Abstract
Two novel flavonoids (
Tagetes patula L. (French marigold), family Asteraceae, is widely known for its phytochemical and medicinal properties. It is native to Mexico and other warmer parts of America and naturalized elsewhere in the tropics and subtropics. 1 Currently, in China, T. patula is a common ornamental cultivated throughout the country. Traditionally, T. patula was used to treat various diseases, such as cough, colic, constipation, diarrhea, rheumatism, and eye problems. Today, the plant is specially used as an antimicrobial, antiseptic, hepatoprotective, 2 blood purifying, and diuretic agent. 3 Previous phytochemical investigations of T. patula have resulted in the isolation of carotenoids, 4,5 flavonoids, 6,7 and monoterpenoids. 8 In our previous studies, we reported the obvious antidepressive effects of T. patula, 9 and, in this article, we report 2 new and 3 known flavonoids isolated from T. patula by various chromatographic techniques. Furthermore, the injury of human neuroblastoma SH-SY5Y cells induced by glutamate was examined to determine the neuroprotective action of these flavonoids.
Results and Discussion
Structural Elucidation of the Isolated Compounds
Compound
1H NMR and 13C NMR Data for Compound 1.

Recorded in dimethyl sulfoxide-d 6, 500 MHz for 1H and 125 MHz for 13C, δ in ppm.

Structure of the isolated compounds from Tagetes patula.
Compound
1H-Nuclear Magnetic Resonance (NMR) and 13C NMR Data for Compound 2.


Heteronuclear multiple bond correlations of compounds 1 and 2.
The spectral properties of the known compounds we isolated were compared with published values, and the compounds were identified as patuletin (
Cytotoxic Activity Assay
Compared with the negative control group, the compounds at concentrations of 50-100 µmol/L significantly decreased the cell viability, whereas concentrations between 1 µmol/L and 10 µmol/L only slightly affected the viability of cells. Concentrations lower than 10 µmol/L was selected for this study (Supplemental Table S1).
Protective Effect on SH-SY5Y Cell Injury Induced by Glutamate
Depression has become the most prevalent mental health problem in developing countries, especially among adolescents. Injury of the human neuroblastoma SH-SY5Y cell line with a low degree of differentiation and metabotropic glutamate receptors induced by glutamate has been applied for the screening of antidepressive active natural products. 17 Here, the isolated compounds were examined by this method.
The isolated compounds were tested for their protective action on neuronal cells. The cells treated with marigold biflavone at a concentration of 0.1 µmol/L produced maximal cell viability of 86.0% ± 0.6%, and at concentrations of 0.01-1 µmol/L significantly increased cell viability (Figure 3). Treatment with patuletin-3-O-α-

Protective effects of isolated compounds on glutamate (Glu)-induced cytotoxicity in SH-SY5Y cells.
Materials and Methods
Materials and Reagents
Tagetes patula was provided by Dalian Wuzhou Holy Herb Scientific and Technological Co. Ltd., China) and identified by Professor Bing Wang of the Liaoning University of Traditional Chinese Medicine; the voucher specimens (No. 20160911) were deposited in the specimen herbarium, Liaoning University of Traditional Chinese Medicine. Optical rotations were measured with a Jasco P-2000 polarimeter; for HPLC, an Agilent Technology Co, Ltd model was used; semipreparative HPLC was performed using a HITACHI 7100 instrument (Hitachi Corporation, Tokyo, Japan); column chromatography was performed using silica gel (200-300 mesh, Marine Chemical Factory, Qingdao, China) and polyamide resin (200-300 mesh, Marine Chemical Factory, Qingdao, China; 80-100 mesh, Taizhou Luqiao Sijia Biochemical Plastic Factory, Taizhou, Zhejiang, China). Thin-layer chromatography (TLC) was carried out on silica gel GF 254 plates (Qingdao Marine Chemical Co., Qingdao, China).
Extraction and Isolation
The dried stems and leaves (15 kg) of T. patula were extracted with 60% ethanol (EtOH) at low temperature (45 °C; 3 × 120 L, 2 hours each). After filtration, the combined extracts were concentrated in a vacuum to furnish the crude extract. This was extracted with petroleum and ethyl acetate successively several times to give the ethyl acetate layer and water (H2O) layer, which was subjected to column chromatography over AB-8 macroporous adsorptive resins using EtOH/H2O (0:100, 60:40, and 95:5) as eluent. The 60% EtOH eluate and ethyl acetate layer (total 300 g) were combined and treated by column chromatography on silica gel, eluting with dichloromethane (CH2CL2)/methanol (MeOH), then with increasing amounts of MeOH to afford 6 fractions (Fr.A1-A6). Fr. A4 was passed over a silica gel column, using CH2CL2/MeOH as eluent, with increasing amount of MeOH to give 6 fractions (Fr.B1-B6). Then, B4 was purified on an ODS column equipped with HPLC eluting with MeOH/H2O. Compounds
The dried flowers (8 kg) of T. patula were extracted 3 times with 95% EtOH for 6 hours each time. After filtration, the residues were extracted with 60% EtOH again and the combined extracts concentrated under reduced pressure. Then, the dry residue was extracted with butyl alcohol and methylene chloride to give butyl alcohol and methylene chloride layers. The butyl alcohol layer was subjected to silica gel column chromatography using CH2CL2/CH3OH (100:0→75:25→75:50 →50:50→25:75→0:100) as eluent to give 6 fractions (Fr.C1-C6). Fr.C2 was subjected to polyamide column chromatography, using MeOH/H2O as eluent, with increasing amounts of MeOH to give 5 fractions (Fr.C2.1-C2.5). Compound
Identification of compound 1
Yellow powder; 1H-NMR (dimethyl sulfoxide [DMSO]-d 6, 500 MHz) and 13C NMR (DMSO-d 6, 125 MHz), see Table 1; HR-ESI-MS m/z 675.1040 [M − H]−, calculated as 675.0992.
Identification of compound 2
Yellow amorphous powder;
Acid Hydrolysis of Compound 2
Acid hydrolysis was performed using 0.1 mol/L sulfuric acid at 100 °C for 1 hour. The mixture was neutralized with silver carbonate and extracted with ethyl acetate. After the aqueous layer was condensed, both of them were examined by HPLC and compared with authentic samples. Analytical HPLC was performed on an Agilent ZORBAX NH2 column (5 m, 4.6 mm 150 mm) with isocratic elution using acetonitrile-H2O (80:20) for 40 minutes at a flow rate of 1 mL/min. The peaks were detected with an evaporative light scattering detector.
Assay Method for Cytotoxic Activity
SH-SY5Y cells were cultured in a 96-well plate at a density of 5 × 105 cells per well and incubated in a humid atmosphere of 5% carbon dioxide (CO2) and 95% air at 37 °C. Following 24 hours of incubation, the cells were completely attached to the well bottom. The cells were randomly divided into 6 groups (n = 3 per group): the cells of the blank group only had DMEM/F-12. The cells of the administration group were treated with various concentrations of flavonoids (marigold biflavone, patuletin, quercetin, kaempferol-3-O-β-
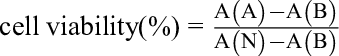
where A refers to the administration group, B is the blank group, and N is the negative control group.
Study on the Protective Effect of Flavonoids on Glutamate-Induced SH-SY5Y Cells
Glutamate, a major excitatory amino acid neurotransmitter in the central nervous system mediates several physiological processes. An increasing body of evidence indicates the important role of the glutamatergic system in the pathophysiology of depression. First, depressed patients exhibit elevated levels of glutamate both in plasma and the limbic brain areas, which are believed to be involved in mood disorders. 18 Additionally, it has been shown that chronic treatment with antidepressants of different mechanisms reduces the glutamate release in rats. 19,20 The isolated compounds from T. patula were evaluated for their protective effect on glutamate-induced SH-SY5Y cells. The protective effect of glutamate-induced SH-SY5Y cells was performed using the MTT assay method.
SH-SY5Y cells were cultured in a 96-well plate at a density of 5 × 105 cells per well and incubated with a humid atmosphere of 5% CO2 and 95% air at 37 °C. Following 24 hours of incubation, the cells were completely attached to the well bottom. The cells were randomly divided into 9 groups (n = 3 per group): the cells of the model group were treated with
Following 24 hours of incubation, 20 µL of MTT stock solution (0.2 mg/mL in phosphate-buffered saline) was added to each well. After 4 hours of incubation, the supernatants were aspirated, and 100 µL of DMSO was added to each well. After oscillation for 10 minutes at 37 °C, the absorbance was measured at 492 nm by a microplate reader. Data were normalized as a percent of vehicle-treated cells (100%) in 3 independent experiments and are presented as the mean ± SEM. The cell viability was calculated using the formula:
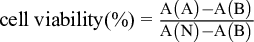
where A refers to the administration group, B is the blank group, and N is the negative control group.
Conclusions
In this work, 2 novel (
Supplemental Material
Supplementary Material 1 - Supplemental material for Chemical Constituents of Tagetes patula and Their Neuroprotecting Action
Supplemental material, Supplementary Material 1, for Chemical Constituents of Tagetes patula and Their Neuroprotecting Action by Linlin Liu, Simin Luo, Miao Yu, Ahmed M. Metwaly, Xiaoku Ran, Chunyan Ma, Deqiang Dou and Decheng Cai in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by the Liaoning Province Distinguished Professor Support Program, China.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
