Abstract
To evaluate the relationship between tree peony cultivars and the quality consistency of Cortex Moutan, a sensitive, reliable, and validated method based on high-performance liquid chromatography-electrospray ionization/mass spectrometry was developed for the quantitative analysis of paeonol and chemical fingerprinting of Cortex Moutan. Results from quantitative analysis showed that the content of paeonol in Paeonia ostii “Feng Dan” was the highest (24.51 ± 0.83 mg/g), followed by Paeonia suffruticosa “Luoyang Hong” (14.29 ± 0.76 mg/g), P. suffruticosa “Taiping Hong” (13.99±1.13 mg/g), and P. suffruticosa “Zhaofen” (13.08±0.85 mg/g). Paeonia ostii “Luoyang Feng Dan” was found to have the lowest content (8.76±0.46 mg/g) of paeonol. In qualitative analysis, 5 tree peony cultivars collected from different plantations in China were used to establish the fingerprint. For the fingerprint analysis, 17 characteristic peaks were used to evaluate similarities among tree peony cultivars, and they were found to show similarities. In short, the results of quantitative and qualitative analyses suggested that there was no significant difference in the chemical composition of Cortex Moutan from different tree peony varieties; however, there were significant differences in the levels of chemical components. The method developed in this study provides an important reference to establish a quality control method for other related traditional Chinese medicinal preparations.
Paeonia suffruticosa Andrews, commonly called the tree peony, belongs to the Ranunculaceae family and is native to China. 1 The root bark of tree peony is called Cortex Moutan and is an important crude drug that is widely used in traditional Chinese medicine. 2 According to the theory of traditional Chinese medicine, Cortex Moutan has the properties of clearing excessive heat, cooling the blood, promoting blood circulation, and removing blood stasis without inducing bleeding. 3 Modern pharmacological studies show that Cortex Moutan has anti-inflammatory effects. It has been frequently used as a therapeutic remedy for cardiovascular disorders, extravasated blood, stagnated blood, and genital diseases in women. 4 -7 The Chinese Pharmacopeia stipulates that the original plant for Cortex Moutan is P. suffruticosa (s.l.), which includes P. ostii and P. suffruticosa. Paeonia suffruticosa (s.l.) is cultivated in different areas and is known by vernacular names. Cortex Moutan is known as “Feng Danpi” in Tongling (Anhui province), “Chuan Danpi” in Dianjiang (Sichuan province), and “Cao Danpi” in Heze (Shandong province). 8 Compared with other peony varieties, “Feng Danpi” is considered a Daodi medicinal material and is widely recognized as having therapeutic benefits. 9 Quality criteria for Cortex Moutan in the Chinese Pharmacopeia and other East Country Pharmacopeias rely on only a single or a few constituents as chemical makers, such as paeonol. 8,10 Pharmacological evidence suggests that paeonol has antibacterial, antiaggregation, antianxiety, and anti-inflammation effects. 8,10 -14 In fact, unlike single chemical entities aimed at a specific single target, traditional Chinese medicines are complex mixtures containing hundreds of diverse chemical constituents. 15 Therefore, traditional Chinese medicines are too complicated to investigate comprehensively their quality consistency effectively with a single detection technique. 16 -18 As a herbal medicine with complicated compounds, the quality evaluation and quality control of Cortex Moutan tends to be a difficult task for modern researchers and traditional Chinese medicine practitioners alike. Fingerprint analysis has been used successfully as a powerful quality control tool for multicomponent herbal extracts and medicine. Several methods have been reported for the analysis of Cortex Moutan including thin-layer chromatography, high-performance liquid chromatography (HPLC), capillary gas chromatography, and liquid chromatography (LC)-mass spectrometry (MS)/MS. 8 HPLC-electrospray ionization (ESI) tandem/MS has been used for the structural analysis of chemical constituents in herbal extracts and has demonstrated a superior advantage over other methods owing to its high sensitivity, rapidity, and low sample consumption. 19 In this study, to understand more comprehensively the chemical composition of Cortex Moutan from different areas, the chemical components of 5 varieties of Cortex Moutan were systematically quantitatively and qualitatively analyzed, and their differences compared using the fingerprint technique. The results of this study will help select the best quality of Cortex Moutan from different origins and also provide basic data for the establishment of a more comprehensive, rapid, and effective method for quality control to guarantee its efficacy in clinical applications.
Materials and Methods
Sample Collection and Pretreatment
Paeonia ostii “Feng Dan” belongs to the section Moutan of the genus Paeonia and the family Paeoniaceae. Five-year-old P. ostii “Feng Dan” plants, which are the raw material of Daodi medicinal material, were collected from Phoenix Mountain Peony Garden, Tongling city, Anhui province, China. Paeonia ostii “Feng Dan” seedlings were grown in Anhui province for 2 years and then transplanted in Luoyang city for 3 years. These 5-year-old peony samples were collected from the peony cultivation base of the National Flower Park of China, Luoyang, Henan, China. In order to distinguish them from the above peony variety, we named them as P. ostii “Luoyang Feng Dan”. In essence, P. ostii “Luoyang Feng Dan” and P. ostii “Feng Dan” are the same peony varieties, but their origins are different. Paeonia ostii in Tongling city and Luoyang city were named P. ostii “Feng Dan” and P. ostii “Luoyang Feng Dan”, respectively, in this study.
Five-year-old P. suffruticosa “Luoyang Hong” plants were also collected from the peony cultivation base of the National Flower Park of China, Luoyang city, Henan province, China. Five-year-old P. suffruticosa “Zhaofen” plants were collected from Caozhou Peony Garden, Heze City, Shandong province, China. Five-year-old P. suffruticosa “Taiping Hong” plants were collected from Dianjiang Peony Garden, Chongqing City, China. A total of 30 individuals of each kind of tree peony were collected. The roots of the peony plants mentioned above were peeled, and the internal cores were eliminated according to the method of use in the Chinese Pharmacopeia (2010 edition, Vol.1, p. 160-161). The remaining material, called Cortex Moutan, was immediately dried at 40 °C for 24 hours. 20 The dried Cortex Moutan materials were ground and strained using a 60-mesh screen. The ground sample (0.5 g) was transferred to a conical flask (total volume 100 mL) and extracted with methanol (25 mL) by vortexing for 30 seconds, followed by continuous ultrasonication for 30 minutes (frequency 50 kHz and power of 100 W). The resultant stock solutions were centrifuged at 8000 rpm for 10 minutes (Anke TCL-16G desktop high-speed centrifuge, Shanghai Anting Scientific Instrument Factory, Shanghai, China). The supernatants were stored at 4 °C and filtered through a 0.45-µm membrane filter before HPLC-MS analysis.
Chemicals
Standard paeonol (Cat.0708‐0504) was purchased from National Institutes for Food and Drug Control (Beijing, China) and methanol (HPLC grade) and formic acid (HPLC grade) from Tianjin Kermel Chemical Reagent Co., Ltd (Tian Jin, China). Ultra-pure water was produced using a Milli-Q system (Millipore, Bedford, MA, USA). All solvents and samples were filtered through a 0.22-µm filter (Xinjinghua Co., Shanghai, China) before injecting into the HPLC.
Instrumentation and Chromatographic Conditions
The HPLC system consisted of a Surveyor Autosampler and an Accela 600 LC pump (Thermo Fisher Scientific, San Jose, CA, USA). The separation of the compounds was carried out using a gradient elution program at a flow rate of 0.2 mL/min, at 30 °C using a Hypersil GOLD AQ C18 (250 mm × 4.6 mm, 5 µm i.d.) column supplied by Thermo Fisher (Thermo Fisher Scientific, San Jose, CA, USA). The injection volume used for HPLC was 10 µL, and the mobile phase consisted of water:formic acid (99.9:0.1, v/v) (A) and methanol (B). The following linear gradient was applied: 0-5.0 minutes, A (90%),B (10%);5.0-20.0 minutes, A (90%~20%), B (10%~80%); 20.0~50.0 minutes, A (20%), B (80%); 50.0~55.0 minutes, A (20%~90%),B (80%~10%);55.0~60.0 minutes, A (90%),B (10%), followed by re-equilibration of the column for 2.5 minutes before the next run. The HPLC was connected to a TSQ Quantum Access Max triple-quadrupole mass spectrometer (Thermo Fisher Scientific, Basel, Switzerland) equipped with an ESI operating in the positive mode. The quantitative determination of paeonol in Cortex Moutan was based on HPLC-MS. Peaks were detected in the positive ion mode of MS detection (ESI+). ESI-MS conditions were as follows: nitrogen was used as a drying gas for solvent evaporation. The vaporizer and capillary temperatures were maintained at 250 °C and 350 °C, respectively. The peak width was set at 0.7, and sheath gas (nitrogen) was set to 42 arbitrary units. The auxiliary gas was set to 9 arbitrary units and the ion spray voltage to 3.1 kV. The full-scan mass covered the range from m/z 100-1000. Qualitative analysis of the main active components of Cortex Moutan was based on HPLC-MS/MS. Argon was used as the collision gas and the scan time was 0.2 minutes. For positive-ion analyses, the spray voltage, collision energy, and collision gas pressure used were 3100 V, 35 eV, and 1.5 m Torr, respectively. In the negative ionization mode, the spray voltage, collision energy, and collision gas pressure were 3000 V, 30 eV, and 1.5 m Torr, respectively.
Standard Solutions and Calibration
A stock solution of paeonol was prepared by dissolving 10.24 mg paeonol in 25 mL of the mobile phase (stock working solution). Dilution of the stock solution with methanol yielded the working solution at concentrations of 0.5, 2, 5, 10, and 20 µg/mL. Triplicate 20-µL injections were made for each concentration and chromatographed using the conditions described above. The regression curve was obtained by plotting concentration versus peak area.
Tandem Mass Spectrometric Fingerprint Analysis
Data analysis was performed using the Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine software (Version 2004A) according to the recommendation made by the State Food and Drug Administration (SFDA) of China, which is mainly used in the similarity study of chromatographic and spectral patterns. In our study, this software was used to synchronize chromatographic peaks and to calculate the correlation coefficients between entire chromatographic profiles, as well as to compute and generate the mean chromatogram as a representative standard fingerprint chromatogram from a group of chromatograms. The similarities between chromatographic patterns were analyzed among tested samples.
Results and Discussion
Optimization of Chromatographic Conditions for Fingerprint and Quantitative Analysis
Chromatographic conditions including column, column temperature, mobile phase composition, gradient elution procedure, and flow rate were optimized. Mobile phases (methanol-water and acetonitrile-water) containing different concentrations (0.05%, 0.1%, 0.2%) of acids (formic, acetic, phosphoric acids) were compared, indicating that methanol-0.1% formic acid generated better chromatographic separation using the following linear gradient: 0-5.0 minutes, A (90%),B (10%);5.0-20.0 minutes, A (90%~20%), B (10%~80%); 20.0~50.0 minutes, A (20%), B (80%); 50.0~55.0 minutes, A (20%~90%),B (80%~10%);55.0~60.0 minutes, A (90%),B(10%). A Thermo Hypersil GOLD AQ C18 column and a Waters Xbridge C18 column were compared, and the former was finally selected owing to its higher resolution. Meanwhile, the optimal column temperatures among 25 °C, 30 °C, 35 °C, and 40 °C were studied and 30 °C was finally selected. Different flow rates were considered and 0.2 mL/min proved to be optimal.
Validation of the Quantitative Analysis
Linearity
In this study, an HPLC-ESI/MS calibration curve was constructed using 5 different concentrations ranging between 0.5 and 20 µg/mL. Three replicates of each concentration were used. The assay was performed according to the experimental conditions previously described. Second-order polynomial calibrations of HPLC-ESI/MS (peak area against the corresponding concentration) were observed in the range of 0.5-20 μg/mL. The data provided a linear calibration following the equation: Y = A + BX, where Y is the value of the peak area, X represents the concentration of the standard solution, A is the intercept, and B is the slope. The regression equation was Y = 3.12948 × 106 X−3.88882, and the regression coefficient r 2 was 0.9999.
Range
The calibration range was established by considering the practical range necessary based on paeonol concentrations present in Cortex Moutan extracts to give precise, accurate, and linear results. The calibration range was from 0.5 to 20 μg/mL. The limit of detection (LOD) was defined as the lowest concentration of paeonol that gave an average signal to noise ratio greater than 3 over 5 replicate injections based on peak area and was determined to be 0.2 µg/mL.
Precision
The precision was evaluated using 5 repeated injections of the sample solution. The relative SD (RSD) value of the peak area was determined to be 0.3024% (n = 5).
Selectivity
Paeonol could be separated without the interference of other compounds in the samples (Figure 1). Peak identification was carried out using a direct comparison of the retention time, precise molecular mass, and the MS/MS data between the analyte and the authentic standard. The retention time of the chromatographic peak of paeonol standard sample was 31.86 minutes.

Total ion chromatograms in the positive ion mode of extracts of Cortex Moutan from different tree peony cultivars. (A) Cortex Moutan from Paeonia ostii “Feng Dan”, (B) Cortex Moutan from Paeonia suffruticosa “Luoyang Hong”, (C) Cortex Moutan from Paeonia ostii “Luoyang Feng Dan”, (D) Cortex Moutan from Paeonia suffruticosa “Taiping Hong”, and (E) Cortex Moutan from Paeonia suffruticosa “Zhaofen”.
Accuracy
To determine the accuracy, a known amount of paeonol was added to the Cortex Moutan samples, and the resultant solutions were analyzed using the valid HPLC-ESI/MS method. Results are shown in Table 1. The recovery of paeonol was between 89.9% and 96.7%, and RSD was between 2.22% and 2.55%.
Recovery Studies of Paeonol Added to Selected Samples of Cortex Moutan.
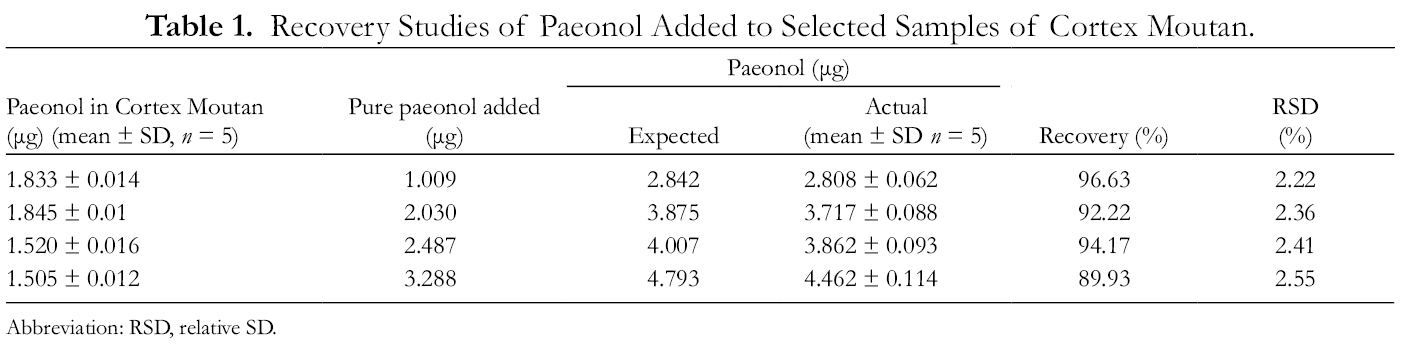
Abbreviation: RSD, relative SD.
Stability
To evaluate the stability, the Cortex Moutan extract and paeonol standard were dissolved in the mobile phase, stored in the autosampler vials for 30 hours, and then assayed. No significant difference in the mean results and no chromatographic changes were observed.
Differences in the Contents of Active Ingredient Contents Among Tree Peony Cultivars
In this study, samples of 5 tree peony varieties were collected from different medicinal peony producing areas in China. Thirty tree peonies were collected from each tree peony variety and analyzed. The contents of paeonol in 5 kinds of Cortex Moutan from different tree peony cultivars of China were determined using the above developed HPLC-ESI/MS method following the same sample preparation method. The results are shown in Table 2. Paeonol was the main component isolated from the root bark of P. suffruticosa. The contents of paeonol in the 5 types of Cortex Moutan ranged from 8.76 to 24.51 mg/g D.M (dry matter). The content of paeonol in Paeonia ostii “Feng Dan” was the highest (24.51 ± 0.83 mg/g), followed by P. suffruticosa “Luoyang Hong” (14.29 ± 0.76 mg/g), P. suffruticosa “Taiping Hong” (13.99 ± 1.13 mg/g), and P. suffruticosa “Zhaofen” (13.08 ± 0.85 mg/g). The lowest content of paeonol was found in P. ostia “Luoyang Feng Dan” (8.76 ± 0.46 mg/g). In our study, the content of paeonol in P. ostii “Feng Dan” was found to be the highest among the 5 kinds of Cortex Moutan. These levels are consistent with that of P. ostii “Feng Dan” used clinically. In fact, in China, Cortex Moutan produced from P. ostii “Feng Dan” is considered a Daodi medicinal material. As the essence of Chinese medicinal materials, Daodi medicinal materials have been widely used and have proven to be efficacious. Our results show that the method using paeonol content as an index to evaluate the quality of Cortex Moutan has a certain rationale, although not very comprehensive. In addition, P. ostii “Luoyang Feng Dan”, which was grown in Luoyang for 3 years and comes from the same variety as P. ostii “Feng Dan”, had the lowest content of paeonol (8.76 ± 0.46 mg/g). This finding suggested that the geographical environment of the growth of tree peony had an important effect on the accumulation of paeonol in Cortex Moutan.
Content of Paeonol in 5 Kinds of Cortex Moutan From Different Tree Peony Cultivars of China.

HPLC-Tandem Mass Spectrometric Qualitative Analysis
Characterization of chemical constituents in Cortex Moutan
HPLC-ESI-MSn was used to analyze the chemical constituents of Cortex Moutan, and the typical total ion chromatography profiles were generated in the positive and negative ion modes. By comparison, the positive ion mode responded much better than the negative. The ESI-MS (positive ion mode) full scan analysis of Cortex Moutan showed abundant protonated molecules and the molecular weight of each peak was also obtained. As shown in Figure 2, 17 peaks were detected in Cortex Moutan extract. The structures were identified using their MS and MS/MS spectra data and by comparing the generated data with previous reports. The identification results are shown in Table 3.
Comparison of fingerprints and similarity analysis among different samples

Representative total ion chromatogram in the positive ion mode of Cortex Moutan extract. 1: Mudanoside A; 2: Catechin; 3: Paeoniflorin; 4: 3,4,5-trihydroxybenzoic acid 3-O-apiofuranosyl (1→6)-gIucoypranoside; 5: 1.2.3.6-tetra-O-gaIIoyI-beta-d-gIucose; 6: Paeonoside C15H20O8; 7: PaeonifIorin; 4”-hydroxy, 6’-(4- Hydroxybenzoyl); 8: Paeoniflorin C23H28O11; 9: not identified; 10: Benzoylpaeoniflorin; 11: not identified; 12: not identified; 13: catechin glucoside; 14: 4-O-ethylpaeoniflorin; 15: Benzoylpaeoniflorin; 16: Paeoniflori -genone; 17: Paeonol.
The Identification Results of Chemical Composition of Cortex Moutan.

To standardize the fingerprints, we analyzed 5 batches of samples from different geographical regions using our optimized HPLC-ESI/MS method. The Similarity Evaluation System for Chromatographic Fingerprint of Traditional Chinese Medicine (Version 2004A) software was used to evaluate chromatograms. After carefully analyzing the fingerprint profiles of these samples, 17 common peaks with acceptable peak height and good resolution were assigned as common peaks. A simulative median chromatogram was generated as a reference. Fingerprints of the 5 batches of Cortex Moutan are illustrated in Figure 3. The similarities among the chromatograms of the 5 batches of samples to the reference fingerprints were calculated. Based on the results of qualitative analysis, it can be seen that the similarity values of all 5 batches of Cortex Moutan are higher than 0.904 (Table 4). It can be seen that the main characteristic peaks are the same in all samples. These results suggest that there are no significant differences in the types of active components of Cortex Moutan prepared from the 5 tree peony varieties. In addition, the 17 common peaks detected using HPLC-MS fingerprints were useful in assessing the quality of the Cortex Moutan.

High-performance liquid chromatography-electrospray ionization/mass spectrometry chromatographic fingerprints of Cortex Moutan from different tree peony cultivars. S1, Cortex Moutan from Paeonia suffruticosa “Zhaofen”; S2, Cortex Moutan from Paeonia suffruticosa “Taiping Hong”; S3, Cortex Moutan from Paeonia ostii “Luoyang Feng Dan”; S4, Cortex Moutan from Paeonia suffruticosa “Luoyang Hong”; S5, Cortex Moutan from Paeonia ostii “Feng Dan”; R, simulative median chromatogram.
Similarity Results of 5 Batches of Cortex Moutan From Different Tree Peony Cultivars.

S1, Cortex Moutan from Paeonia suffruticosa “Zhaofen”; S2, Cortex Moutan from Paeonia suffruticosa “Taiping Hong”; S3, Cortex Moutan from Paeonia ostii “Luoyang Feng Dan”; S4, Cortex Moutan from Paeonia suffruticosa “Luoyang Hong”; S5, Cortex Moutan from Paeonia ostii “Feng Dan”; R, simulative median chromatogram.
Conclusions
In this study, a reliable and sensitive HPLC-ESI/MS method was developed for the routine quantification of paeonol from P. suffruticosa Andrews. This method has been used to determine the content of paeonol in Cortex Moutan from different geographical regions. Results showed that P. ostii “Feng Dan”, a Daodi medicinal material, had the highest content of paeonol. In addition, the HPLC-ESI/MS fingerprint of Cortex Moutan was generated. For fingerprint analysis, 17 characteristic fingerprint peaks were used to evaluate the similarities among the 5 batches of tree peony cultivars and were found to show good similarities. To summarize, the results of quantitative and qualitative analysis of the chemical components of Cortex Moutan from different regions suggested that there were no significant differences among the chemical composition of Cortex Moutan from different tree peony varieties; however, there were significant differences in the levels of chemical components.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: National Natural Science Foundation of China (31302133).
