Abstract
Recently, natural ingredients have focused on the inhibition of bacteria-induced alveolar bone resorption in orthodontic treatment. Jixueteng (Jix), a Chinese traditional medicine, contains several kinds of flavonoids given their biological properties. We evaluated the effects of Jix on experimental periodontitis during orthodontic tooth movement (OTM) in rats. To this end, 9-week-old male Wistar rats, which were equipped with orthodontic appliance, were orally infected with Porphyromonas gingivalis (Pg), while Jix was administered in their drinking water. A total of 28 days after the beginning of OTM, alveolar bone resorption on the right side of the upper jaws was scanned with micro-computed tomography. These were also used as histological specimens and underwent tartrate-resistant acid phosphatase (TRAP) staining. TRAP-positive multinucleated cells were counted as osteoclasts. As a result, the distance of tooth movement in the OTM and Pg infection with Jix administration (OTM + Pg + Jix) group was the same as that of the sham-infected group. The amount of bone resorption and number of osteoclasts in the OTM + Pg + Jix group was more significantly decreased than that in the OTM and Pg-infected group (P < 0.05). Hence, Jix had little effect on OTM and inhibited Pg-induced alveolar bone destruction. We suggested that the administration of Jix can support tooth movement and contribute to the prevention of periodontitis during orthodontic treatment.
Orthodontic treatment has been linked to the improvement of malocclusion and facial esthetics. In orthodontic treatment, a mechanical force applied to teeth induces an inflammatory response with tooth movement and results in bone resorption on the compression side and osteogenesis on the tension side of the periodontium. 1 -7 Osteoclasts and osteoblasts play a critical role on orthodontic tooth movement (OTM). Osteoclast differentiation is regulated by the binding of receptor activator of nuclear factor-kappa B ligand (RANKL) of osteoblasts derived from stromal stem cells and RANK, a receptor on the surface of osteoclasts and their precursor cells, derived from macrophages. 8 Comparing to the tension side of periodontium, osteoclast formation is induced by the activation of RANKL/RANK signaling on the compression side.
Recently, the rising number of elderly patients needing orthodontic treatment continuously has led to a need to better prepare for prosthodontic treatment or prevent the occurrence of periodontitis. 2 Periodontitis is an infectious disease that causes a pathological inflammatory reaction in the periodontium. The primary etiological agent of periodontitis has been identified as oral bacteria in dental plaque. More than 700 species of oral bacteria colonize on the tooth surface and form dental plaque. 9 Fixed orthodontic appliance, which consists of orthodontic brackets and arch wires, is commonly used for tooth movement in orthodontic treatment. Bacterial cells adhere to the brackets and arch wire increase plaque accumulation and gingival inflammation that make it difficult to keep clean in the oral cavity. 2,10,11 Porphyromonas gingivalis (Pg), an obligate anaerobic gram-negative bacterium, is one of the keystone pathogens in the development of periodontitis. 9,12 This bacterium is frequently detected from human periodontal pockets. In particular, the prevalence of Pg in elderly patients is significantly higher than that in young patients. 12 Pg has various virulence factors on alveolar bone resorption and induces inflammatory responses in the periodontium. 12 -14 The inflammatory cytokines are also detected from gingival crevicular fluid on OTM. 6 The inflammatory reaction caused by periodontal bacteria during OTM will expand root resorption, alveolar bone destruction, and gingival regression. 2,7 Therefore, the control of inflammation is important to maintain periodontal health during OTM.
To reduce the bacterial infection on periodontium, chemical agents have been used with mechanical plaque removal. 15 -20 The prolonged use of chemical agents is known to induce side effects 18,21 and the presence of resistance bacteria. 18,20 Herbal medicine, which consists of natural products with medical properties, is known to have low cytotoxicity to the human body. 22,23 Herbal medicine also has great bactericidal effects on oral bacteria. 23,24 Jixueteng (Jix) specified in Chinese Pharmacopoeia derives from the dried stems of Spatholobus suberectus (S. suberectus) Dunn of the family Leguminosae that improves blood circulation 25,26 and has an antioxidant effect. 27,28 Previous studies have reported that Jix has a strong bactericidal effect against Pg and suppresses osteoclast differentiation on alveolar bone resorption. 29 However, little is known with regard to the influence of Jix on OTM. In the present study, we investigated the effect of Jix on experimental periodontitis during OTM in rats.
Results and Discussion
In Figure 1(A) and (B), the increasing of alveolar bone resorption and osteoclasts induced by Pg inoculation were confirmed. Jix administration was not related bone resorption. To investigate the inhibitory effect of periodontitis on adult orthodontic treatment, the following experiments were performed according to the time schedule shown in Figure 2.

Effect of Jix on alveolar bone resorption without an orthodontic appliance. (

Experimental design. Rats were divided into 4 groups (n = 5/group). Arrow heads indicate infection timing with Pg. Arrows indicate experimental orthodontic tooth movement.
Inhibitory Effect of Jix on Alveolar Bone Resorption
The body weight of the animals was not affected by Pg infection and orthodontic force until the final day in this experiment. Figure 3 shows the bone levels of cemento-enamel junction (CEJ) to alveolar bone crest (ABC) in all groups. The increasing distance between CEJ to ABC reflects the loss of alveolar bone volume. OTM was increased the levels of alveolar bone resorption. The bone volume of the OTM + Pg group was 2.6 times greater than that of the OTM group (P < 0.05). However, the bone volume of the OTM + Pg + Jix group was 0.7 times lower than in the OTM + Pg group (P < 0.05). No significant difference was found between the OTM + Pg + Jix group and the OTM group. Alikhani M et al. reported that the applied force on OTM increase the several kinds of inflammatory cytokine production. 30 In our experiment, OTM may promote of the inflammatory cytokine productions from chemokines and activate osteoclast formation. Jix, the extract of S. suberectus Dunn, contains several types of isoflavones extracted from soybeans such as formononetin, genistein, daidzein, ononin, and medicagol. Isoflavones, a large family of polyphenols, are used as an antioxidant that have bactericidal and anti-inflammatory effects. 31,32 Formononetin and daidzein have a bactericidal effect on Pg. 33,34 Genistein and ononin inhibit the inflammatory cytokine production induced by lipopolysaccharide (LPS). 35 -37 LPS, one of the bacterial virulence factors on Pg, promotes osteoclast formation and increase inflammatory legions. Previous studies have been reported showing that some herbal medicines inhibit the growth on Pg 23,24,29,38 and proteolytic enzyme production on this bacterium. 38 In our results, Jix was found to have possibly decresed the amount of Pg and inhibited its-induced inflammation. The administration of herbal medicine may reduce the bacteria-induced inflammatory response and normalize the oral environment. 39 Results demonstrated that alveolar bone resorption-induced Pg infection was suppressed by Jix administration.
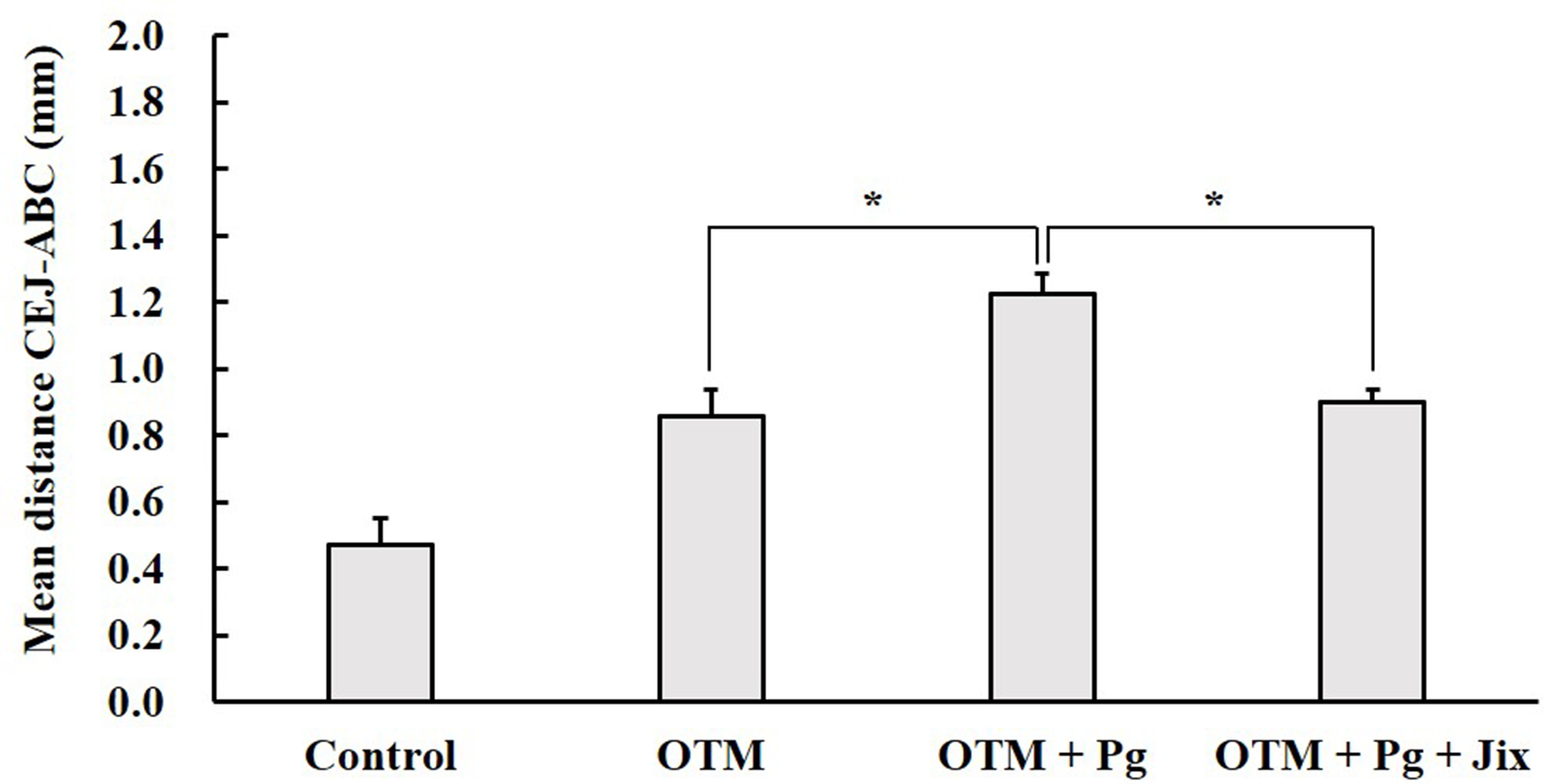
Effect of Jix on alveolar bone resorption with an orthodontic appliance. The bone levels of CEJ to ABC were measured. The increasing mean distance between CEJ to ABC indicates the loss of bone volume. n = 5/group. *P < 0.05.
Inhibitory Effect of Jix on Osteoclast Formation in Periodontium
Figure 4(A) represents tartrate-resistant acid phosphatase (TRAP) staining images of the buccal distal root of the upper right first molar. TRAP-positive cells with more than 3 nuclei were defined as osteoclasts. In the control group, few osteoclasts were observed (Figure 4(A)). In the OTM group, osteoclasts were slightly detected on the surfaces of alveolar bone and distal root (Figure 4(A)). OTM and Pg infection was induced by the presence of a number of osteoclasts. Moreover, the lacuna of bone resorption and root resorption was observed (Figure 4(A)). Root resorption, which is one of side effect in orthodontic treatment, causes demineralize of the dentin and cementum leading to the loss of root apex. The excessive orthodontic force is mainly risk factor of root resorption. In this study, the areas of root resorption were slightly observed that received 0.25N (25 g) force at day 35. Generally, orthodontic force correlates the surface area of root surface and tends to indicate different histological responses in animal species. A previous study revealed that a force of 10g to 25g is an ideal orthodontic force to promote OTM and rat root resorption was little observed. 30 Therefore, we suggested that the value of orthodontic force used in this study was appropriate to promote tooth movement normally. In the OTM + Pg + Jix group, the distribution of osteoclasts resembled that of the OTM group (Figure 4(A)). As shown in Figure 4(B), the number of osteoclasts was significantly increased by OTM and Pg infection (P < 0.05). However, the number of osteoclasts in the OTM + Pg + Jix group was decreased to one-third of that in the OTM + Pg group (P < 0.05). The number of osteoclasts in the OTM + Pg + Jix group was nearly the same as that of the OTM group. OTM and Pg infection was increased the number of TRAP-positive multinucleated cells on the external bone surface. Adding the orthodontic force in tooth, increasing of the vascular permeability in periodontal ligament promotes migration of mononucleated osteoclast precursor cells to the alveolar bone surface and induce the formation of multinucleated giant cell. 40 Especially, these cells are formed rapidly multinucleated giant cell in the compression side. 41 Our data indicate that the TRAP-positive cells increasing by OTM and Pg infection changed to multinucleated cells, and the cell size were greater than that of internal surface of bone. Besides, Jix administration reduced the number of TRAP-positive cells on external bone surface by the inhibition of Pg infection. A previous study has reported that a 0.1% concentration of Jix inhibits osteoclastogenesis, mostly in the presence of RANKL. 29 Some herbal medicines regulate RANKL and osteoprotegenin signaling and suppress osteoclast formation. 42,43 On the other hand, formononetin, genistein and daidzein individually induce the inhibition of osteoclastogenesis on the RANK-RANKL pathway or inflammatory cytokine-induced cascade. 44,45 The synergistic effect of formononetin, genistein and daidzein can be expected to reduce the excessive osteoclastogenesis induced on inflammatory bone resorption.
Effect of Jix on OTM
The micro-computed tomography (micro-CT) images of the upper first right molar moved by OTM are shown in Figure 5. Table 1 represents the mesial distance of tooth movement on the upper right first molar analyzed by micro-CT. In the control group, no tooth movement was observed. The extent of tooth movement in the OTM + Pg group was 1.6 times higher than that in OTM group (P < 0.05). The extent of tooth movement in the OTM + Pg + Jix group was significantly decreased, to 0.5 times than that in the OTM + Pg group (P < 0.05). The extent of tooth movement in the OTM + Pg + Jix group was similar to that of the OTM group. To inhibit pathological bone resorption, bisphosphonates and anti-RANKL antibody, which has been used mainly for the treatment of osteoporosis, induces osteoclast apoptosis. However, bisphosphonates and anti-RANKL antibody inhibits tooth movement and affects the improvement of malocclusion in the orthodontic treatment. 46,47 Some herbal medicines have strong effects on osteoporosis and increase bone mass. 48 In this research, Jix administration affected the extent of tooth movement. A previous study suggested that Jix can improve the infected-harmful vascular networks and promote blood flow mass in the gingiva. 26 To induce the bone mass and tooth movement applied by an orthodontic force, Jix may stimulate angiogenesis and activate bone metabolism.

Effect of Jix on osteoclast formation in orthodontic tooth movement. (

Micro-CT images of the upper first right molar after 28 days on OTM.
The Distance of Tooth Movement With Pg Infection and Jix Administration on OTM.

These data represented mean ± SD (n = 5/Group), *P < 0.05.
Isoflavones, which have unique chemical structures, point to the estrogenic effect on bone formation. 49,50 Formononetin enhances osteoblastic differentiation on bone formation 51,52 and regeneration. 53 It can bind estrogen receptors and promote angiogenesis. 54 Besides, genistein increases alkaline phosphatase activity and affects the expression of calcification-related genes in the hard tissue. 55 In addition, genistein and daidzein also induce osteoblastic differentiation and angiogenesis on bone formation. 56 -58 In the present study, Jix inhibited Pg infection and decreased alveolar bone resorption. Osteoblasts appear on tension side to promote new bone formation. Jix may contribute to promote bone formation at tension side. A future study will seek to examine the effects of Jix-induced angiogenesis and osteoblast function on tooth movement at tension side.
This research focused on the efficacy of Jix in avoiding periodontal pathogens in the context of orthodontic treatment. Affecting the activity of tooth movement and bone metabolism by Jix, the term of orthodontic treatment may be completed faster than usual. Therefore, Jix will be expected to be used as a natural extract for the stability of tooth movement and infection control in the context of orthodontic treatment.
Conclusions
Our results revealed that the oral administration of Jix was not affected OTM. Jix reduced the number of osteoclasts and inhibited alveolar bone resorption caused by Pg infection. Our findings indicated that Jix reduced bacteria-induced bone resorption during OTM. In addition, we suggested that Jix is a natural ingredient contributing to the prevention of periodontitis during orthodontic treatment.
Experimental
Preparation of Jix Extract
Dried vines of S. suberectus Dunn (Tochimototenkaido, Osaka, Japan) were used in this study. Jix extract was solved by distilled water and was finally designed such as to have an 8% concentration. 26,28,29
OTM on Pg-Induced Experimental Periodontitis
Pg American Type Culture Collection 33277 was cultivated as described previously. 26,29,56 The experimental procedures of this study were reviewed and approved by the Committee of Ethics on Animal Experiments of Kanagawa Dental University (No. 18‐046). Forty 9-week-old male Wistar rats (CREA Japan, Tokyo, Japan) were used in this study. The animals were fed powdered standard pellets diet (MF; Oriental Yeast, Tokyo, Japan) throughout the experimental period. First, to confirm the inhibition effect of Pg infection on Jix administration without OTM, twenty rats were randomly divided into 4 animal groups without an orthodontic appliance such as control group (sham-infected control), Pg group (Pg-infected group), Jix group (Jix extract administration group), and Pg +Jix group (Jix extract administration group with Pg-infection). This experiment was performed by according to a previous study. 29 Next, to evaluate the inhibitory effect of Pg-induced alveolar bone resorption during OTM, the remaining 20 rats were randomly divided into 4 groups as follow. Sham-infected groups consisted of the distilled water administration group without an orthodontic appliance (Control group), and the distilled water administration group with an orthodontic appliance (OTM group). Pg-infected groups consisted of the distilled water administration group with an orthodontic appliance (OTM +Pg group) and the Jix extract administration group with an orthodontic appliance (OTM +Pg + Jix group). Experimental periodontitis was performed according to previous reports. 26,29,59 Figure 1 shows the outline of the experimental design. Briefly, the rats were given antibiotics in their drinking water to reduce original oral microorganisms for 4 days. Each rat orally received 0.5 ml (2.1 × 1011 cells/mL) of the bacterial suspension in 5% carboxymethylcellulose (CMC; Sigma-Aldrich) at 8, 11, 13 and 15 days. Sham-infected groups (Control and OTM) orally received only 5% CMC. A total of 7 days after the start of the experiment, a nickel-titanium closed coil tension spring (0.25 n; Tomy International, Tokyo, Japan) was used as an orthodontic appliance in rat oral cavities, except in the control group. The animals were treated under general anesthesia using a combination of pentobarbital sodium (Kyoritsuseiyaku, Tokyo, Japan) and isoflurane (Wako Pure Chemical Industries, Osaka, Japan). The orthodontic appliance was fixed between the upper incisors and the upper right first molar using wire ligature (ø 0.25 mm; Ormco, Orange, CA, USA) and dental composite resin (Tokuyama Dental, Ibaraki, Japan).
Measurement of Alveolar Bone Resorption by Micro-Computed Tomography
A total of 28 days after the start of tooth movement, the animals were sacrificed and the right side of the upper jaw was removed under general anesthesia. The upper jaw was scanned with a micro-CT system (ScanXmate-L080; Comscantecno, Kanagawa, Japan). The image of the upper jaw was obtained at 80 kV and 100 µA, with an image pixel size of 67.36 µm. The software used for measurement was OsiriX Lite (Newton Graphics, Inc., Hokkaido, Japan). The distance between CEJ to ABC was measured at 5 sites of the mesial root and distal root on the upper right first molar.
Histological Analysis
After micro-CT scanning, the right sides of the maxilla were used for histological analysis, as described previously. 29,59 The sections of the maxilla were stained with TRAP (TRAP Staining Kit; Cosmo Bio, Tokyo, Japan), performed in accordance with the manufacturer’s instructions. Red-stained multinucleated (TRAP-positive) cells were defined as osteoclasts and counted in the compression side of distobuccal root of the upper right first molar under an optical microscope (200 × magnification, BX53, OLYMPUS, Tokyo, Japan).
Statistical Analysis
All data are shown as mean ± standard deviation (SD). The software EZR (Ver.1.40, Jichi Medical University Saitama Medical Center, Saitama, Japan) was used for all statistical analyses. 60 The results were analyzed by Kruskal–Wallis-H tests with Steel-Dwass post hoc tests. A P value of less than 0.05 was defined as statistically significant.
Footnotes
Statement of Human and Animal Rights
All procedures in this study were conducted in accordance with the Committee of Ethics on Animal Experiments of Kanagawa Dental University (No. 18-046) approved protocols.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by JSPS KAKENHI Grant Number JP17K18193.
