Abstract
The evidence regarding a deficiency of manganese (Mn) in humans is scarce. So the aim of this narrative review was to consider the state of the art on the relation between manganese and bone health in humans and the effectiveness of manganese supplementation (alone or with other micronutrients) on bone mineralization. This review included 4 eligible studies. All the literature published is in agreement in showing that osteoporotic women have lower serum Mn levels than women with normal bone mineral density, thus confirming the essential role of manganese in the synthesis of cartilage and bone collagen, as well as in bone mineralization and confirming the studies on the animal model. Considering the human studies that evaluated the effectiveness of an oral Mn supplement for a long period (2 years) on the bone mineral density of menopausal women, both of the clinical trials showed that bone loss was significantly greater in the placebo group than in the group taking supplementation, equal to 5.0 mg Mn/day in the study by Strause, and equal to 2.5 mg Mn/day in the study by Saltman, considering, however, that supplementation was represented by a set of microelements (Mn, copper, and zinc) and by calcium.
Manganese (Mn) is a metal element present both naturally and as a consequence of contamination of the soil, sediments and water; it can exist in different oxidation states, of which Mn2 + and Mn3 + are the most important from a biological point of view. 1 Food represents the main source of Mn exposure for the general population; its level in drinking water varies from 0.001 to 0.1 mg/L, while daily intake through the ambient air is significantly lower. Mn has proven essential for various species; it is a component of arginase, pyruvate carboxylase and superoxide dismutase, plays the role of cofactor of various enzyme systems and activates others (glycosyl transferases, for example, are specifically activated by Mn). 1,2 Due to its poor solubility, only a small percentage of food Mn is absorbed; this element is largely excreted very quickly in the intestine via bile and minimally with urine. It tends to accumulate in tissues rich in mitochondria, such as the liver and pancreas, and in the brain, in particular in the globus pallidus, striatum and substantia nigra. 1,3
Mn deficiency in animals can lead to several adverse effects, including growth alterations, skeletal abnormalities, reproductive defects, ataxia, and lipid and carbohydrate metabolism defects. On the contrary, the evidence about a deficiency of Mn in humans is scarce and no specific manganese deficiency syndrome has been described so far (effects such as hair color change from black to red, slow nail growth and scaly dermatitis have been observed only in experimental conditions). 1,2
Studies on Animal and in Vitro Models
According to data in the literature, bone represents one of the main deposition tissues of Mn (containing about 43% of total body Mn). 4 Mn, in fact, seems to be an important osteotropic element: In addition to stimulating the synthesis of the bone matrix, it seems to have an effect on calcification in general. 5 In a 1986 study, 150 female rats were divided into 3 groups according to the quantities of Mn and/or copper (Cu) possibly added to their diet: (1) group N, normal intake of Mn (66 ppm) and Cu (5 ppm); (2) group L, low intake of Mn (2.5 ppm) and Cu (0.5 ppm); (3) group D, with no addition of Mn and normal supply of Cu (5 ppm). After a period of 12 months, the rats belonging to group D (without Mn intake) showed significantly higher serum calcium and phosphorus levels than the rats of group N. On the other hand, calcium and Mn concentrations were found to be significantly lower in group D than in group N, with an inverse correlation between femoral concentration and serum calcium concentration in rats in groups L and D. These observations could be the consequence of alterations in the regulation of calcium at bone level (decreased mineralization) associated with increased bone resorption, in turn caused by long-term manganese and copper deficiencies. 6
In order to investigate the possible relationship between Mn deficiency and bone development, Liu and collaborators administered to 3 groups of chicks the same basal diet, supplemented with 60 (control group), 40 (Mn I deficient group) and 8.7 mg Mn/kg of food (group deficient in Mn II), respectively. Compared to the control group, the Mn deficient groups showed, in a dose-dependent manner, a markedly less trabecular reticulum at the tibial level, with a reduction in the number and thickness of the trabeculae. At an ultrastructural level, the presence of damaged osteoblasts (rupture of the nuclear and external mitochondrial membranes, loss of the mitochondrial crests, and alterations in the endoplasmic reticulum) was observed in both groups with low Mn intake, with a higher incidence, in particular, in group II. Furthermore, in these 2 groups (and to a greater extent in II), a reduced level of expression of the OPG (osteoprotegerin) and RANKL (receptor activator of nuclear factor kB ligand) genes was found with an increase in the RANKL/OPG mRNA ratio, an index of an alteration in the dynamic balance between resorption and bone formation in favor of the former. Overall, therefore, Mn deficiency could interfere with normal bone development by inhibiting the viability of osteoblasts and altering the RANKL/OPG mRNA ratio. 7 The same authors, in a second study carried out on chicks, divided in a similar way to the previous one, in 3 groups based on the Mn content of their diet, observed a significant reduction in the size of the proliferative zone and an increase in the rate of apoptosis of chondrocytes at the level of the tibial growth cartilage in animals deficient in Mn. The deficit of this element would therefore be related to disorders in bone development and growth. 8
A study conducted in 1998 on young male rats on a poor Mn diet (- Mn: 0.5 µg/g of food) for 3 months had already shown a reduction in the growth rate, with a significant decrease in the body weight of these animals, compared to the control group (+Mn: rats fed a diet containing 45 µg Mn/g of food). The deficiency status induced by the poor Mn diet (confirmed by the finding of a 15-fold reduction in the concentration of Mn in the liver) was associated with an alteration in the metabolism of GH and IGF-1: in -Mn rats, a decrease in circulating concentrations of IGF-1 and insulin and an increase in GH levels compared to controls had been observed, suggesting how these modifications could, at least in part, be responsible for the growth anomalies observed in –Mn animals. 9
Using ovariectomized rats as the model of osteopenia, Rico et al. investigated the effects of Mn supplementation, alone and in association with Cu, on vertebral and femoral bone mass. The animals were divided into 4 groups of each of 15 subjects: non-ovariectomized (Sham-OVX), ovariectomized but without supplements (OVX), ovariectomized and receiving integration with 40 mg Mn/kg of food (OVX + Mn) and integration with Mn and 15 mg/kg of Cu (OVX + Mn + Cu) for 30 days. BMD (assessed by DXA) and bone mineral content (BMC) at the femoral level were significantly lower in the OVX group than in the Sham-OVX group and the 2 groups receiving supplementation with Mn and Mn +Cu; overlapping results were also found at the vertebral level (fifth lumbar vertebra). The authors therefore concluded that Mn supplementation is able to inhibit effectively the loss of bone mass induced by ovariectomy, both at the level of the axial skeleton and at the appendiceal level, an effect not further accentuated by the addition of Cu, assuming that this inhibitory action of Mn can be explained by the fact that this element represents an indispensable co-factor for some enzymes involved in carbohydrate metabolism, in particular for the synthesis of mucopolysaccharides in the bone, and is able to counteract the bone resorption induced by free radicals. 10
In another rat study, the level of Mn in the teeth and mandibular bone, which was significantly decreased following ovariectomy, increased again after administration of 17 β-estradiol, suggesting that the deposition of Mn at the bone level could be influenced by the estrogenic state. 5,11
In a Bae & Kim study, a group of ovariectomized rats (OVX) and a group of non-ovariectomized rats (sham-operated) were further divided into a group fed a diet with an adequate concentration of Mn (Mn 0.001%) and one fed a high concentration diet (0.01% Mn) for 12 weeks. Lumbar BMD was significantly higher in sham rats supplemented with Mn than in non-supplemented sham rats; femoral BMD showed an increase following Mn supplementation in both the OVX and sham groups. Furthermore, serum levels of osteocalcin, a sensitive marker of bone formation, were considerably higher in the groups with the highest intake of Mn. These results indicate how Mn plays an important role in the formation of bone tissue and in the increase of BMD, suggesting its possible use in the treatment of postmenopausal osteoporosis. 12
That Mn can stimulate the growth of osteoblasts and bone regeneration has also emerged from in vitro studies on bioactive glass, in which it has been observed how the addition of Mn is able to promote, in cultures of human osteoblasts, the expression of alkaline phosphatase (ALP) and some bone morphogenetic proteins (BMPs), 13 and to stimulate the osteogenetic differentiation of human mesenchymal stem cells (hMSCs) and influence the mineralization process by increasing the expression of markers of osteogenetic differentiation, such as type I collagen, osteopontin and osteocalcin. 14
Daily Intake of Manganese
Mn is one of the most abundant metals on the earth’s crust 15 ; it occurs naturally in surface and underground waters, especially in conditions of anaerobiosis or low oxidation, and in numerous foods, which are the main source of Mn for humans. 16
Nuts, chocolate, cereal products, crustaceans, molluscs, legumes and fresh fruit are foods rich in Mn; the greatest contribution to the daily intake of this element is provided by whole grains and derivatives, vegetables and fruit, while dairy products, meat and fish provide minimal amounts.
Among drinks, tea is particularly rich in Mn, while 2 L/day of drinking water provide on average only 40‐64 μg of Mn (corresponding to about 2% to 3% of the quota supplied by food) 15,17,18 The absorption of Mn from foods introduced with the diet is quite low, about 3% to 4%, 15 and occurs in the small intestine with an active mechanism and by passive diffusion. 19
In humans, it has been reported that Mn deficiency can be associated with hypocholesterolemia, bone demineralization and reduced growth in children; however, to date, the deficiency has not been clearly correlated with reduced diet intake. 20 Since Mn is an abundant element in food, especially of plant origin, the consumption of a varied and balanced diet will be reasonably sufficient to ensure the proper contribution of this microelement. 18 On the basis of these observations, together with the uncertain validity of the studies carried out to determine the need for Mn, in 1989 a daily dietary intake for adults equal to 2.0‐5.0 mg of Mn was provisionally recommended. 18 In 2013, the European Food Safety Agency (EFSA), judging the available data still insufficient to be able to derive an average requirement (Average Requirement, AR) and a reference intake for the population (Population Reference Intake, PRI) for Mn, proposed an adequate intake (Adequate Intake, AI) for adults (including pregnant and breastfeeding women) equal to 3.0 mg/day (considering it is not necessary to provide sex-specific values) based on the average contributions observed in the countries of the EU. 17 The Italian Society of Human Nutrition (SINU), on the other hand, in the IV Review of LARN (Reference Assumption Levels of Nutrients and Energy for the Italian population), established for adulthood, based on the median value of the content of Mn emerged from studies conducted on the total diet in Italy, an AI equal to 2.7 mg Mn/ day for males and 2.3 mg/day for females. 20
Toxicity phenomena (specifically neurotoxicity) in humans have been observed only in workers professionally exposed to high concentrations of Mn in inhaled dusts and fumes, but not as a consequence of its high intake through diet; an occasional intake of 10 mg/day was considered safe for humans. 18 However, considering the available data concerning humans insufficient and not definitive, EFSA has not established any tolerable upper intake Level (UL) for Mn. 2 Sharing the recommendations of EFSA, and also the SINU, the IV Review of the LARN did not consider it possible to indicate a UL for Mn, either for the various age groups or for pregnancy and breastfeeding. 20
The constancy found in the concentration values of Mn in different human tissues and in different ages of life seems to suggest an adequate dietary intake together with a strong homeostatic control by the human body. 18
From the data collected through national dietary questionnaires, duplicate diet studies, total diet studies and market basket studies in several European countries, an estimated average daily intake of Mn between 2 and 6 mg in adults emerges, with most values around 3 mg/day; inter-individual variations may be due to the characteristics and dietary habits of the individual subject (eg., vegetarian diet vs mixed diet). 17
In a recent Italian study, after determining, through inductively coupled plasma mass spectrometry, the Mn content of different foods and drinks usually consumed by the population, the average daily intake was estimated in a sample of inhabitants of the region Emilia-Romagna (719 subjects, 319 males and 400 females, aged between 18 and 87 years). Each participant was subjected to a validated, semi-quantitative Food Frequency Questionnaire (FFQ), specifically developed for the Italian population of Central and Northern Italy. From the data collected, an average intake of 2.34 mg Mn/day emerged, with the major sources represented by cereals, vegetables, legumes (in particular chickpeas, soy and beans), sweets (especially dark chocolate) and drinks such as coffee and tea. 21 This value appears in line with what is estimated, in the United States, through the Total Diet Study (average daily dietary intake of Mn equal to 2.7 mg for men and 2.2 mg for adult women) 22 and with the figure calculated for the French adult population (2.5 mg/day, with 75% of the total Mn supplied by cereals and vegetables). 15 From a duplicate meal study conducted in Belgium, however, higher average daily intake values emerged (3.1 mg Mn/day, SD 1.8; range 0.6‐8.8), 23 as well as from a total diet study that investigated the intake of Mn through the diet in the United Kingdom (approximately 4.6 mg/day), where almost half of this microelement was found to derive from drinks, especially from tea (with a further 30% coming from cereals and derivatives and about 15% from vegetables and fruit). 24
Given this background, the aim of this narrative review was to consider the state of the art on the relation between Mn and bone health in humans and the effectiveness of Mn supplementation (alone or with other micronutrients) on bone mineralization.
Results and Discussion
Manganese and Bone: Human Studies
Several authors have investigated the possible relationship between Mn and bone health in humans. Osteoporotic women, for example, showed significantly lower serum Mn levels than healthy subjects (0.02, 0.04 mg/L, respectively). 25 In a prospective, placebo-controlled, double-blind study, Saltman & Strause then assessed the importance of calcium supplementation (Ca, 250 mg in the form of a citrate-malate complex 4 times/day) with or without the addition of copper (Cu, 5.0 mg), Mn (Mn, 2.5 mg) and zinc (Zn, 15 mg) (collectively referred to as TMIN, trace minerals, and contained in a single tablet taken once/day) in a sample of 137 healthy Caucasian postmenopausal women over 50 years of age (mean age 64.6 years, SD 7). The subjects were divided into 4 groups: placebo, integration with only Ca, integration with only TMIN and integration with Ca +TMIN. After 2 years of supplementation, a greater loss of lumbar BMD (L2-L4) was observed in the placebo group in women not undergoing hormone replacement therapy (n = 76), with significantly different values compared to the baseline and with the only difference between statistically significant groups found between placebo and Ca +TMIN. As for women undergoing estrogen therapy during the study, however, no significant changes in bone mineral density were detected in any of the 4 treatment groups. Finally, considering the cohort as a whole, bone loss was significantly greater in the placebo group and in the group with only TMIN supplementation compared to the Ca +TMIN group. The merit of this study was to extend from the animal model to humans the evidence of an essential role of Cu, Mn and Zn in the maintenance of BMD, even if each element has not been investigated individually. 25
A study very similar to the one just described was conducted by the same authors on 59 post-menopausal women over 50 years of age (average age 66 ± 7 years), not on estrogen therapy, also divided in this case into 4 groups: placebo (n = 18); only supplemented with 1000 mg of calcium citrate malate/day (n = 13); only integration with TMIN, comprising 15.0 mg Zn, 2.5 mg Cu and 5.0 mg Mn/day (n = 14); Ca + TMIN (n = 14). After a period of 2 years, the loss of BMD at the L2-L4 level (assessed by DXA) was greater in the placebo group, with a significant difference compared to the baseline only in that group. By comparing the 4 groups with each other, Ca +TMIN proved to be the only treatment to differ significantly from placebo in the change in lumbar bone density. These results highlight how the lumbar bone loss observed in post-menopausal women receiving calcium supplementation can be further slowed through the simultaneous increase in trace mineral intake. 26
That serum Mn levels may be related to bone quality and density in postmenopausal women was also suggested by a prospective cross-sectional pilot study of 41 postmenopausal women who received no treatment, 21 of whom had osteoporosis (lumbar and/or femoral T-score ≤−2.5), in which the blood concentrations of some trace elements were investigated, including Mn, Cu, magnesium (Mg), Zn and cadmium (CD). The analysis of the collected data revealed a significant positive correlation between serum Mn levels and lumbar or femoral BMD and a negative correlation between Cu levels and BMD at both measurement sites. On the contrary, a respectively negative and positive correlation was found between the serum levels of Mn and Cu and the number of fractures, but no significant relationship between the concentrations of Mg, Zn and Cd and bone parameters. This study has therefore shown how the concentrations of manganese and copper in serum have a certain predictive value toward lumbar and femoral BMD, as well as bone quality; but considering the small number of participants, these results should be confirmed by further investigations. 5,27
In a study of 100 subjects between 30 and 60 years of age (average age 41.2 ± 4.9 years, 47 women and 53 men), of which 30 normal controls (T-score between 0 and −0.99), 40 osteopenic (T-score between - 1 and −2.49) and 30 osteoporotic (T-score ≤−2.5), in addition, significantly lower serum levels of calcium, Mn, Cu, and Zn were found in osteopenic and osteoporotic subjects compared to controls. Furthermore, following a 12 week period of moderate aerobic physical activity (1 hour/day for 3 days/week), a significant increase in serum Ca and Mn concentrations and a decrease in serum concentrations of Cu and Zn were observed in all 3 groups. These changes were significantly correlated with the increase in serum bone alkaline phosphatase (sBAP) levels and with the improvement of BMD values (detected by DEXA) at both femoral and vertebral levels. These observations seem to confirm the essential role of some trace elements, including manganese, in the synthesis of cartilage and bone collagen, as well as in bone mineralization. Furthermore, moderate aerobic exercise could be protective for bone and cartilage by regulating the body levels of these trace elements, involved in the biosynthesis of matrix structures and in the inhibition of bone resorption processes. 28
In the study by Friedman and colleagues in 1987, the manganese balance and clinical effects of a diet poor in this element had been investigated in a group of 7 male subjects aged between 19 and 22 years. The study protocol included an initial 21-day period characterized by a baseline diet, with a daily intake of 2.59 mg of Mn, followed by 39 days of a “purified” diet containing 0.11 mg Mn/day (depletion period) and subsequently by 2 periods of 5 days each with a progressively higher daily intake of Mn (1.53 mg the first 5 days and 2.55 mg/day the last 5; repletion period). The participants showed a slightly negative Mn balance during the depletion period, which then turned positive in the 10 following days of Mn reintegration. Furthermore, a significant increase in serum concentrations of calcium, phosphorus and alkaline phosphatase (ALP) had been found in the 39 days of the Mn-poor diet, suggesting that the deficiency of Mn could influence bone remodeling. 25,29
Finally, a clinical trial conducted in Mexico in 2001 demonstrated how a multiple supplement with vitamins and microelements, including Mn, {administration of a drink containing the recommended daily dose -RDA- of vitamins D3, E, K1, B6, niacin, thiamine, biotin, folic acid, pantothenic acid and minerals (iodine, Cu, Mn and selenium); 1.2 times the RDA of vitamin A and 1.5 times the RDA of ascorbic acid, riboflavin, vitamin B12, iron and Zn)} given 6 days/week for an average period of 12.2 months, is able to improve growth in length (with a gain of almost 5 mm) in children aged between 8 and 14 months compared to the placebo group . In particular, the effect of supplementation was greater in children less than 12 months of age at the start of the study (average increase in length of 8.3 mm) compared to children ≥ 12 months (average increase of 2.0 mm). 30
Unfortunately, to date, the available data are insufficient to establish an AR and a PRI for Mn; for European countries, however, the EFSA (European Food Safety Agency) has proposed an AI for adults equal to 3 mg/day. 17 Similarly, the SINU, in the IV revision of the LARN (Reference Assumption Levels of Nutrients and Energy for the Italian population), has established, for the adult Italian population, an AI equal to 2.7 mg/day for males and 2.3 mg/day for females. 20 Furthermore, currently, again due to the scarcity and inadequacy of the available data, a tolerable Upper intake Level of Mn has not yet been established. 2,20
Research conducted in several European countries has shown an estimated average daily intake of 2‐6 mg, with most values around 3 mg/day, 17 values which are in line with what is also estimated in a recent Italian study (average adult intake of 2.34 mg Mn/ day). 21
All the literature published to date is in agreement in showing that osteoporotic women have lower serum Mn levels than women with normal bone mineral density), 25 thus confirming the essential role of manganese in the synthesis of cartilage and bone collagen, as well as in bone mineralization 5,27 and confirming the studies on the animal model.
Conclusion
Considering the human studies that evaluated the effectiveness of an oral manganese supplement for a long period (2 years) on the bone mineral density of menopausal women (in total 196 women), both of the double blind clinical trials that evaluated this topic showed that bone loss was significantly greater in the placebo group than in the group taking supplementation, equal to 5.0 mg Mn/day for the Straus study, 26 and 2.5 mg Mn/day for the study of Saltman et al., 25 but considering, however, that supplementation was represented by a set of microelements (Mn, Cu and Zn) and by calcium.
Material and Methods
The present narrative review was performed following the steps by Egger et al. 31 as follows:
Configuration of a working group: 3 operators skilled in clinical nutrition (one acting as a methodological operator and 2 participating as clinical operators).
Formulation of the revision question on the basis of considerations made in the abstract: “the state of the art on the relation between Mn and bone health in humans and the effectiveness of manganese supplementation (alone or with other micronutrients) on bone mineralization”
Identification of relevant studies: a research strategy was planned on PubMed (Public MedIine run by the National Center of Biotechnology Information (NCBI) of the National Library of Medicine of Bethesda (USA)) as follows: (a) Definition of the keywords (manganese, bone health, humans, supplementation, bone mineral density), allowing the definition of the interest field of the documents to be searched, grouped in quotation marks (“…”) and used separately or in combination; (b) use of: the Boolean (a data type with only 2 possible values: true or false) AND operator, that allows the establishments of logical relations among concepts; (c) Research modalities: advanced search; (d) Limits: time limits: papers published in the last 20 years; humans; adults; languages: English; (e) Manual search performed by the senior researchers experienced in clinical nutrition through the revision of articles on the state of the art on the relation between manganese and bone health in humans and the effectiveness of manganese supplementation (alone or with other micronutrients) on bone mineralization
Published in journals qualified in the Index Medicus.
Analysis and presentation of the outcomes: we create paragraphs about the state of the art on the relation between manganese and bone health in humans and the effectiveness of manganese supplementation (alone or with other micronutrients) on bone mineralization and the data extrapolated from the “revised studies” were collocated in tables; in particular, for each study we specified the author and year of publication and study characteristics.
The analysis was carried out in the form of a narrative review of the reports. At the beginning of each section, the keywords considered and the type of studies chosen are reported. We evaluated, as is suitable for the narrative review, studies of any design which considered the state of the art on the relation between manganese and bone health in humans and the effectiveness of manganese supplementation (alone or with other micronutrients) on bone mineralization
Table 1 shows studies regarding manganese and bone health in humans.
Studies Regarding Manganese and Bone Health in Humans.

Figure 1 shows flow chart of literature research.
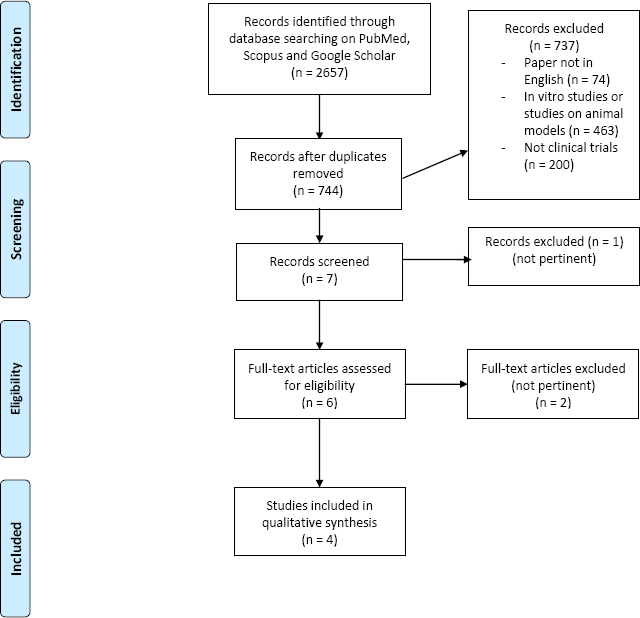
Flow chart of literature research
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
