Abstract
The chemical composition of essential oils (EOs) extracted from the aerial parts of 2 species of Juniperus was determined by Gas Chromatography-Mass Spectrometry (GC-MS). In total, 65 and 58 compounds accounting for 90.3% and 89.8% of the whole chemical composition of Juniperus oxycedrus (JO) and Juniperus phoenicea (JP) were identified, respectively, with α-pinene, α-amorphene, terpinen-4-ol, α-terpinene, and β-elemene, as major components. For the first time, the capacity to inhibit quorum-sensing for Chromobacterium violaceum CV026 and CV12472 by the investigated EOs was evaluated. Both oils exhibited good violacein inhibition on CV12472 with 100.0 ± 0.0% inhibition at minimal inhibition concentration (MIC) values. Besides, the quorum-sensing inhibition of CV026 was high at MIC for JO essential oil from fruits (JOF, 16.3 ± 2.0 mm), JO leaves (JOL, 12.5 ± 3.5 mm), JP fruits (JPF, 19.7 ± 2.5 mm), and JP leaves (JPL, 21.1 ± 5.0 mm). On both CV12472 and CV026, essential oil from J. phoenicea leaves was the most active inhibitor. All investigated EOs inhibited swarming motilities in flagellated Pseudomonas aeruginosa (PA01) in a concentration-dependent manner, and those from JP were more active than EOs from JO. Moreover, these EOs showed good antioxidant potential according to DPPH● and FRAP methods, especially the EO from JO leaves with an IC50 DPPH● inhibition value of 20.2 ± 1.0 mg/mL. Based on the obtained results, the investigated EOs are good candidates to combat microbial resistance be used as alternatives to conventional antibiotics, and equally find applications in food biosafety as preservatives.
Research on aromatic plants, their medicinal properties and the discovery of their active components has contributed to the improvement of human life.. 1 -3 These plants represent a great source of compounds, with diverse chemical structures possessing numerous biological activities. 4 -8 In different parts of the world, several aromatic plants are used in folk medicine as remedies and antimicrobials, and are being approved by the scientific world. 9,10 Due to their potential as sources of drugs and food, plant components have attracted consumer’s interest, leading to high demand for natural food and products, including EOs, which are famous for their antimicrobial properties and aromas. 11 -15 Juniper plants are widely used by indigenous populations, including those in Algeria for health purposes. 16,17
Due to their antimicrobial properties and specific compositions, Juniperus species find numerous applications in traditional medicine, as skin drugs, cosmetics, fragrance, and flavoring, particularly in European cuisine and beverages. 18 -23 The most common species of Juniperus are Phoenician juniper (Juniperus phoenicea L.) and Cade juniper (Juniperus oxycedrus L.). Both are evergreen, but are quite distinct, with scaled leaves and berry-like fleshy fruits that are a dark red color when ripe for J. phoenicea, and needle-like leaves with brownish or red-brown berries for J. oxycedrus. Ethnobotanical and ethnopharmacological studies show the use of EOs and all parts of Juniperus as a remedy for several ailments. J. phoenicea has antirheumatic, antiseptic, antihypertensive 24 and anti-inflammatory 25 properties, while J. oxycedrus has hypoglycemic, 26 antirheumatic, and anti-inflammatory 24,27 properties. The chemical analyses report a large variety of compounds in the EOs of both species, 28,29 including monoterpenes, with α-pinene as the main compound in leaves and fruits. 29 -31 J. phoenicea essential oil has other major components, like β-phellandrene, α-terpinyl acetate and δ−3-carene, 18,30,32 while 13-epi-manoyl oxide and (Z)−6-pentadecen-2-one are amongst the major components found in J. oxycedrus. 33
Many studies report the antimicrobial activity of Juniperus plants EOs by conventional means, without studying their ability to inhibit quorum-sensing traits in microorganisms, which are responsible for the virulence of infections and emergence of resistant strains. This work aims to evaluate the anti-quorum sensing activities of bacteria produced by essential oils extracted from J. phoenicea and J. oxycedrus (leaves and fruits). Likewise, and due to the importance of natural products as antioxidants agents, the estimation of the EOs antioxidant potential was also investigated.
Materials and Methods
Plant Material
Plants collection and preparation
The investigated species were collected in the Tebessa Region (East of Algeria)
Essential oil distillation
EOs were extracted by hydro-distillation using a Clevenger apparatus following the European Pharmacopoeia method. This process yielded 4 EOs, J. oxycedrus fruits (JOF) J. oxycedrus leaves (JOL), J. phoenicea fruits (JPF) and J. phoenicea leaves (JPL). Distillation took approximately 6 hours from the first drop of distillate until exhaustion of the EO contained in the plant. The resulting oils were stored in dark vials at a temperature of 4 °C in a refrigerator, pending analyses.
GC/EID/MS (Gas Chromatography With Electronic Impact Detector/Gas Chromatography-Mass Spectrometric) Analyses
EOs characterization was undertaken in the Scientific and Technical Research Center in Physico-Chemical Analysis (Bousmail, Algiers-Algeria). Samples were analyzed using a Hewlett Packard Agilent 6890 gas chromatography (GC) system, with electronic impact detector (EID) and Hewlett Packard Agilent 5973 mass spectrometer (MS), fitted with a quadrupole mass analyzer and capillary flow technology enabling simultaneous analysis of the sample on both detectors. A HP-5MS (30 m x 0.25 mm, film thickness 0.25 µm) capillary column, with 5% phenyl and 95% dimethylpolysiloxane as a stationary phase was used. Operating conditions were as follows: oven temperature 60 °C (8 minutes) 2 °C/min to 250 °C; isotherm during 10 minutes (total analysis time: 113 minutes); flow rate of 0.5 mL/min (helium with a purity of N6); injector T = 250 °C; 0.2 µL injection volume at split ratio 50:1. The mass spectrometry conditions were as follows: ionization voltage 70 eV, ion source temperature 230 °C, transfer line temperature 270 °C with a solvent delay of 3.5 minutes, and mass range from 30 to 550 Da. The MS were operated in scan TIC mode and identification was achieved with the aid of AMDIS (Automated mass spectral deconvolution and identification system), and by comparison with data from literature libraries and estimated Kovat′s (Retention) indices (determined using a mixture of a homologous series of normal alkanes from C9 to C25 in n-hexane, under the same conditions mentioned above). The identification of the components contained in the analyzed EOs was made and the percentage ratios of EO components were deduced by the normalization method of the GC/EID peak areas.
Antioxidant Activity
DPPH-free radical scavenging assays
The 2,2-diphenyl-1-picrylhydrazyl (DPPH●) free radical scavenging assay was used according to the protocol described previously, 34 with slight modifications. One mL of each EO in methanol solution at different concentrations (from 0.0 to 92.0 mg/mL) was added to 3 ml of 0.004% DPPH● methanolic solution. A mixture of 1 ml of methanol with 3 ml of DPPH● methanol solution made up the negative control. The absorbance was taken at 515 nm, after 60 minutes of incubation in the dark, at room temperature. The positive control consisted of a methanol solution of a standard antioxidant, BHT, measured under the same conditions as the samples. The following formula expressed the reducing power (or the inhibition) of the DPPH● 35 :
Where, Abs c is the absorbance of the negative control (the solution of DPPH● in the absence of the EO or BHT), Abs e is the absorbance of the DPPH● solution in the presence of the EO, or BHT. The experiment was conducted in triplicate and the IC50 value, which is the concentration of the substrate causing the loss of 50% of the DPPH●, determined for each EO.
Iron reducing power (FRAP)
The EOs reducing power was evaluated using the iron-reducing antioxidant power (FRAP) method, according to the procedure described by Oyaizu, 36 with some modifications. This technique is an uncomplicated, fast and reproducible test. 37 Into a glass test tube, 0.5 ml of the sample at different concentrations, 1.25 ml of 0.2 M phosphate buffer (pH 6.6) and 1.25 ml of potassium ferricyanide 1% ([K3Fe(CN)6]) were introduced. The mixture was incubated in a water bath at 50 °C for 20 minutes and then cooled to room temperature. To stop the reaction, 2.5 ml of trichloroacetic acid (TCA, 10%) was added to the obtained mixture and the tubes were centrifuged at 3000 rpm for 10 minutes. Distilled water (1.25 ml) and 250 µL of freshly prepared ferric chloride (FeCl3) 1% were added to 1.25 ml of a supernatant. The absorbance of the reaction mixture was read after 30 minutes, at 700 nm using a Shimadzu UV-1700 Pharma spectrophotometer. A blank was prepared by replacing the EO with methanol and the positive control involved ascorbic acid solution, at the same selected concentrations and under the same experimental conditions.
Microbial Strains and Determination of Minimum Inhibitory Concentration (MIC)
The microorganisms used in this study were Pseudomonas aeruginosa PA01, Chromobacterium violaceum CV12472, and Chromobacterium violaceum CV026. MICs were determined by the microtitre broth dilution method, as recommended by the Clinical and Laboratory Standards Institute. 38 The MIC was defined as the lowest EO concentration that yielded no visible growth. The test medium was Mueller-Hinton broth (MHB) and the density of bacteria was 5 × 105 colony-forming units (CFU)/mL. Cell suspensions (100 µL) were inoculated into wells (96-well microtitre plates) in the presence of EO with different final concentrations (2, 1, 0.5, 0.25, 0.125, 0.0625, 0.03125 mg/mL). The inoculated microplates were incubated at 37 °C for 24 hours before being read.
Violacein Inhibition Assay Using C. Violaceum CV12472
All EOs were subjected to qualitative analysis to find their QSI (Quorum-sensing inhibition) potentials against C. violaceum ATCC 12472. 39 Overnight culture (10 µL) of C. violaceum (adjusted to 0.4 OD at 600 nm) was added to sterile microtiter plates containing 200 µL of LB broth, and incubated in the presence and absence of sub-MIC concentrations of EOs of J. phoenicea and J. oxycedrus. These plates were incubated at 30 °C for 24 hours and then observed for the reduction in violacein pigment production. The absorbance was read at 585 nm. Each experiment was made in triplicate and the percentage of violacein inhibition was calculated via the following formula:
Bioassay for Quorum-Sensing Inhibition (QSI) Activity Using C. Violaceum CV026
Quorum sensing inhibition was evaluated as described elsewhere. 39,40 Five mL of warm molten Soft Top Agar (1.3 g agar, 2.0 g Tryptone, 1.0 g sodium chloride, and 200 ml deionized water) was seeded with 100 µL of overnight CV026 culture and 20 µL of 100 µg/mL C6HSL was added as exogenous AHL (acyl homoserine lactone) source. This preparation was mixed and poured immediately over the surface of a solidified Luria Bertani Agar (LBA) plate as an overlay. Wells 5 mm in diameter were made on each plate after solidification of the overlay. Each well was filled with 50 µL of sub-MIC concentration filter-sterilized EO. A white or cream-colored halo around this well against a purple lawn of activated CV026 bacteria indicated QSI. The activity detection limit was determined by applying serial dilutions of the EO (1:1 to 1:8, using LB broth as diluent) and the endpoints were estimated as the lowest dilution of EO leading to discernible inhibition of violacein synthesis. Each experiment was repeated 3 times. The assay plates were incubated at 30 °C for 3 days, and then the diameters of the quorum sensing inhibition zones were measured.
Swarming and Swimming Motility Inhibition of P. Aeruginosa PA01
The swarming motility inhibition assay was carried out as described previously. 39,41 Briefly, overnight cultures of P. aeruginosa PAO1 strain were point inoculated at the center of swarming plates consisting of 1% peptone, 0.5% NaCl, 0.5% agar, and 0.5% of filter-sterilized D-glucose with various concentrations of EO (50, 75, and 100 µg/mL). A plate without the EO was maintained as a control and the plates were incubated at an appropriate temperature in an upright position for 18 hours. The swarming migration was recorded by following swarm fronts of the bacterial cells. For the swimming motility assay, the P. aeruginosa PAO1 strain was inoculated at the center of the swarming agar medium consisting of 1% peptone, 0.5% NaCl, 1.5% agar, and 0.5% of filter-sterilized D-glucose with increasing concentrations of EO (50, 75, and 100 µg/mL). Plates were then wrapped with Saran Wrap to prevent dehydration, and incubated at 37 °C in an upright position for 16 hours. The reduction in swimming migration was recorded by measuring the swim zones of the bacterial cells after 16 hours.
Results and Discussion
Chemical Composition of Essential Oils
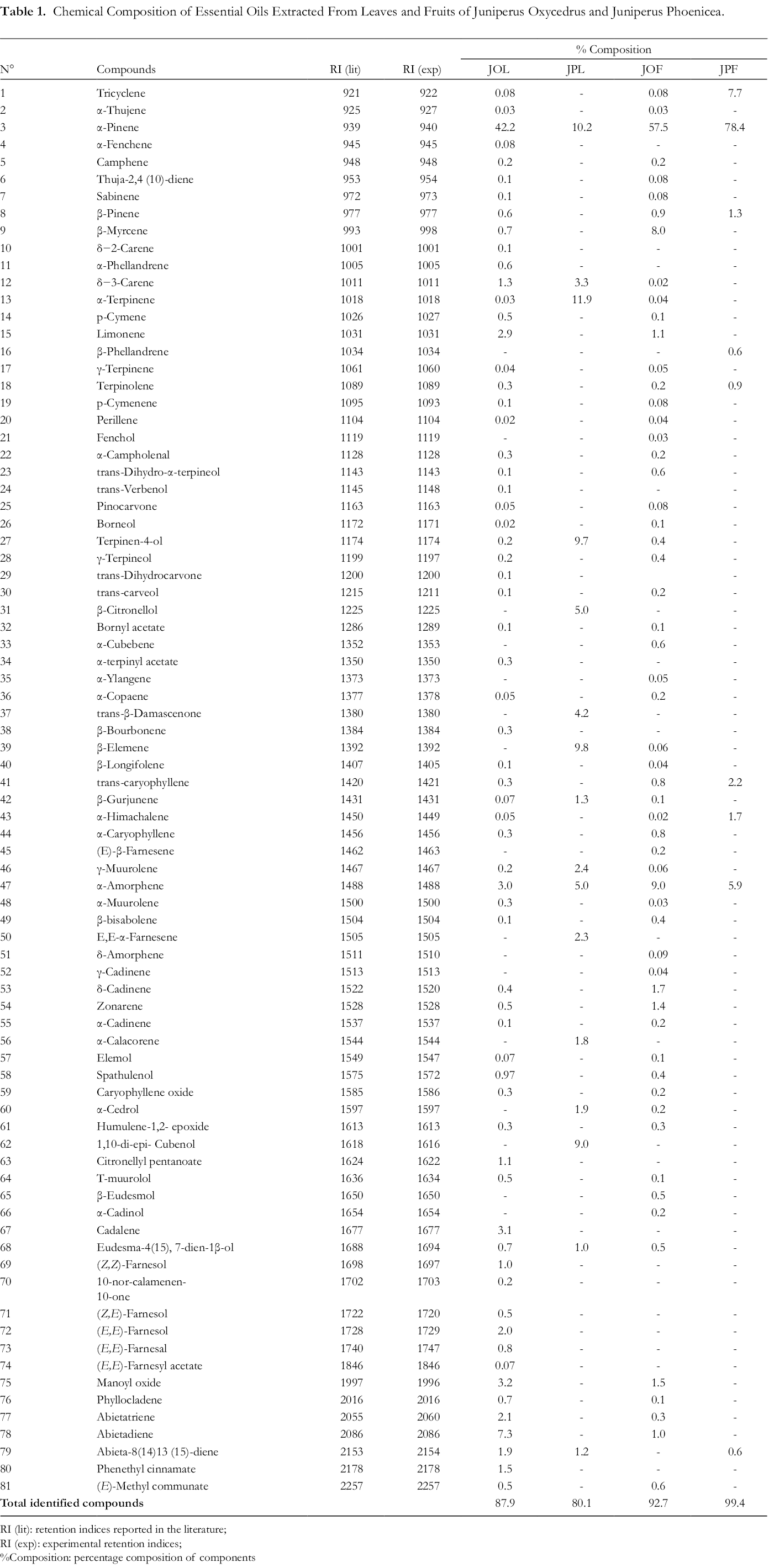
Monoterpenes were the most abundant components in all the investigated oils. In the leaf and fruit oils of J. oxycedrus, 65 and 58 compounds, respectively, were identified and presented in Table 1.
Chemical Composition of Essential Oils Extracted From Leaves and Fruits of Juniperus Oxycedrus and Juniperus Phoenicea.
RI (lit): retention indices reported in the literature;
RI (exp): experimental retention indices;
%Composition: percentage composition of components
The main components found in the fruit oil were α-pinene (57.5%), α-amorphene (9.0%), β-myrcene (8.0%), δ-cadinene (1.7%), manoyloxide (1.5%), and zonarene (1.4%). For the leaf oil, the major components were α-pinene (42.2%), abietadiene (7.3%), manoyloxide (3.2%), cadalene (3.1%), α-amorphene (3.0%), limonene (2.9%), and abietatriene (2.1%). Most of the compounds identified in the EOs extracted from J. oxycedrus in this study have been previously described for the same subspecies, although in different amounts, 42,43 and the monoterpenes are usually in higher amounts in the leaf EO than in that of the fruits. However, α-pinene was abundant in the EOs from the different plant parts, although some studies on J. oxycedrus oils from other Algerian regions did not report this molecule as the main component, indicating a regional variation in chemical composition and also a variation between different subspecies. 22,42
In most investigations, the major components of J. oxycedrus leaf EO have been α-pinene and manoyl oxide, while those of J. oxycedrus fruit essential oils are α-pinene and α-amorphene, showing a resemblance to the results reported here. J. oxycedrus EOs from Kosovo contain β-myrcene, α-pinene, limonene and α-amorphene as major components. Therefore, monoterpenes and sesquiterpenes are the predominant class of volatile compounds in Juniperus oxycedrus EOs.
44
Major components detected in J. oxycedrus collected in Bulgaria showed α-pinene, α-amorphene, limonene, δ-cadinene, γ-cadinene, α-curcumene, manoyl oxide, α-caryophyllene, β-caryophyllene, and caryophyllene oxide, and they were used in grouping the oils from this plants collected from different regions into chemotypes of 5 chemical groups showing varying аntimicrobial activity.
21
Analysis of both the EOs of J. phoenicea revealed few compounds compared to those from the other species. In the leaf and fruit EOs, 16 compounds (80.1%) and 9 compounds (99.4%) were identified, respectively (Table 1). The main components in the leaf oil were α-terpinene (11.9%), then α-pinene (10.2%), β-elemene (9.8%), terpinen-4-ol (9.7%) and 1,10-di-epi-cubenol (9.0%). The main components in the fruit oil were α-pinene (78.4%), followed by tricyclene (7.7%), α-amorphene (5.9%), trans-caryophyllene (2.2%), α-himachalene (1.7%) and β-pinene (1.3%). In previous work, similar compounds were identified in J. phoenicea, but as usual, the amounts of each compound vary. For J. phoenicea collected from Tunisia, α-pinene, δ−3-carene, α-terpineol, α-himachalene, β-caryophyllene, and γ-cadinene were the main components occurring in variable amounts in the leaf and fruit EOs.
45
Some compounds were detected in both J. oxycedrus and J. phoenicea, while the most abundant compound α-pinene occurred in both species and in all investigated plant parts, but in different amounts {in J. oxycedrus leaf EO (42.2%), J. oxycedrus fruit EO (57.5%), J. phoenicea leaves EO (10.2%), and J. phoenicea fruit EOs (78.4%)}. As reported in a study for J. oxycedrus and J. phoenicea growing under the same ecological conditions in Morocco,
46
the chemical composition variation of Juniperus EOs could be due to multiple factors, such as collection region, harvesting season, species and subspecies, plant parts and the fruit ripeness, as well as the extraction methods used. Potent compounds, such as limonene, sabinene, myrcene, α-pinene, and β-pinene, in Juniperus are responsible for its excellent antifungal and antibacterial properties. For example, EOs of these plants have shown good bacterial and fungal inhibitions, and interestingly, against certain fungi that attack the skin, leading to skin diseases such as ringworm, yeast, and mouth infections..
47
Mycobacteria are known to cause serious humans health effects, but M. avium, M. intracellulare, and M. gordona
Antioxidant Activity
DPPH-free radical scavenging assays
When the free radical DPPH● accepts a proton from an antioxidant compound, it is reduced and turns from purple to yellow, which can be measured quantitatively from the change in its absorbance value. 49 Each IC50 value of the tested EOs is given on the histogram (Figure 1). Each value represents the mean of 3 replicates ±the standard deviation. According to the obtained results, all EOs had IC50 values below 75 mg/mL, indicating good antioxidant potential (good free radical scavenging). Amongst these EOs, those from J. oxycedrus fruits and J. phoenicea leaves showed the greater free radical scavenging capacity. The highest trapping power of DPPH● was observed for the fruit EO of J. oxycedrus with an IC50 value of 20.2 ± 1.0 mg/mL, followed by that of J. phoenicea leaves (36.1 ± 2.1 mg/mL). The other EOs exhibited lower potencies; the fruits of J. phoenicea and leaves of J. oxycedrus had IC50 values of 74.9 ± 2.5 mg/mL and 62.6 ± 3.8 mg/mL, respectively.

Representation of the percentage reduction variation of the DPPH● as a function of the concentration of investigated EOs with the antioxidant BHT as standard.
Figure 2 shows that the percentage of free radical inhibition increases proportionally with the EO concentration before reaching a saturation level. However, the antioxidant power of J. phoenicea fruits was 2 times lower than that of its leaves, but 3 times lower than that of J. oxycedrus fruits and close to that of J. oxycedrus leaves. According to these graphs, at 32.2 mg/mL, the free radical scavenging power was shown to be almost the same for the leaves of both studied species. Moreover, J. oxycedrus fruit EO was the most active amongst them with an inhibition percentage up to 92.2%, which is very close to that recorded for BHT.

Graphical representation of the inhibition variation of the DPPH● as a function of different concentrations of tested EOs compared with BHT.
Iron reducing power (FRAP)
To confirm the EOs antioxidant potential, previously evaluated by the DPPH● free radical scavenging test, kinetic monitoring by the FRAP method was employed. The results of the EOs iron-reducing power of each species and ascorbic acid are shown in Figures 3 -7. The obtained curves demonstrate that the increase in absorbance correlated with both concentration and time, due to the probable presence of antioxidant compounds capable of offering electrons, thus reducing Fe3+ to Fe2+. These results indicate that all the tested EOs had more or less reducing power. At a concentration of 9.2 mg/mL, EOs from JPL and JOL exhibited a high absorbance of 0.68 and 0.60, respectively, while the EOs from JOF and JPF had absorbance values of 0.56 and 0.48, respectively. However, the obtained values were low compared to those of ascorbic acid.

Kinetic monitoring of the effects of J. oxycedrus leaf EO on iron reduction.
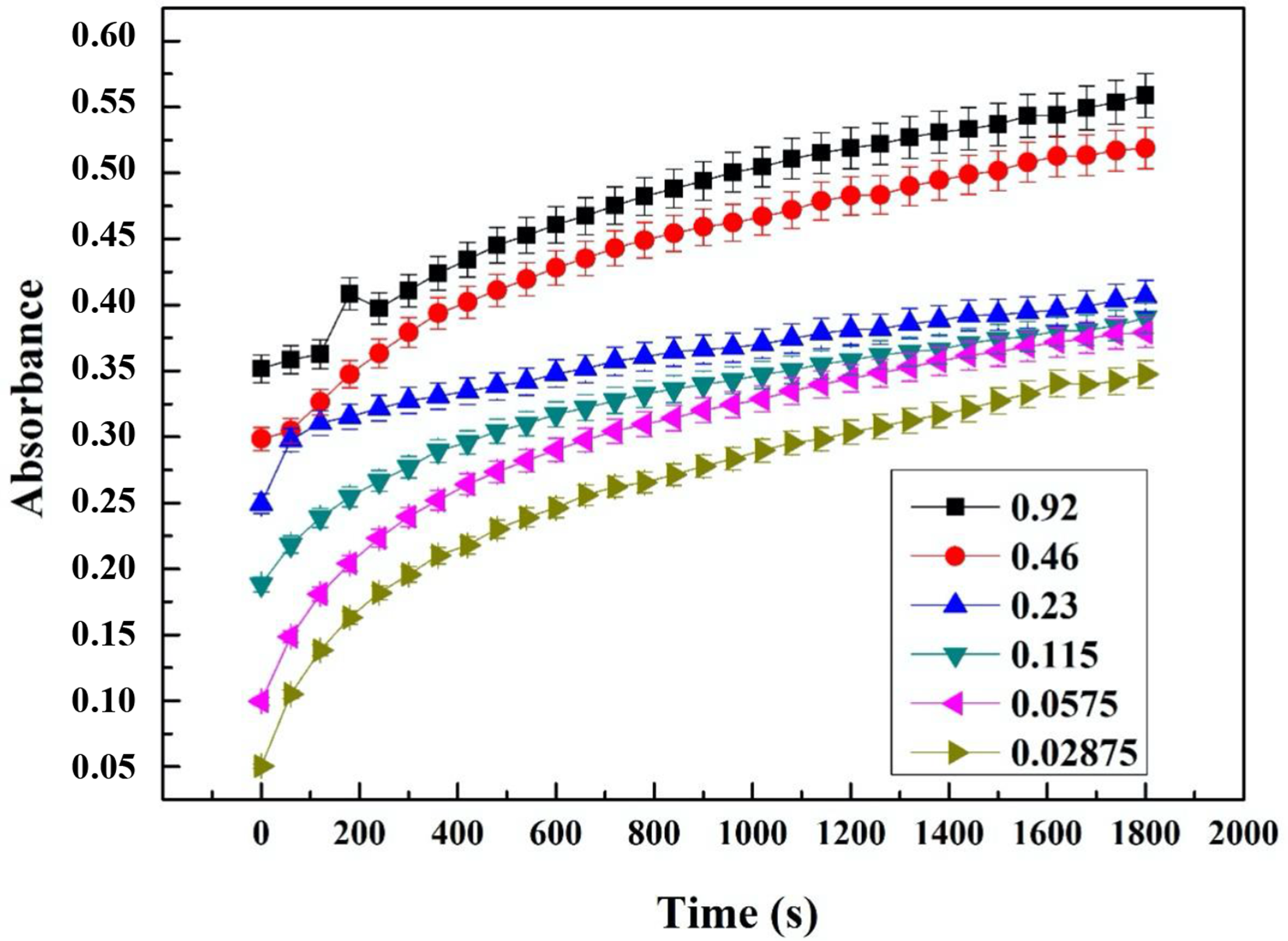
Kinetic monitoring of the effects of J. oxycedrus fruit EO on iron reduction.

Kinetic monitoring of the effects of J. phoenicea leaf EO on iron reduction.

Kinetic monitoring of the effects of J. phoenicea fruit EO on iron reduction.
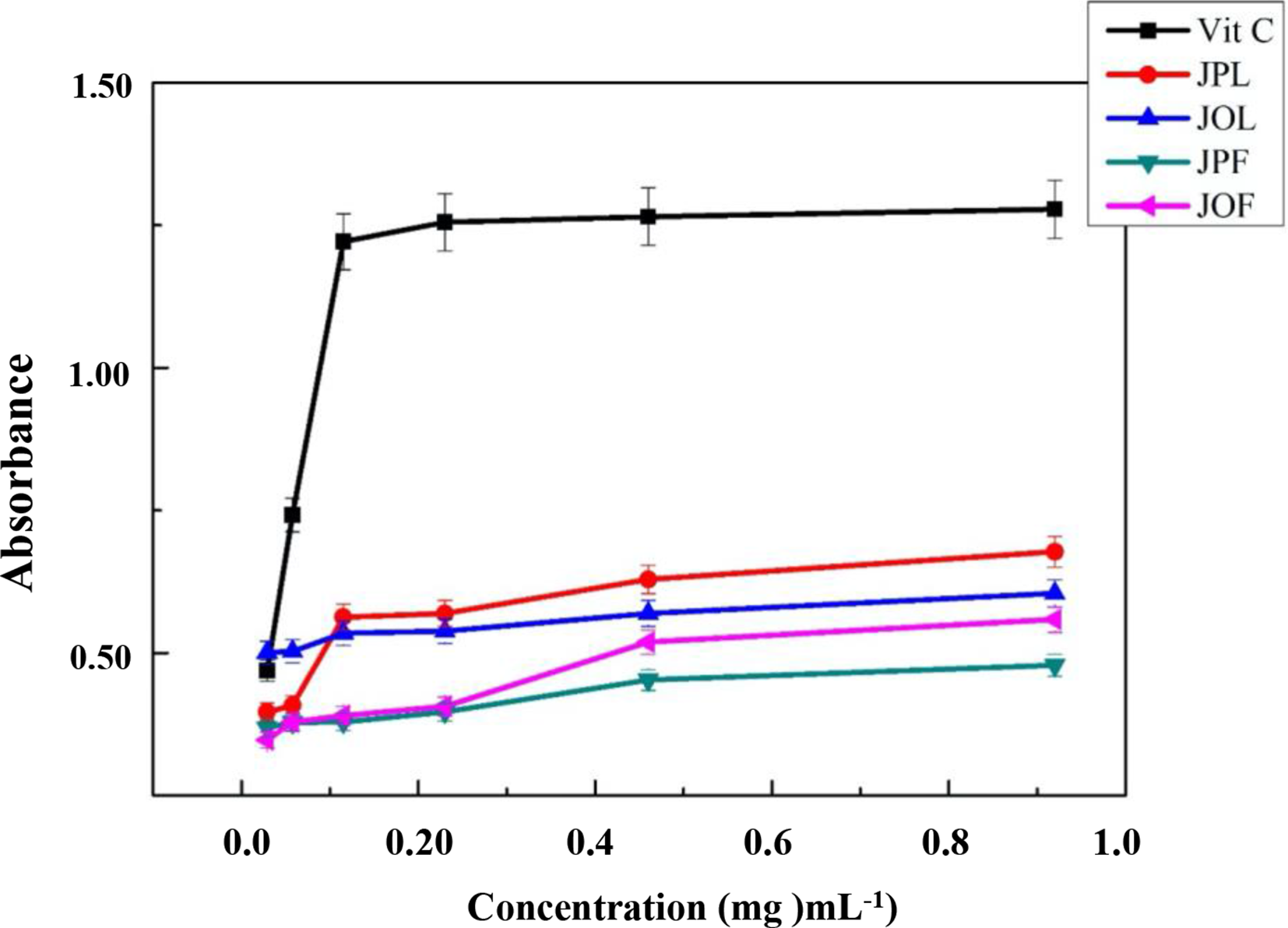
Reducing power of iron exerted by EOs compared to ascorbic acid.
Violacein Inhibition Using C. Violacein CV12472
C. violaceum CV12472 produces a violet coloration while growing by a quorum sensing (QS) mediated process. This color is due to the presence of violacein and can be observed and quantified easily as a marker-trait that makes this bacterium suitable for quorum sensing activity research. 50 MIC values of EOs were determined so that the assays could be affected at sub-MIC concentrations before measuring violacein production inhibition. All EOs from fruits and leaves of J. oxycedrus showed MIC values of 0.1 mg/mL, and those from fruits and leaves of J. phoenicea exhibited MIC values of 0.05 mg/mL. Thus, the antimicrobial activity of J. phoenicea was higher than that of J. oxycedrus against C. violaceum CV12472. The JOF EOs showed better violacein inhibition than the EOs from JOL. The percentage inhibition of violacein of JOF was 100.0 ± 0.0% at MIC and MIC/2, while JOL had 100.0 ± 0.0% only at MIC. At MIC/8, JOF had 07.5 ± 1.1% inhibition of violacein, while JOL had no inhibition at this concentration. On the other hand, the JPL EO had higher violacein inhibition than the JPF EO. The violacein inhibition of JPL was 100.0 ± 0.0% from MIC to MIC/4, and this inhibition reduced to 06.0 ± 1.5% at MIC/16, which is the lowest activity. JPF exhibited violacein inhibition of 100.0 ± 0.0 only at MIC, and JPF showed no activity beyond MIC/4. JPL showed the best violacein inhibition activity amongst all tested samples.
Quorum-Sensing Inhibition (QSI) Activity Using C. Violaceum CV026
MIC values of the various EOs against the biomonitor strain, C. violaceum CV026 were determined with their QSI at sub-MIC concentrations. The flower EOs of both plants had MIC values of 0.0625 mg/mL. For the leaf EOs, JOL had a MIC of 0.25 mg/mL, while JPL exhibited a MIC of 0.125 mg/mL. The observation of a white or cream-colored halo around this well against a purple lawn of activated CV026 bacteria indicated a QSI.
For J. oxycedrus EOs, JOF had higher anti-QS activity showing inhibition zones of 16.3 ± 2.0 mm at MIC and 09.0 ± 0.7 mm at MIC/4, while JOL had an inhibition zone of 12.5 ± 3.5 mm at MIC and showed no activity at MIC/4. For J. phoenicea, JPL had higher activity, with inhibition zones of 21.1 ± 5.0 mm at MIC and 08.0 ± 0.5 mm at MIC/8, as compared to JPF, which had QS inhibition zones of 19.7 ± 2.5 mm at MIC and 08.5 ± 3.0 mm at MIC/4. Amongst all the samples, JPL had the highest anti-QS activity at the same concentration, and it was the only sample, which displayed a QS inhibition zone at MIC/8 concentration.
Swarming Inhibition of P. Aeruginosa PA01
In the early stages of biofilm formation, swarming migrations have an important role mediated by QS. In this assay, the anti-swarming potential of the EOs was performed using the flagellated bacterium P. aeruginosa PAO1 as the test strain. All EOs were able to inhibit swarming motility at 100 µg/mL and 75 µg/mL concentrations. However, at 50 µg/mL, all EOs displayed anti-swarming activity, except JOL. For EOs of the same plant, EOs from fruits had higher swarming inhibition activity than those from leaves. The J. phoenicea fruit EOs had higher swarming inhibition than the other tested EOs. It is worth mentioning that, since swarming is a key step for bacteria to move and colonize surfaces and to form biofilms, this activity is very necessary and shows that EOs from J. oxycedrus and J. phoenicea can be used in protecting surfaces from microbial invasion.
EO compounds are capable of triggering the expression of genes that promote the production of autoinducer molecules, which are responsible for virulence factors and anti-quorum sensing activity of the Gram-negative bacterium C. violaceum. 51 This is so because the bacterial intercellular communication is mediated by various auto-inducers or signaling molecules and the disruption of this communication is a promising strategy for overcoming virulence and resistance observed in bacterial infections. Since, in our study, the investigated EOs could inhibit violacein production in CV12472, it is clear that these EOs can interrupt acyl-hormoserine lactone (AHL) production. On the other hand, since externally supplied acyl-hormoserine lactone to CV026 could not permit it to produce violacein, it is also clear that these EOs blocked signal reception. Therefore, the studied Juniperus species EOs had the double advantage of blocking acyl-hormoserine lactone production and reception. Without fully investigating the mechanism, it was noticed in previous studies that EOs acted through either inhibition of the production of acyl-hormoserine lactone or by blocking the cell-to-cell communication system in bacteria. 52
Various mechanisms explain the anti-quorum sensing activity of plant EOs such as inhibition of acyl-hormoserine lactone production, deactivation of acyl-hormoserine lactone binding to the cognate receptor, or signal reception. 53 In most bacteria and their infections, motility, such as swarming, plays a key and primordial role in the movement towards and colonization of surfaces involving spreading of bacteria across the surface and subsequent establishment of biofilms. 54 All investigated EOs in this study were able to inhibit swarming motility in P. aeruginosa, which is a flagellated bacterium and is believed to use swarming movements to attach, spread, and colonize surfaces before forming resistant biofilms on them. Hence, this swarming inhibition is very beneficial in protecting surfaces and food substances onto which bacteria can attach and promote their spread and contamination.
Conclusion
Most conventional antibiotics on the market, which are based on inhibiting growth and killing bacteria, are falling out of use due to the development of microbial resistance. Most resistance and virulence traits in bacteria occur through quorum sensing mechanisms involving bacterial cell-to-cell communication. Therefore, disrupting quorum sensing is a good and promising strategy to remedy this situation. In this work, EOs obtained from aerial parts of J. oxycedrus and J. phoenicea (leaves and fruits), by hydro-distillation were analyzed by GC-MS, and monoterpenes were the most abundant in all samples. Evaluation of the antioxidant activity of the investigated EOs was achieved using 2 methods to reveal their antioxidant potential. All EOs showed a potent antioxidant activity, especially the JOF EO with an IC50 value of 20.2 ± 1.0 mg/mL via DPPH● scavenging, due certainly to the chemical composition of each EO and to the synergy between the different compounds. As a result, these Juniperus essential oils could be used as an alternative to synthetic antioxidants since natural products are more compatible with living systems and can be safer than synthetic ones. J. phoenicea EOs exhibited the best violacein inhibition activity with the highest anti-QS activity at MIC/8 concentration. Juniperus EOs could be suitable alternatives to conventional antibiotics against resistant bacterial strains since they can inhibit bacterial growth and destroy the quorum-sensing networking.
Footnotes
Acknowledgments
The authors are grateful to Larbi Tebessi University and Mugla Sitki Kocman University for providing facilities towards the realization of this collaborative research.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.


