Abstract
Repeated exposure to ionizing radiation has been reported to increase the risk of chronic metabolic disorders such as systemic hyperlipidemia and intracellular lipid accumulation that might lead to diabetes-induced heart disease. The purpose of this study was to investigate the effect of pterostilbene on high-fat diet rats suffering from ionizing radiation-induced hyperlipidemia. High-fat diet rats showed an increase in body weight and body fat compared with rats fed with normal chow. Pterostilbene and Orlistat treatments resulted in lower body weight and body fat gain, insulin resistance, reduced lipid peroxidation with attenuated liver enzyme levels, and regulated lipogenesis-related genes in the HFD + IR rat group. Regulation of Peroxisome proliferator-activated receptor-γ (PPAR-γ) mRNA enhanced paraoxonase-1 (PON-1) and arylesterase (AE) activities and inhibited that of 3-hydroxy-3-methylglutaryl coenzyme A reductase (HMG-CoA). It also increased the activities of plasma lecithin-cholesterol acyltransferase (LCAT) and lipoprotein lipase (LPL). Pterostilbene and Orlistat also corrected the alterations of serum leptin and adiponectin levels in lipidemic rats. Such findings provide evidence that Pterostilbene and Orlistat can act as normolipidemic agents that possess lipid-lowering effects and potential as a radioprotector.
Radiation therapy for multiple diseases has significant side-effects on healthy tissues and a wide number of metabolic diseases are encountered by survivors of long-term cancer treatment. 1 -4 Generation of reactive oxygen species is the main mechanism of action of radiotherapy, inducing damage to cell structural and functional components, including proteins, lipids, and DNA. Mutations caused by ionizing radiation (IR) alter fat metabolism in tandem with the accumulation of fat in rat adipose tissue. 5 On the same line, ionizing radiation reprogrammed skeletal muscle and adipose progenitor cells, leading to altered differentiation and subsequent alterations in cell function and whole-body glucose homeostasis when tested in a high-fat diet (HFD) model. 6 Wellen & Hotamisligil 7 and Wang et al. 8 stated that the inflammatory response could be attributed to the up-regulation of reactive oxygen species after exposure to ionizing irradiation, which can cause accumulation of lipid in liver tissue leading to inflammatory cytokine production and insulin resistance. Ionizing radiation caused an increased expression of genes related to lipid and glucose metabolism and fatty acid synthesis. Lipid metabolism involves different cell types, such as pre and mature adipocytes, vascular cells, and macrophages; the last secrete adipokines and cytokines that act in harmony via a variety of signaling pathways, resulting in deregulation of gene expression in adipose tissue. 9 For example, the major adipokines formed by adipocytes are adiponectin and leptin; adiponectin is an anti-inflammatory cytokine that acts mainly through modulating the phagocytic potential of macrophages; in contrast, leptin is a pro-inflammatory cytokine whose main functions are modulating appetite, angiogenesis, immunity, and neuroendocrine circulation. 10 The activities of the main lipid metabolizing enzymes were previously reported to be affected by ionizing irradiation. The first of these is HMG-CoA reductase, the rate-limiting step in cholesterol synthesis. The second is acyl-CoA cholesterol acyltransferase (ACAT) and lecithin cholesterol acyltransferase (LCAT), and, thirdly, cholesterol 7-hydroxylase, which is the rate-limiting enzyme in cholesterol catabolism to bile acid low-density lipoproteins (LDL) receptor, high-density lipoproteins (HDL) receptor, and scavenger receptor class B type 1 (SRB-1). 11,12 In correlation with the above, Meneses et al. 13 stated that PON-1/ARE-1 possesses antioxidant properties and is closely associated with HDLs, which detoxify lipid peroxides and protect LDLs from oxidation. Besides, Phua et al. 14 noted that peroxisome proliferator-activated receptor (PPAR) controls adipocyte differentiation, glucose metabolism, and lipid homeostasis. PPAR is a ligand-binding transcription factor that belongs to the nuclear receptor superfamily, expressed throughout the body. Furthermore, PPAR-γ has been reported to regulate the expression of genes involved in lipid metabolism, inflammatory responses, and other biological processes. 15 Orlistat is one of five approved drugs by the US Food and Drug Administration (FDA) for the long-term management of obesity via the reduction of fat absorption from the gastrointestinal tract by inhibiting pancreatic and gastric lipases, and so encourages weight loss. However, some side effects can affect its use, such as fecal urgency, oily stools and fecal incontinence; Orlistat also has the potential to affect the absorption of other drugs. 16 In the present study, Orlistat was used as a reference standard in order to evaluate the comparable effect(s) of Pterostilbene aiming to encourage the use of natural products rather than synthetic drugs to avoid their side effects. Pterostilbene (PS), a resveratrol analog, is a compound derived from fruits such as blueberries and Pterocarpus marsupium. Substantial evidence suggests that PS may have many preventive and therapeutic properties (eg, antioxidant, anti-inflammatory, and anticancer) in a vast range of human diseases. 17 Compared with resveratrol and other stilbenes, the 2 methoxy groups of Pterostilbene result in improved bioavailability, with improved lipophilic and oral absorption. 18 Research conducted by Sun et al. 19 showed that PS treatment of diabetic rats might alleviate the metabolic complications of hyperinsulinemia, hyperglycemia, and apoptosis reduction. Zhang et al. 20 revealed that PS has anti-atherosclerosis defense potential, which enhances aspects of autophagy and helps to mitigate the pro-atherosclerosis impact of oxidized LDL. Over the past few decades, some survivors of cancer chemoradiotherapy have an increased incidence of obesity and type 2 diabetes mellitus with significant metabolic disturbance conditions related to late effects of treatments. Some agents (eg, N-acetyl cysteine, vitamin C, R-α-lipoic acid, quercetin-3-O-rhamnoside-7-O-glucoside, carvacrol, GANRA-5) have the potential to fight various illnesses, with potential use in the treatment of metabolic disorders caused by radiotherapy. 21 In the current study, we focused on the potential of pterostilbene’s anti-dyslipidemic effect on 2 stress factors, obesity with irradiation disturbance impact. As adipose tissue dysfunction widely affects metabolism throughout the body and the functions of the vital organs, we aimed to gain insight into the role of pterostilbene on irradiation-induced dyslipidemia when challenged with HFD, which alters metabolic progenitor cells with consequent alterations in lipometabolism factors.
Materials and Methods
Chemicals and Reagents
PS was obtained from Source Naturals (Scotts Valley, CA, USA). Orlistat and all other chemicals were purchased from Sigma–Aldrich (St. Louis, MO, USA).
Irradiation Process
Whole-body gamma irradiation was performed at the National Centre for Radiation Research and Technology (NCRRT, Cairo, Egypt) using Canadian gamma cell-40 (137Cesium) at a dose rate of 0.67 Gy•min−1 for a total dose of 6 Gy. 22
Animals
Sixty-four male albino Wistar rats, 5 weeks-old, weighing 200 to 220 g, from the breeding unit of the Egyptian Holding Company for Biological Products and Vaccines (Giza, Egypt) were housed in standard cages (a cage 1,800 cm2 floor area with a height of 22 cm). Six animals per cage with free access to water and standard laboratory diet (El-Nasr Co. Cairo, Egypt) were maintained in an air-conditioned (25 ± 2 °C) animal house under standard conditions of humidity (50 ± 5 %) and subjected to a 12:12 hours light-dark cycle. The treatment of the experimental animals was consistent with the guidelines of Ethics by the Guide for the Care and Use of Laboratory Animals 1997, published by Clark et al. 23 in accordance with the recommendations of the animal care committee of the National Center for Radiation Research & Technology (NCRRT), Cairo, Egypt.
Experimental Design
Some of the animals, after an acclimatization period of 1 week, were fed standard chow, and others a high-fat diet (HFD), for 4 weeks. The HFD, obtained from El-Nasr Co. (Cairo, Egypt), consisted of 50% carbohydrates/starch, 27% fat, 10% protein, 10% sucrose, 1.5% fiber, and 1.5% vitamins. Animals were divided into eight groups, with eight animals per group:
Throughout the experiment, the body weight of the rats was recorded, before and after the administration of drugs. Blood samples were collected via retro-orbital bleeding into tubes. Fasting blood glucose was estimated then at ‒ 80 ͦ C and the blood aliquots were stored for further analysis. Visceral adipose tissues were excised and weighed, as previously mentioned 24,25 as the sum of epididymal weight (g) and retroperitoneal weight (g); the results were expressed in g of fat per 100 g body. All rats were sacrificed by cervical dislocation; the liver was dissected, washed with saline, dried on a filter paper, weighed, and stored at up to ‒ 80 ͦ C for further mRNA extraction.
Biochemical Analyses
A commercial kit (BIOMED, Cairo, Egypt) was used to measure plasma glucose levels, according to Lott and Turner. 26 Insulin plasma levels were evaluated (Merck Millipore, Billerica, MA, USA) using an enzyme-linked immunosorbent assay (ELISA). Estimating HMG-CoA in hepatic tissue was as described. 27 Plasma LPL (EC 3.1.1.34) and LCAT (EC 2.3.1.43) levels were assessed according to the methods described by Korn, 28 and Hitz et al., 29 respectively. Serum cholesterol, TG, and HDL-c were measured using ELISA kits (R&D System, USA), following the manufacturer’s instructions, and LDL-c was calculated by the Friedewald equation, as stated. 30 Lipid peroxidation, indicated by the formation of malondialdehyde (MDA), was assessed in plasma and liver homogenates using a described method. 31 In serum and liver tissue, PON-1 and ARE activities were measured spectrophotometrically according to the method described by Gan et al. 32 Serum adiponectin and leptin were measured using an ELISA kit (R&D Systems). Using kits supplied by BIOMED, the activities of serum alkaline phosphatase (ALP), alanine aminotransferase (ALT), and aspartate amino transaminase (AST) were assessed.
Detection of Gene Expression by Real-Time Quantitative Polymerase Chain Reaction (PCR)
Isolation of RNA and Reverse Transcription:
The mRNA expression of PPAR-γ, fatty acid synthase (FAS), fatty acid-binding protein (aP2), and CCAAT/enhancer-binding protein alpha (C/EBPa) were examined (Table 1 for primer sequences used in these investigations). Using the TRIzol reagent (Life Technologies, USA) according to the manufacturer’s instructions, total RNA was isolated from 30 mg liver tissues. Agarose gel electrophoresis (1%) was used with ethidium bromide staining to confirm the integrity of RNA. Synthesis of the first strand complementary DNA (cDNA) was achieved with reverse transcriptase (Invitrogen) using 1 µg total RNA as the template, according to the manufacturer’s protocol. RT-PCRs were performed using the Sequence Detection Program (PE Biosystems, CA) in a thermal cycler stage one plus (Applied Biosystems, USA). A 25 µL total volume reaction mixture consisted of 2X SYBR Green PCR Master Mix (Applied Biosystems), 900 nM of each primer and 2 µL of cDNA. The conditions for PCR thermal cycling included an initial step at 95 °C for 5 minutes, 40 cycles at 95 °C for 20 s, 60 °C for 30 s, and 72 °C for 20 s. At the end of the reaction, a curve analysis was conducted. Using the GAPDH gene that was amplified in each series of PCR experiments, the results were normalized. The relative expression of target mRNA was determined using the method of comparative Ct described by Livak and Schmittgen. 33
Primer Sequences Used for RT-PCR.

Peroxisome proliferator-activated receptor-gamma (PPAR-γ); Fatty acid synthase (FAS); Fatty acid binding protein (aP2); CCAAT/enhancer binding protein alpha (C/EBPa); Glyceraldehyde-3-phosphate dehydrogenase (GAPDH).
Statistical Analysis
Data were subjected to statistical analysis and tests of significance were performed using the statistical package SPSS (Statistical Program for Social Science) version 15.0 by applying a one-way ANOVA test, followed by a post hoc test for multiple comparisons. All data are expressed as a mean of 8 values; SE and difference between means are considered significant at P < 0.05.
Results
Effect of Pterostilbene on Metabolic Dysfunction Markers Under Nutritional and Irradiation Stress
In the present study, in all study groups, the initial body weights were similar. The HFD and HFD +IR group’s final body weight increased by 266 ± 18 g and 354 ± 14 g (P < 0.05), which were significantly different when compared to the normal control group. The visceral fat was high (P < 0.05) in the HFD and HFD +IR rat groups 7.5 ± 3.2 and 13 ± 1.4 g, respectively, in comparison with the normal control ones. Also, serum glucose, 8.8 ± 0.6 and 10.5 ± 1.5 mmol/L in HFD and HFD +IR rat groups respectively, was significantly raised in comparison with that of the control. A significant decrease in plasma insulin level 5.71 ± 0.7 µU/mL, (P < 0.05) was recorded for the HFD +IR rat group, but the change was not significant for the HFD rat group in comparison with the control group. Treatment with either PS or Orlistat significantly (P < 0.05) ameliorated the change in body and visceral fat weight resulting from HFD and HFD +IR, along with returning plasma glucose and insulin concentrations close to those of the control (Figure 1(A-D)).

(A-D) The effect of PS and Orlistat on body weight and visceral fat weight (g), plasma glucose (mmol/L) and plasma insulin (µU/mL) in HFD rats exposed to γ-radiation. Values are represented as the means ± S.E, N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
The appropriate mevalonate in the biosynthesis of cholesterol is generated by HMG-CoA reductase action. Increasing HMG-CoA activity contributes to the development and production of cholesterol. In this study, there was a significant increase within the liver of activation of HMG-CoA reductase due to irradiation of the HFD +IR rats. As shown in Figure 2(A), HFD and HFD +IR rats had significantly increased activity of HMG-CoA reductase in the liver, with mean activities of 3.70 ± 0.66 and 4.69 ± 1.21 (HMG-CoA/mevalonate ratio), respectively, as compared with the control group. Both PS and Orlistat inhibited the effects of HFD and HFD +IR by revealing enhancement in liver tissue levels of HMG-CoA reductase compared with the HFD and HFD +IR groups.

(A-C) The effect of Pterostilbene and Orlistat on liver HMG-CoA reductase activity (HMG-CoA/mevalonate ratio), plasma LCAT activity (µmol cholesterol esterified/h/mL) and plasma LPL activity (µmol glycerol liberated/h/mL) in HFD rats exposed to γ-radiation. Values are represented as the mean ± S.E. N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
Lecithin: Cholesterol Acyltransferase is the main enzyme that catalyzes free cholesterol esterification of lipoprotein plasma and plays a crucial role in the metabolism of high-density lipoprotein. Conversely, the plasma activities of LCAT and LPL were significantly inhibited by HFD +IR 18.15 ± 1.18 µmol cholesterol esterified/h/mL and 25.38 ± 2.4 µmol glycerol liberated/h/mL, respectively} (Figure 2(B and C)). The administration of pterostilbene mitigated the metabolic syndromes in the hyperlipidemic rats by significantly increased the activities of LPL and LCAT. PS was more efficient at ameliorating the decrease in the activities of both LCAT and LPL in HFD and HFD +IR rats, the difference being more pronounced in the case of HFD +IR rats.
Serum cholesterol, triglycerides, and LDL-c concentrations in the HFD group increased in comparison with the controls. The HFD +IR rat group showed highly significant increases in serum levels of total cholesterol, TGs, and LDL-c, and a decline in HDL-c (5.1 ± 0.31, 2.05 ± 0.07, 0.30 ± 0.02, and 3.93 ± 0.24), respectively, while the PS treatment revealed improvement in all serum levels compared to the HFD +IR group, but the values were not significantly different from those of the HFD +IR + Orlistat group (Figure 3). Even though PS decreased the values of TG, they were still not normal. As to Orlistat, the behavior was similar, even though the decrease in TG levels in the HFD +IR group was less pronounced.

(A-D) Effect on lipid profile of Pterostilbene and Orlistat in HFD rats exposed to γ-radiation. Values are represented as the mean ± S.E. N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
Elevated levels of lipid peroxidation in both plasma and liver tissues of hyperlipidemic rats indicated an increase in oxidative stress. As a result of the increased lipid profile, lipid peroxidation accumulated. High-fat diet rats resulted in a significant rise in MDA levels (plasma and liver) compared to the control group. The MDA level in the plasma of the HFD +IR rat group was recorded as 60.1 ± 2.9 nmol/mL (Figure 4(A)) and 302 ± 12 nmol/mg in the liver tissue (Figure 4(B)), which were significantly different (P < 0.05) compared to the control group. The serum MDA levels in the PS and Orlistat groups were similar to those of the control. However, the liver MDA values for the HFD +IR + PS and HFD +IR + Orlistat groups (Figure 4B) were higher than those of the control. PS and Orlistat treatments were able to correct this imbalance in the radiation-induced dyslipidemia marker serum, and the liver MDA levels in the HFD +IR + PS and HFD +IR + Orlistat groups relative to the HFD +IR group (Figure 4(B and C)).
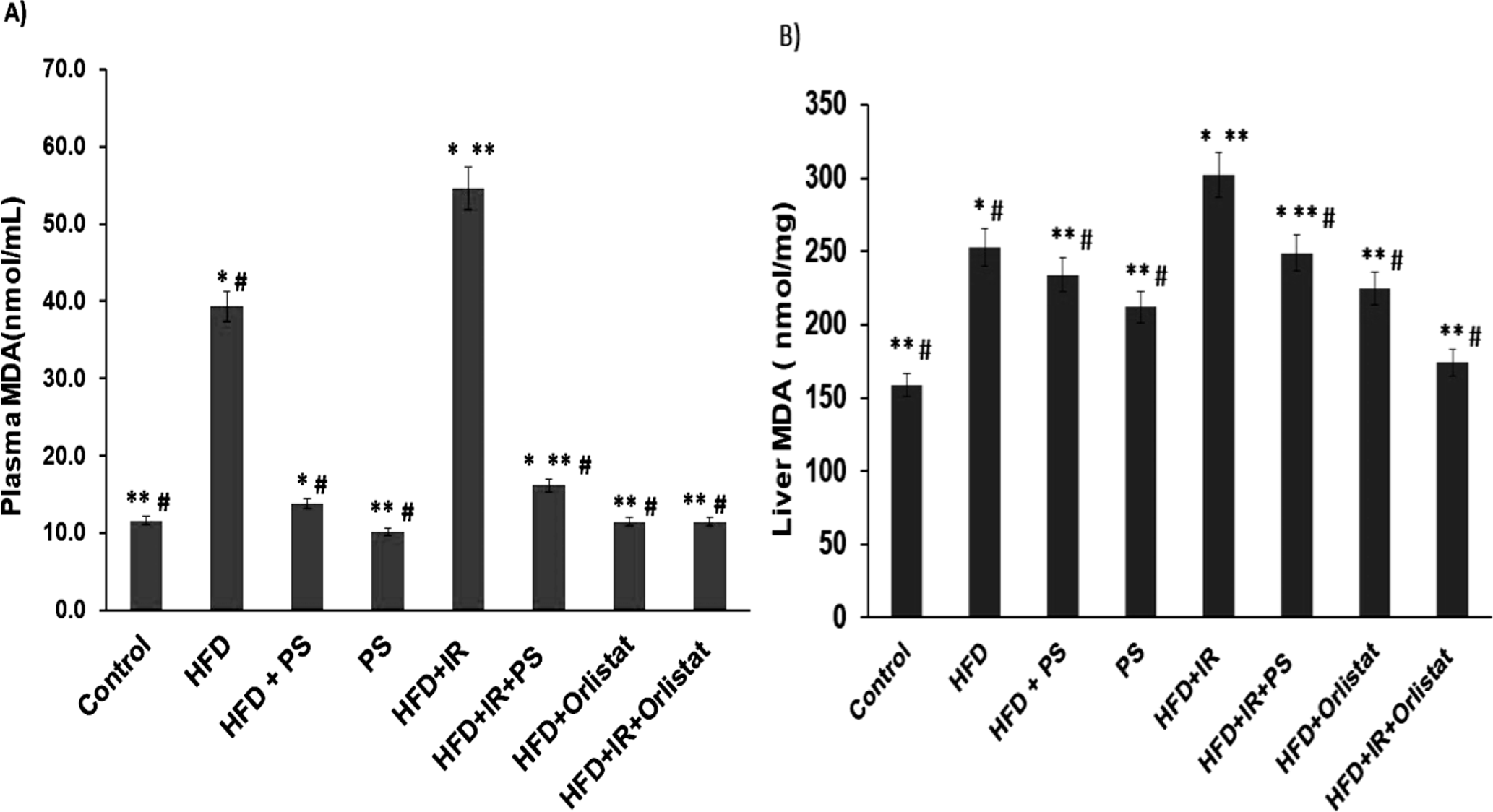
(A & B) The effect of Pterostilbene and Orlistat on MDA serum (nmol/mL) and liver tissue (nmol/mg) levels in HFD rats exposed to γ-radiation. Values are represented as the mean ± S.E. N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
Effect of Pterostilbene and Orlistat on the Adipogenesis Transcription Factors Under Nutritional and Irradiation Stress
Gene expression of PPAR-γ amounted to 0.256 ± 0.022 fold change in the HFD +IR rat group as compared to the control 1.02
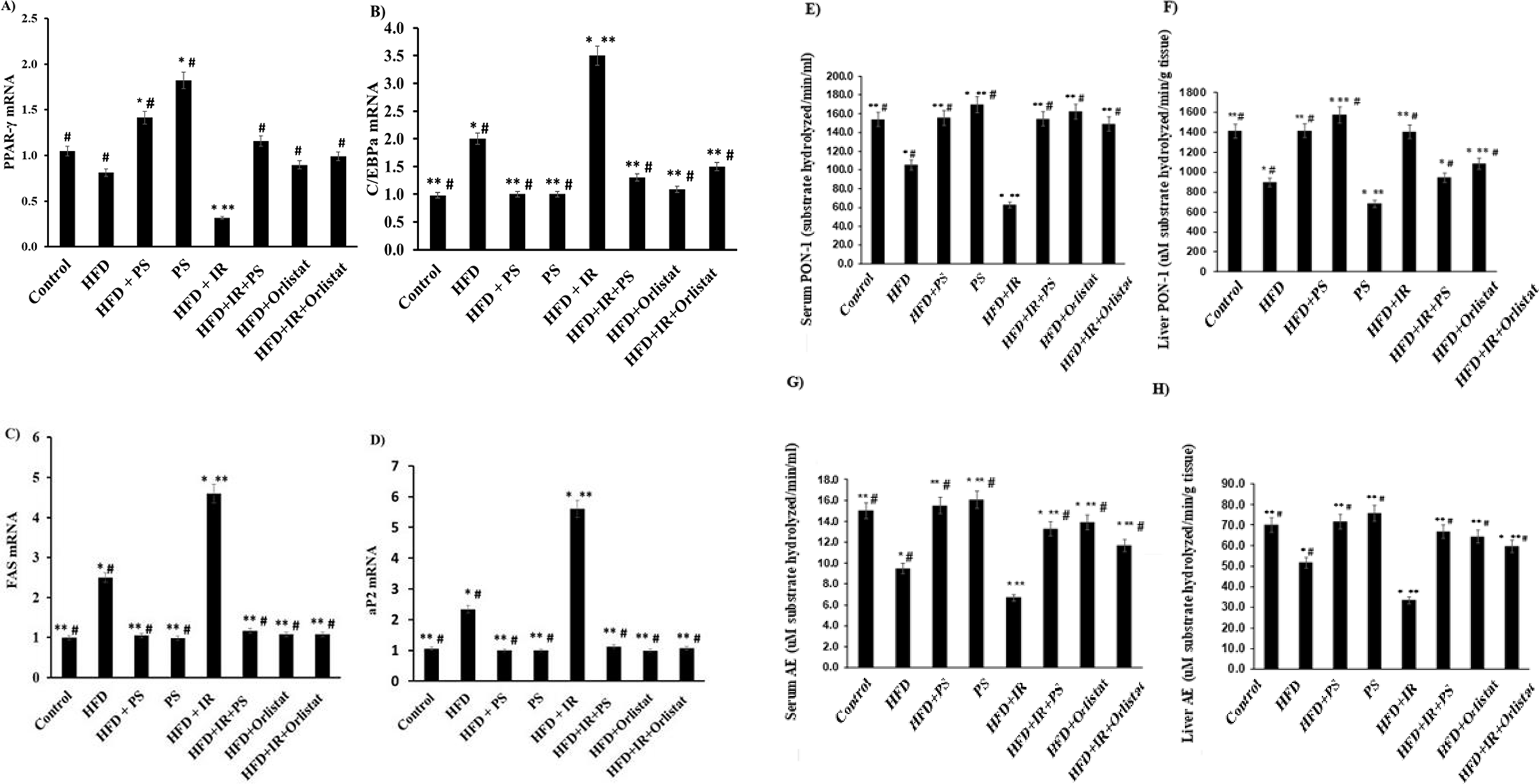
(A-H) Effect of Pterostilbene and Orlistat on the mRNA levels of PPAR-γ, C/EBPa, FAS, aP2 and PON-1/AE activity in HFD rats exposed to γ-radiation. Values are represented as the mean ± S.E. N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
Low PON-1 levels in obesity are a potential marker of metabolic syndrome development. Significant (P < 0.05) decline in PON-1 activity was noted for the serum (63.11 ± 2.31 µM substrate hydrolyzed/min/mL; Figure 5(E)) and liver (648.7 ± 12.5 µM substrate hydrolyzed/min/g tissue; Figure 5(F)) of the HFD +IR group compared with the normal group. Both PS and Orlistat normalized the activity of serum PON-1 in the HFD and HFD +IR groups (Figure 5). However, a similar effect for both compounds was not observed in the PON-1 activity of the liver; PS was much more efficient than Orlistat. In the HFD +IR group, the data show a significant (P < 0.05) decline in AE activity levels in both serum and liver {6.4 ± 2.7 µM substrate hydrolyzed/min/mL in serum (Figure 5(G)) and 33.5 ± 3.4 µM substrate hydrolyzed/min/g tissue in liver (Figure 5(H)) when compared to the control group. On the other hand, when hyperlipidemic rats were treated with PS a significant (P < 0.05) increase in activity levels was observed (serum and liver) in comparison with both the HFD and HFD +IR groups. PS was more efficient in overcoming the HFD effects than the HFD +IR effects and overall was more efficient than Orlistat.
Effect of Pterostilbene and Orlistat on Serum Leptin and Adiponectin Under Nutritional and Irradiation Stress
The influence of Pterostilbene and Orlistat treatment on both serum leptin and adiponectin levels in HFD +IR rats is presented in Figure 6(A). In high-fat diet rats, there was a significant (P < 0.05) increase in the level of serum leptin (3641 ± 15.3 pg/mL) and a significant (P < 0.05) drop in adiponectin level (4224 ± 13.5 pg/mL) (Figure 6(B)) in comparison with the controls. Administration of either Pterostilbene or Orlistat significantly modulated the increase in the leptin level and reduced it to a nearly normal value. PS effectively modulated the decrease in adiponectin levels compared with the HFD and HFD +IR groups.

(A & B) Effect of Pterostilbene and Orlistat on leptin (pg/mL) and adiponectin (pg/mL) in HFD rats exposed to γ-radiation. Values are represented as the mean ± S.E. N = 6. *P < 0.05, **P < 0.05, # P < 0.05, compared with normal (Control) and HFD and HFD +IR groups, respectively.
To determine possible hepatic impairment in the HFD and HFD +IR groups, ALP, AST, and ALT activities were assessed and a significant increase was detected in comparison to the normal group. Significant changes in the levels of liver function enzymes in the PS and Orlistat treated groups were detected when compared to the HFD +IR group (Table 2).
Effect of PS and Orlistat on Liver Function Enzymes in Rats Under Nutritional and Irradiation Stress.

Values are represented as the mean ± S.E. N = 6 *P < 0.05, **P < 0.05, # P < 0.05, compared with controls and HFD and HFD +IR groups, respectively.
Discussion
The adipogenesis process includes morphological changes, lipid accumulation, and the expression of genes characteristic of fat cells, for example, fatty acid binding protein-4 (FABP4) and adipokines, such as leptin and adiponectin. 34 The adipocyte-differentiation markers include PPARγ, PPARα, and C/EBPα, and the genes related to lipid metabolism include LPL, AP2, sterol regulatory element binding protein (SREBP) 1 c, FAS, acetyl-CoA carboxylate 1 (ACC1) and carnitine palmitoyltransferase 1B (CPT1B). 35 PPARγ and C/EBPα play synergistic roles in the activation and expression of genes that are activated in differentiation, and participate in a major adipocyte change. 36
In the present study, HFD rats exposed to irradiation showed a significant increase in body weight, accompanied by immense visceral adipose tissue. Such elevation was reduced in the groups that received either PS or Orlistat treatment, which reflected the efficiency of both treatments for the control of weight gain in those who develop metabolic syndrome by accumulating fat and dyslipidemia. In agreement with the above, the recent study of Aguirre et al. 37 reported that obese rats treated with PS maintain body weight when compared with that of healthy rats.
The current results also showed that rats fed HFD and irradiated (6 Gy) had elevated blood glucose levels, accompanied by loss of insulin secretion, which increases the risk for insulin resistance and adipose homeostasis of total body weight. This is consistent with Nylander et al. 6 that irradiated mice fed a HFD showed a fat mass and insulin sensitivity. This may be due to adipose tissue accumulation and consequent lipodystrophy following irradiation. We observed that either PS or Orlistat treatment leads to normoglycemia and a shift from a catabolic to an anabolic state when fed a high-fat diet. We thought that the better glycemic levels may result from the normalized insulin use by cells that oxidize glucose as a mitochondrial source of energy to preserve adipose and muscle tissue and regain body weight in the PS and Orlistat treatment groups. Thus, PS appears to mimic insulin action instability to normalize tissue responsiveness in both HFD and HFD +IR rat groups. In brief, the protective effect of PS may be explained by the ability of the drug to eliminate free radical intermediates of glucose-oxidation reactions along with an anti-hyperglycemia activity that is attributable to insulin release from established pancreatic β-cells. 38 Fatty acid formation is a key mechanism of lipogenesis that takes place in the adipose tissue. Fatty acids stimulate the induction of triglycerides and adipogenesis processes in the white adipose tissue, via the upregulation of PPARs, which activate CCAAT/enhancer-binding protein α. Thus, increased levels of SREBP-1c boost adipogenic gene expression, fatty acid synthesis, and triglyceride production. 39 In adipose tissue of the HFD +IR rat group, treatment with either PS or Orlistat prevents adipogenesis and endorses lipolysis. Moreover, Orlistat moderately inhibits triglyceride hydrolysis, consequently diminishing the absorption of monoglycerides and free fatty acid. 40 As shown from the results, the significant elevation observed in the key adipogenic target gene mRNA levels (including C/EBPa, FSA, and aP2) were inhibited by both treatments. PPAR-γ activation has been reported to be linked with regulation of antioxidant enzyme paraoxonase expression. 41,42 On the other hand, up regulation of the PPAR-γ gene expression associated with lower PON-1 activity seen in this study can consequently be explained by the significant increase in the oxidative stress marker MDA and irregularities in the lipid profile of the HFD groups. The increased ROS affects large molecules such as poly-unsaturated fatty acids to initiate lipid peroxidation, which further changes the fluidity and permeability of the cell membrane, 43 which may lower the PON-1 activity levels in the HFD group. Meanwhile, the expression of genes that promote selective utilization of lipid substrates is augmented during the early oxidative stress states that are associated with increased systemic delivery of free fatty acid. 44 This agreed with the increment in PPAR-γ gene expression as an early response in the HFD group. Shunmoogam et al. 45 reported that PON-1 is a protein with HDL-related properties and hydrolysis capabilities for oxidized LDL cholesterol with potential arterioprotection and capable of acting as a screening tool for metabolic stress, vascular stress, and resistance to insulin. Our results demonstrate that PS and Orlistat significantly attenuate HFD +IR induced lipid accumulation concomitant with a significant decrease in PON-1/AE activity and PPAR-γ gene expression. Satheesh & Pari 46 documented that Pterostilbene has hypolipidemic interaction that offers a possible alternative to dyslipidemia therapy. Another study has also documented that treatment with Pterostilbene reduced cell growth, triacylglycerol deposition, and lipid production. 17 Ma et al. 47 have clarified that hypercholesterolemia may be linked to stimulation of the HMG-CoA reductase enzyme, the main regulatory enzyme in the overall cholesterol synthesis process. They explained that the elevated activity of liver HMG-CoA reductase, accompanied by a reduction in plasma LPL and LCAT enzyme levels, besides a high elevation in plasma cholesterol, triglyceride, and LDL cholesterol levels was associated with a significant decline in HDL level, as affected by induced dyslipidemia in the HFD +IR rat group. These results are consistent with those in previous studies of irradiation consequences. 11 Afonso et al. 48 found that the reduction in LCAT activity led to the free accumulation of cholesterol and excess plasma lipoprotein as LCAT is the essential enzyme for esterification and trans-esterification in the cholesterol process. It has been confirmed that PS therapy normalizes to a large extent the pathological changes in tissues of dyslipidemic rats. 49 PS and Orlistat treatments of HFD plus IR rats significantly attenuated the radiation-induced increase in HMG-CoA reductase activity. In contrast to the effect of either PS or Orlistat on LCAT and LPL plasma activities, displaying elevated LCAT activity, a significant increment in LPL activity was observed. This observation is in agreement with Cho et al., 50 who declared that nutritional resveratrol reduces serum cholesterol through hepatic down-regulation of HMGR mRNA in hamsters that had been fed a high-fat diet. PS, which is related to resveratrol, may act as an inhibitor of HMG-CoA reductase, a rate-limiting enzyme in sterol biosynthesis. The cellular energy status sensor and primary controlling tool for monitoring entire-body energy homeostasis is AMP-activated protein kinase (AMPC). Resveratrol plays an essential role in lipid metabolism by activating fatty acid oxidative processes and inhibiting lipid synthesis by activating AMPK. 17 We consider the lipid profile abnormality a stimulation key for oxidative stress generation and lipid peroxidation 15 that were detected in the current study. Obese rats exposed to γ-radiation share a common linkage with oxidative stress and accumulation of adipose tissue cells. Ionizing radiation-induced liver oxidative stress status leads to the pathogenesis of steatosis, which is evidenced by depletion of reduced glutathione and increased lipid peroxidation. 45 The marked increase in liver and plasma MDA recorded in this study might be due to the interaction of ionizing-radiation with water, major cellular constituent consequences of which are the generation of various reactive oxygen spices (ROS), including hydroxyl radical (•OH), hydrogen peroxide (H2O2), superoxide radical (•O−2), and ultimately oxygen (O2) as an attack on the fatty acid composition of membrane lipids. 51 These findings could be explained by the antidyslipidemic effects of pterostilbene, which show a significant reduction in the absorption of gastrointestinal (GI) tract enzymes involved in carbohydrate and lipid metabolism by blocking the most effective comportment. Resveratrol is known as an α-glucosidase inhibitor. 52 The potent pharmacological features of pterostilbene have been related to ring A, which contains methoxy groups at C3 and C5. With these structural characteristics, the lipophilic pterostilbene improves its membrane permeability, bioavailability, and bioactivity. Pterostilbene, with an aromatic ring, contains two–OCH3 groups and its hydrogen bonding interaction pattern 53 could inhibit HMG-CoA reductase activity, which is the critical enzyme of lipid-lowering significance and hence improving the unbalanced adipogenesis gene mediated by γ-irradiation.
Adiponectin reduces triglyceride content in muscular and liver tissues by decreasing insulin resistance in dyslipidemia rats, which gives rise to increased hepatic insulin sensitivity and raises peripheral glucose efficiency in the liver. Adiponectin expressions are negatively correlated with body mass levels, in comparison to leptin, which correlates positively with adipocyte size and lipid content. 54 Anagnostis et al. 55 reported that body mass index and total blood cholesterol, LDL, triglyceride, and leptin levels fell significantly in obese sibutramine and Orlistat treated subjects. The present work is parallel to the previous publication which stated that metabolic syndrome induced an increase in serum leptin and a decrease in adiponectin levels in rats. 56 Administration of either PS or Orlistat led to marked enhancement in adiponectin, which modulated the increase in serum leptin level and returned it to a nearly normal level compared with a high-fat diet and exposed to radiation group. Also, PS and Orlistat treatment gave protection from liver damage by improving liver function enzymes.
Conclusion
Finally, it could be concluded that the findings of the current study provide preliminary evidence that pterostilbene and Orlistat can act as normolipidemic agents that affect lipid and inhibit activation of the progenitor adipose gene induced under radiation and high-fat diet stressors, and maybe have the potential for beneficial use on the metabolism of patients with metabolic disorders caused by radiotherapy.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
