Abstract
Natural products have presented potentiality to improve the outcomes of cancer therapies. Impressic acid (E12-1) and acankoreanogein (E13-1), important activity compounds in
Pancreatic cancer is one malignant tumor of the digestive system accompanied with high cancer mortality and its morbidity and mortality significantly rise in recent years.1-3 In the United States, pancreatic cancer is the fourth leading cause of cancer mortality, and the 5-year survival rate of pancreatic cancer patients is only 6%.4,5 In China, the annual incidence of pancreatic cancer is about 5.1 cases/100 000 people, with 5~60 000 new sufferers every year. 6 Gemcitabine (GEM, Figure 1(A)) had been approved as the first-line drug for the treatment of advanced pancreatic cancer by the US Food and Drug Administration in 1996, and been applied in clinical treatment in China in 1999.7 -10 The standard treatment for pancreatic cancer is chemotherapy with GEM and erlotinib, since 2005. However, their efficacies are still poor, with median survival of 8.5 months. 11 Moreover, high doses of GEM ultimately result in the resistance and toxicity. One popular explanations on the limited effect of GEM is the faster-forming resistance of GEM in pancreatic cancer. 12 To improve the efficacy of GEM in the treatment of advanced pancreatic cancer, some other drugs were used in combination with GEM, such as cisplatin, capecitabine, and docetaxel, and showed improvement on the efficacy and tumor-related symptoms. 13

The structure of (A) GEM, (B) E12-1, and (C) E13-1.
In recent years, natural products had received considerable attention due to their beneficial effect and low toxicity.
14
Research results showed that many natural products could overcome the intrinsic resistance of tumor cell apoptosis. Thence the application of clinical drugs with natural products obtained more and more attention.
Combination drugs strategy targeting multiple pathways has attracted an increasing interest to target multiple pathways. Therefore, in continuation to our interest in combination drugs and their biological activities, herein we isolated E12-1 and E13-1 from
Results and Discussion
Isolation of E12-1 and E13-1
The powder of
Effect of E12-1, E13-1 and GEM on the Growth of Panc-1 Cells
First, the effects of E12-1, E13-1, and GEM on Panc-1 cells were evaluated by 3-(4,5-dimethylthiazol-2-yl)−2,5-diphenyltetrazolium bromide (MTT) assays. As shown in Figure 2, treatment with E12-1, E13-1, or GEM alone resulted in the inhibition of panc-1 cells viability in a dose-dependent manner. The half-maximal inhibitory concentration (IC50) values of E12-1, E13-1, and GEM were 66.13, 98.38, and 2.78 µM, respectively.

Effect of E12-1, E13-1, and GEM on the growth of Panc-1 cells. E12-1, impressic acid; E13-1, acankoreanogein; GEM, gemcitabine.
According to our previous studies, 18 the treatment concentration of compound with cell lethality ~20% was selected to evaluate the synergistic effect of combination drugs. Based on MTT assay results, concentrations of E12-1, E13-1, and GEM were selected as 10, 35, and 0.2 µM, respectively. As shown in Figure 3, treatment with E12-1, E13-1, and GEM alone resulted in cell viability of 80.74%, 75.46%, and 77.85%, respectively. However, treatment with E12-1 + GEM or E13-1 + GEM leaded to cell viability to 49.70% and 44.21%, respectively, which were significantly lower than E12-1, E13-1, or GEM alone. The results indicated that E12-1 + GEM or E13-1 + GEM presented synergistic effect on the growth of Panc-1 cells.

Synergistic effect of E12-1, E13-1, and GEM on the growth of Panc-1 cells. E12-1, impressic acid; E13-1, acankoreanogein; GEM, gemcitabine. **
Effect of E12-1, E13-1, and GEM on the Apoptosis of Panc-1 Cells
Apoptosis that is controlled by multiple genes is the basic measure to maintain the dynamic balance of cell number in the organism. 25 Also, apoptotic cells assay is a key method to evaluate the anticancer effect of drugs. So the apoptotic effect of E12-1, E13-1, and GEM in Panc-1 cells was investigated. As seen in Figure 4(A), treatment with E12-1, E13-1, and GEM alone caused chromatin condensation and nuclear shrinkage, the hallmarks of apoptotic cells compared to control group. And treatment with E12-1, E13-1, and GEM alone resulted in the apoptosis of 25.43%, 22.61%, and 24.25%, respectively (Figure 4(B)). However, treatment with E12-1 + GEM or E13-1 + GEM not only caused more morphological change of cells (Figure 4(A)), but also leaded to the apoptosis of 44.26% and 46.18%, respectively (Figure 4(B)).

Effect of E12-1, E13-1, and GEM on the apoptosis of Panc-1 cells. (A) Morphological change 200×, (B) percent apoptosis. DMSO, dimethyl sulfoxide; E12-1, impressic acid; E13-1, acankoreanogein; GEM, gemcitabine. *
The combination index (CI) of GEM with E12-1 or E13-1 was calculated to be 0.25 and 0.46, respectively. These results showed that coadministration treatment of GEM with E12-1 or E13-1 could synergistically enhance the apoptosis of Panc-1 cells compared to E12-1, E13-1, and GEM alone (
Effects of E12-1, E13-1, and GEM on Cell Migration
Then the effect of E12-1, E13-1, and GEM on cell migration was assayed by wound healing experiment. 26 Figure 5(A) and (B) verified that treatment with E12-1, E13-1, and GEM alone or in combination decreased the migration rate. The cell migration rates of E12-1, E13-1, and GEM group were 87%, 83%, and 79%, respectively. However, treatment with E12-1 + GEM and E13-1 + GEM markedly decreased the migration rate to ~57% and ~47%, respectively.

Effect of E12-1, E13-1, and GEM on cell migration. (A) Cell migration assay 200×, (B) migration rate. DMSO, dimethyl sulfoxide; E12-1, impressic acid; E13-1, acankoreanogein; GEM, gemcitabine. **
Effect of E12-1, E13-1, and GEM on NF-κB, p-Stat 3 Activity
Finally, we investigated the underlying molecular mechanisms of E12-1, E13-1, and GEM. Nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) was found to play a key role in apoptosis and survival of prostate cancer.27-29 Compared with the control group, treatment with E12-1, E13-1, and GEM alone showed low inhibitory effect on the expression of NF-κB; however, treatment with E12-1 + GEM and E13-1 + GEM exhibited obvious inhibition effect on the expression of NF-κB (Figure 6).
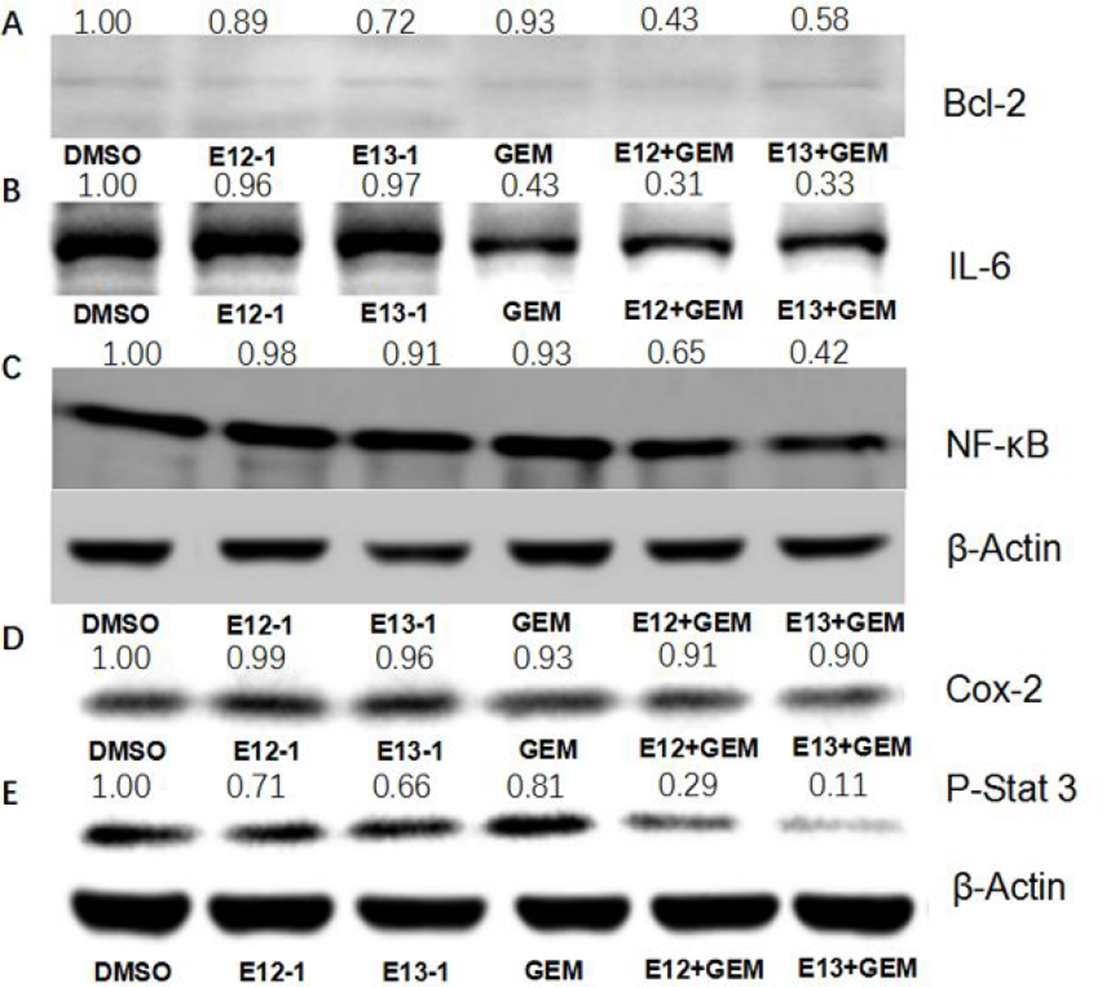
Effect of E12-1, E13-1, and GEM on NF-κB, p-Stat 3 activity. COX-2, cyclooxygenase-2; DMSO, dimethyl sulfoxide; E12-1, impressic acid; E13-1, acankoreanogein; GEM, gemcitabine; IL-6, interleukin 6; NF-κB, nuclear factor kappa-light-chain-enhancer of activated B cells; p-Stat 3, phosphorylated signal transducer and activator of transcription 3.
Interleukin-6 (IL-6) and cyclooxygenase-2 (COX-2), 2 important pleiotropic cytokines, were regulated by NF-κB signal pathway. 30 They displayed key role in the specific immune response and inflammatory response and were involved in the malignant transformation and apoptosis of tumor. 31 The effect of E12-1, E13-1, and GEM on IL-6 and COX-2 was further detected, and found that treatment with E12-1 + GEM and E13-1 + GEM could more effectively downregulate the IL-6 and COX-2 than E12-1, E13-1, and GEM alone (Figure 6). B-cell lymphoma 2 (Bcl-2), an important NF-κB downstream target, prevents cells from apoptosis through the mitochondrial pathway. 32 Our results also showed that treatment with E12-1 + GEM and E13-1 + GEM could more effectively regulate the expression of Bcl-2 than E12-1, E13-1, and GEM alone (Figure 6).
Finally, we analyzed the effect of E12-1, E13-1, and GEM on the activity of Stat 3 that is certificated to have high relation with the suppression of apoptosis, tissue invasion, and limitless replicative potential in pancreatic cancer.33,34 As shown in Figure 6, treatment with E12-1 + GEM and E13-1 + GEM had stronger effect on the decreasing expression of Stat 3 in Panc-1 cells than E12-1, E13-1, and GEM alone.
Conclusions
In summary, our study suggests that GEM combined with E12-1 or E13-1 could markedly and synergistically inhibit the growth, induce the apoptosis, and reduce the migration of Panc-1 cells compared to GEM, E12-1, and E13-1 alone. These synergistic effects of coadministration treatment were related to their ability to regulate the expression of NF-κB and phosphorylated signal transducer and activator of transcription 3 (p-Stat 3) activity, and levels of Bcl-2 and IL-6. Hence, the application of GEM combined with E12-1 or E13-1 might be an effective method for the prevention of pancreatic cancer.
Materials and Methods
Chemical Reagents
Dulbecco’s modified Eagle’s medium (DMEM) tissue culture medium, penicillin-streptomycin, fetal bovine serum (FBS), and L-glutamine were obtained from Gibco (Grand Island, NY, USA). Matrigel was obtained from BD Biosciences (Bedford, MA, USA). MTT and propidium iodide (PI) were purchased from Sigma-Aldrich (St. Louis, MO, USA). Trypan blue stain was obtained from Cambrex (Walkersville, MD, USA). β-actin, p-Stat 3, and IL-6 were purchased from Millipore Corporation (Billerica, MA, USA). COX-2 and NF-κB were obtained from Cell Signaling Technology (Beverly, MA, USA). Bcl-2 was purchased from Santa Cruz Biotechnology (Dallas, TX, USA). Secondary antibodies were purchased from Santa Cruz. PANC-1 cells were obtained from the American Type Culture Collection (Rockville, MD, USA).
Isolation of E12-1 and E13-1
The powdered stems and leaves of
E12-1 (150 mg) was yielded from
Cell Culture
Panc-1 cells were cultured in DMEM containing 10% FBS, 100 µg/mL streptomycin, 100 U/mL penicillin, 300 µg/mL L-glutamine, and 2.0 g/L sodium bicarbonate at 37 ℃ in a humidified atmosphere of 5% CO2. E12-1, E13-1 and GEM were dissolved in DMSO and the final concentration of DMSO in the cell culture medium was 0.1%.
MTT Assay
Panc-1 cells (1 × 105 cells/mL) were seeded in 96-well plate and incubated for 24 hours. Then E12-1, E13-1, GEM, E12-1 + GEM, and E13-1 + GEM solution were added into each corresponding well, followed by an incubation for 72 hours. Then 100 µL MTT was added and cells were continuously incubated for 3 hours. After the medium was removed, 100 µL DMSO was added into each well. Thus, the absorbance of each sample at 570 nm was detected with a microplate reader. DMSO-treated cells were set as control, and each sample was done in triplicate. Finally, the IC50 value of compound on the cell growth was calculated.
Apoptosis Assays
The cytospin slide of each sample was made using the cell suspension without treatment of trypan blue stain. After fixed with methanol/acetone (1:1, v/v) for 10 minutes, the cells were stained with PI for 10 minutes. The morphology of cells was observed with a fluorescence microscope. And the apoptotic cells could be confirmed by their morphological features.
Cell Migration
Panc-1 cells were seeded in six-well plate and incubated until density of ~90%. Then the wound line was scratched across the plate with a standard pipette tip (10 μL). After washed with PBS to remove the cell debris, the cells were incubated with E12-1, E13-1, GEM, E12-1 + GEM, and E13-1 + GEM, respectively. The cell-free wound area was observed under a light microscope. The cell migration effect of each sample was assessed by the percentage of remaining cell-free area compared to its initial wound. DMSO-treated cells were set as control.
Western Blotting
Panc-1 cells (1 × 105 cells/mL) were seeded in 100 mm petri dish and incubated for 24 hours. Then cells were treated with E12-1, E13-1, GEM, E12-1 + GEM, and E13-1 + GEM for 24 hours, respectively. DMSO-treated cells were set as control. The cells were collected and lysed in 200 µL RIPA buffer with protease inhibitor cocktail to extract total protein. After determining the whole protein concentrations using the Bio-Rad protein assay kit, the sample was boiled with loading buffer for 10 minutes. For western blot analysis, the protein was separated on 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis gels for 110 minutes at a voltage of 100 V, followed by a transfer to the polyvinylidene fluoride membrane. The membrane was blocked with 5% nonfat-dried milk in Tris-buffered saline Tween (TBST) for 1 hour at room temperature, and subsequently incubated overnight at 4 °C with primary antibodies (1:1000): p-Stat 3, IL-6, COX-2, NF-κB, Bcl-2, and β-actin, respectively. After washed with TBST, the membrane was continuously incubated with secondary antibody (1:5000) for 1 hour at room temperature. Finally, the protein band of each sample was visualized using the chemiluminescent agents.
Statistical Analyses
All data were represented as mean ± SEM and analyzed using one-way analysis-of-variance test.
Supplemental Material
online supplementary file 1 - Supplemental material for Synergistic Anticancer Effect of Gemcitabine Combined With Impressic Acid or Acankoreanogein in Panc-1 Cells by Inhibiting NF-κB and Stat 3 Activation
Supplemental material, online supplementary file 1, for Synergistic Anticancer Effect of Gemcitabine Combined With Impressic Acid or Acankoreanogein in Panc-1 Cells by Inhibiting NF-κB and Stat 3 Activation by Sen Jiang, Dong-Li Li, Jie Chen, Xi Zheng, Pan-Pan Wu, Chen Li, Xue-Tao Xu and Kun Zhang in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the Youth Foundation of Wuyi University (No: 2017td01), Foundation from Department of Education of Guangdong Province (No: 2017KSYS010, 2016KCXTD005, 2017KZDXM084, 2019KZDZX2003 and 2019KZDXM035).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
