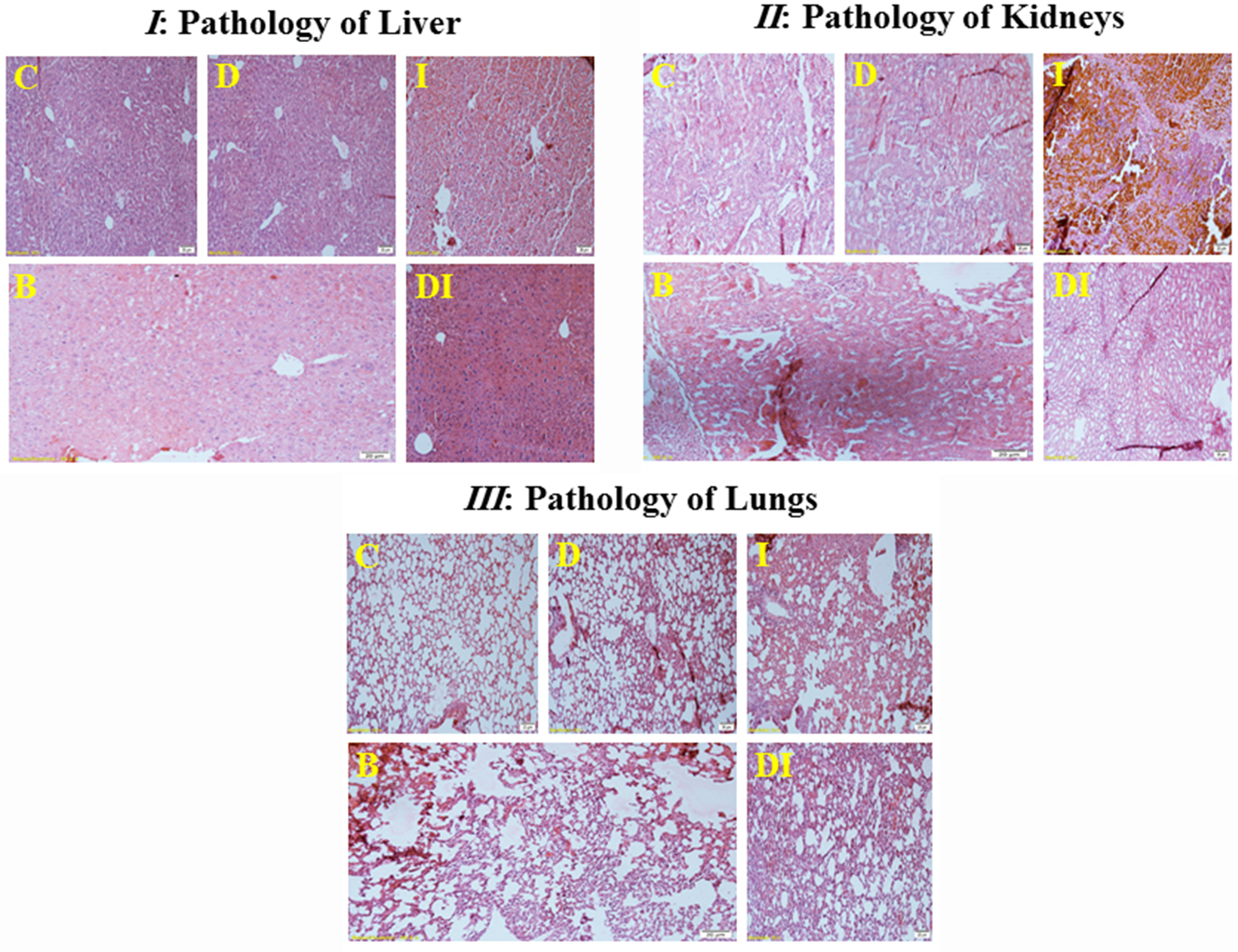
H&E staining and clinical manifestations of iron accumulation in the liver (
In the results of Prussian blue staining, dark blue colored iron deposits were observed in the tissues (Figure 4). As can be seen in the corresponding pictures, in the control groups (C) and DFO (D), which did not receive iron, the liver, kidney and lung tissues were healthy with a natural and vivid color. The most extraordinary amount of iron was observed in the hepatocellular cells. Using either DFO or berberine, excess tissue iron dropped by attaching to these chelators. Berberine appears to be more effective at reducing iron in the liver and kidney tissues than DFO. In the kidneys, most excess iron was deposited in the adipose tissue around the kidneys. With the use of berberine and DFO, a large amount of iron was chelated and excreted from the lungs.

The amount of iron deposition in the liver (
Discussion
In this study, we measured the effect of berberine on iron-overloaded mice, and a significant difference was observed between diverse groups. Researches have previously shown that iron chelators decrease the iron content. 23 -26 Certain natural products are capable of chelating excessive iron in iron-overloaded animals as a substitute for chemical chelators. 8,27,28 The practical use of berberine-containing plants in traditional medicine dates back to 3000 BC. 16,19 Various comprehensive studies have shown that berberine naturally has an antioxidant activity effect in vitro. 20,21 Iron sucrose increased the total iron content in the tissues. However, our results show that berberine drastically reduces the level of total iron in the liver, kidney and lung tissues. The iron chelating potential of berberine was relatively equal to that of DFO (a common iron chelator drug). Tissue damage in the berberine-treated groups was significantly reduced and berberine performed better than DFO (DI group). Prussian blue staining also demonstrated a significant decrease in iron pigments in the tissues of the berberine-treated groups. Previous studies have shown that certain natural products are more effective than DFO for chelating excessive iron. 8,27,29 A number of studies showed that berberine was capable of inhibiting ROS generation and properly serves as a potent antioxidant. 22,30 Berberine has the power of free radical scavenging, superoxide radical scavenging and preventing lipid peroxidation. 17,20 The marked decrease in body iron is presumably due to the direct scavenging power of berberine. 20 Molecular pathways involved in the antioxidant effects of berberine have previously been discussed. 31 Accordingly, berberine, by increasing the levels of SOD and Sirtuin 1, as well as decreasing the expression of NADPH oxidase through the AMPK pathway, reduces the oxidative conditions. 32 -34 Gholampour and colleagues showed that berberine protected the liver and kidneys of rats against the adverse effects of iron sulfate by preventing lipid peroxidation and iron chelating. 35 A 2012 study by Zhao and colleagues showed that berberine possessed antioxidant power and exerts a protective effect on rat liver cells. 22 In order to evaluate the antioxidant power of berberine, the activity of 2 enzymes, superoxide dismutase (SOD) and catalase (CAT), was measured. Antioxidant enzymes are proteins in the body whose job is to convert active oxygen species (e.g., superoxide) into stable, non-toxic molecules, and to defend against oxidants and prevent cell death. Increased free iron in tissues augments the number of active species of oxygen and oxidants. 36 With its antioxidant activity, SOD converts harmful superoxide (O2 -) radicals into oxygen and hydrogen peroxide (H2O2), followed by CAT converting the toxic hydrogen peroxide into water and oxygen. These 2 key enzymes are present in all cells exposed to oxygen and their inactivity causes much damages to cells. 37 The activity of these 2 critical enzymes reduces in iron overload conditions. Therefore, the activity rate of these 2 enzymes is a remarkable indicator for the study of oxidative stress in the body. 36 -38 The activity of catalase and superoxide dismutase in group I significantly reduced, which was consistent with previous studies. 39 -41 Treatment with berberine as an antioxidant sufficiently increased the activity of these enzymes. Likewise, other studies confirm the antioxidant effect of berberine. 41,42 The level of malondialdehyde (MDA) is a notable marker to determine the lipid peroxidation state as a representative of the oxidative stress rate. The higher the oxidative stress, the higher the MDA level. 43 Studies have shown that iron administration increases MDA levels. 28,44 In the current study, iron consumption caused oxidation and the production of extraordinary levels of MDA and subsequent increase in lipid peroxidation in the liver, kidney and lung tissues. Fortunately, berberine was able to effectively reduce the amount of MDA in these tissues. Berberine showed better efficacy than DFO in reducing MDA in the aforementioned tissues. Limitations of the study include stress in mice due to intraperitoneal injection of iron-dextran, as well as preparation of tissue slides with full details of their pathological damage. The increase in iron in the body is accompanied by obvious changes in biomarkers such as SOD, CAT and the amount of MDA. Specifically, iron suppresses and limits the activity of the 2 chief enzymes that reduce free radicals, namely SOD and CAT, through oxidation. However, berberine improves the activity of these enzymes with its chelating and antioxidant properties. Our results show that berberine might represent a valuable therapeutic option for patients at high risk of chronic iron accumulation.
Conclusion
The results of this study show that the natural compound berberine demonstrates a superior ability to reduce excess iron and chelate it in tissues, including liver, kidney and lung. By reducing oxidative stress, it can also improve the activity of the antioxidant enzymes catalase and superoxide dismutase. This remarkable ability of berberine is comparable to and often superior to that of deferoxamine as a common chelator. Berberine was equally capable of reducing malondialdehyde and lipid peroxidation resulting from an iron overdose. Therefore, we recommend further in vivo studies on berberine as an iron chelator and an antioxidant compound in diseases associated with iron-overload.
Methods
Animals and Experimental Treatments
Thirty NMRI male mice with an average weight of 20‐25 g were purchased from the Pasteur Institute of Iran (Amol, Iran). The mice were properly kept in cages in the pet room with light cycles of 12 hours light/12 hours dark under controlled temperature (24 ± 2 °C) and humidity (45 to 55%). The mice had unlimited access to food pellets containing corn flour, fish meal, wheat flour, oil seed, bone meal and salt and sterile purified municipal water during the experiments. The mice were randomly divided into 5 groups of 6. The negative control group (C) and DFO (D) received normal saline during the first month. Other specific groups, including iron (I), iron/DFO (DI) and iron/berberine (B), received 100 mg/kg/day Iron Sucrose (Venofer, Switzerland) for 4 weeks and 5 times a week intraperitoneally to produce iron-overloaded mice. The mice were then left to their own for a month to reach steadiness. 27 Afterward, group B was given 20 mg/kg/day berberine (Aldrich, UK) and groups D and DI received 25 mg/kg/day DFO (Desferal, Novartis, Switzerland), 5 times a week for 4 weeks, intraperitoneally (Figure 5). 45 Groups C and I were treated just with normal saline during the third month of continuous experimentations. 2,30,46

Diagram summarizing the steps taken in performing the experiments.
Sample and Tissue Collection
After the injections were finished, the mice were kept hungry overnight. Next day a 1:1 (v/v) combination of ketamine (90 mg/kg; Kepro, Netherlands) and xylazine (10 mg/kg; Kela, Belgium) were injected intraperitoneally to induce anesthesia in the mice. 20 to 30 minutes after injection, with deep anesthesia accomplished, the animals were euthanized by exsanguination and their liver, lung and kidney tissues were removed. 28 Three samples of each tissue were placed in 10% formalin for histopathological examination and kept at room temperature. Additionally, to measure the enzymatic activity of catalase, superoxide dismutase and malondialdehyde, 3 samples of each mouse tissue were placed in PBS and kept in a freezer at −20 °C. 47 During animal experiments, all ethical issues of working with animals were observed according to the guidelines of the Ethics Committee of Golestan University of Medical Sciences (Approval Numbers: IR.GOUMS.REC.1397.180 and IR.GOUMS.REC.1398.354).
Preparation of Tissue Samples
Mouse liver, kidney and lung tissues (100 mg) were homogenized in 1 ml of cold PBS (Thermo Fisher Scientific, USA), pH 7.4. Then, the tissue samples were centrifuged at 6,000 rpm for 20 minutes at 4 °C. Finally, the supernatant was gently transferred to a new sample tube for subsequent analyses. 27
Total Non-Heme Iron Measurement
Tissue non-heme iron content was measured according to the method described by Rebouche. 48 Briefly, 400 µL of tissue supernatant was mixed in 400 µL of solution A {including hydrochloric acid and trichloroacetic acid (Carlo-Erba, France)} and gently placed in a 95 °C water bath for 1 hour. After centrifugation at 10,000 for 10 minutes, 500 µL of the supernatant was removed and mixed in 500 µL of solution B {including ferrosin (Sigma, USA), thioglycol (Merck, Germany) and sodium acetate (Sigma, Germany)}. After 30 minutes at room temperature, the light absorption of the samples was measured using a spectrophotometer at 562 nm (Unico UV-2100 Spectrophotometer, USA), and total iron was calculated using a standard curve. 48,49
Determination of Lipid Peroxidation Rate
The quantity of malondialdehyde (MDA) was measured using a Malondialdehyde Assay Kit (ZellBio GmbH, Germany). Initially, 50 µL of either tissue sample supernatant or standards were poured into test tubes. Then, 50 µL of R4 reagent was added gently to all independent samples. Next, 1 ml of chromogen solution was poured in, and the test tubes were incubated in a boiling water bath for 1 hour. After cooling, the tubes were centrifuged at 4,000 rpm for 10 minutes. Ultimately, 200 µL of the resultant pink supernatant was transferred to a 96 cell microplate and its absorbance at 535 nm was read by an ELISA Reader (ChroMate ELISA Reader, USA). At the end, the quantity of MDA was assessed from a standard curve. 50
Measurement of Antioxidant Enzymes Activity
The activities of catalase (CAT) and superoxide dismutase (SOD) were calculated using ZellBio Assay Kits (ZellBio GmbH, Germany). To examine the activity of SOD, 10 µL of either the tissue sample homogenate or blank was dissolved in 250 µL of R1 buffer. At that point, 10 µL of R2 and then 10 µL of deionized water were gently poured into all the tubes. Afterward, 20 µL of chromogen was added to the sample tubes and 20 µL of deionized water was added to the blanks only. The samples in the microplate were then mixed well and the light absorption at 420 nm in 2 times of zero and 2 minutes was read by an ELISA Reader. In order to investigate the activity of CAT, 10 µL of either the tissue sample homogenate or normal saline was gently poured into a 96 cell microplate. Then, 100 µL of R1 buffer and 10 µL of R2 peroxide solution were added, respectively. Samples were incubated at 37 °C for 1 minute. Next, 100 µL of chromogen and then 100 µL of R4 were added. Subsequently, the light absorption of the samples at 405 nm was instantly measured by an ELISA Reader. Ultimately, the sum of SOD and CAT activity of the samples was calculated according to the proposed formula in the catalogue. 51,52
Histopathological Analyses
Tissue samples were fixed in 10% formalin immediately after removal. Next, they were immersed in molten paraffin wax to produce tissue blocks. Then, the blocks were sliced in a microtome to produce samples with a diameter of 5 µm. The tissue samples were then placed in hot air oven at 60 °C for 30 minutes to fix the tissues and thoughtfully remove paraffin. Prussian blue staining was performed to visualize iron deposition in the liver, kidney and lung tissues. In addition, hematoxylin and eosin (H&E) staining was implemented to inspect carefully the histopathological indications of iron. 28,53
Statistical Analysis
Statistical data analysis was performed using one-way analysis of variance followed by Newman-Keuls Multiple Comparison Test employed to accurately compare multiple groups. Data are shown as means ± SD with triple repetition. P < 0.05 was considered significant in all experiments.
Footnotes
Acknowledgments
This study was supported by the Golestan University of Medical Sciences and we are profoundly thankful for their generous funding.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed the receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Golestan University of Medical Sciences (Gorgan, Iran) under two projects, Grant Numbers are 110434 and 111343).
