Abstract
In this study, valuable polyhydroxynaphthoquinone (PHNQ) pigments were recovered from sea urchin food waste and were investigated as a potential bioactive ingredient for cosmeceuticals. The crude PHNQ pigment extract from 4 Vietnam sea urchins, Diadema setosum, Diadema savignyi, Stomopneustes variolaris, and Tripneustes gratilla, exhibited effective 2,2-diphenyl-1-picryl-hydrazyl-hydrate scavenging activity, tyrosinase inhibitory activity, and antibacterial activity. The moisturizing cream with 0.5% of PHNQ pigments from D. setosum and Tripneustes gratilla sea urchins showed no dermal irritation over 14 days of mouse skin test. Four major active components in PHNQ were identified via high-performance liquid chromatography coupled with diode array detector and mass spectrometry. Echinochrome A contributed considerably to the antioxidant activity of the extracts while those containing echinochrome A and spinochrome E were significantly active against various bacteria. The promising results laid the foundation for establishing a novel process from food waste to innovative biomaterial and formulating eco-friendly skincare products with PHNQ components from sea urchins as precious ingredient.
Cosmetics were defined as care substances for cleansing, enhancing attractiveness, and changing the human appearance without altering the body’s structure or process. 1 For a long time, the utilization of synthetic chemicals in manufacturing cosmetics posed undesired issues. The inappropriate formulation of composite based on synthetic components induced product instability and generated side effects like skin irritation, photodermatitis, comedogenicity, carcinogenicity, and eczema. 2 -4 Thus, the use of natural active ingredients possessing clear origin, compositions, interactive mechanisms with the skin, and supplemental health benefits garnered widespread attentiveness in current cosmetic industry. Kligman in his study introduced the term “cosmeceuticals” as cosme(tic) products with pharma(ceutical) benefits. 5 The cosmeceuticals such as creams, lotions, and ointments contained bioactive ingredients (eg, phytochemicals, essential oils, vitamins, and antioxidants), which not only improved skin beauty but also were mainstay of treating, mitigating, and preventing skin diseases. 6 Rising demand for cosmeceuticals has promoted manufacturers to replace synthetic chemicals by naturally derived ingredients in production process. Among the published bioactive ingredients in cosmeceuticals (eg, ulvan, 7 sulfate galactofucan, 8 curcumin 9 ), phenolic compounds attracted considerable attention. The compounds found popularly in anti-aging and whitening skin products were considered to enhance antioxidative activity and skin protection against ultraviolet (UV) radiation. 10 -12 In recent years, hundreds of phenolic compounds were determined and quantified from plant species. Flavonoids and gallic acid were phenolic compounds extracted from terrestrial species while marine organisms like algae contained mainly phlorotannin class of polyphenol compounds. 13 Interestingly, phenolics extracted from shells and spines of sea urchins had structures similar to those extracted from terrestrial plants. 14 Typically, the quinonoid pigments called polyhydroxynaphthoquinones (PHNQs) in many sea urchin species were reported to possess several valuable bioactivities such as antimicrobial, antioxidative, and cardioprotective activity. 15 -18 From Scaphechinus mirabilis, the authors successfully isolated echinochrome A (Ech A), which is the active substance of the new drug Histochrome for the treatment of myocardial infarction and eye diseases. 19 Besides, Powell et al in their study also reported that quinonoid pigments from Psammechinus miliaris urchins displayed high anti-UV activity. 17
In Vietnam, the urchin species were diverse and distributed widely across the sea. In Khanh Hoa province, Diadema setosum (Leske), Tripneustes gratilla (Linnaeus), Stomopneustes variolaris (Lamarck), and Diadema savignyi (Audouin) were the most abundant species. According to a national survey program, production of edible urchins in Vietnam in the period of 1996‐2000 reached over 500 tons per year. This corresponded to the fact that huge amount of waste materials (eg, shell, spine) containing valuable quinonoid substances were discharged into the environment.
In this research, valuable quinonoid pigments were recovered from shell and spine of 4 different sea urchin species and were studied for the first time as a potential bioactive ingredient in cosmeceuticals. Quinonoid compounds were fractionated and the compositions were determined by high-performance liquid chromatography coupled with diode array detector and mass spectrometry (HPLC-DAD-MS). PHNQ pigments were examined for their antioxidative activity by 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) radical scavenging test, antimicrobial activity against Candida albicans yeast and 7 Gram-negative and Gram-positive bacteria, and inhibition of tyrosinase enzyme activity. The moisturizing creams with high bioactive PHNQ pigments were tested for dermal irritation on mouse skin. The results provided database for further formulating novel skincare products with quinonoid pigments from sea urchins.
Experimental
Materials
The sea urchin species D. setosum (Leske, 1778), D. savignyi (Audouin, 1809), Tripneustes gratilla (Linnaeus, 1758), and Stomopneustes variolaris (Lamarck, 1816) were collected at a depth of 5 m (Vietnam, Khanh Hoa province, Nhatrang Bay, 12°11 n, 109°14 E and 12°15 n, 109°20 E ) (Supplemental Figure S1). These species were identified by morphological method.
The reagents utilized for this study were all purchased from Sigma-Aldrich Co., which included sulfuric acid (H2SO4), chloroform (CHCl3), ethyl acetate (EtOAc), ethanol (EtOH), and DPPH, and tested microorganisms are Bacillus cereus (ATCC-11778), Staphylococcus aureus (ATCC-25923), Listeria monocytogenes ATCC 19111, Escherichia coli (ATCC-25922), Klebsiella pneumonia (ATCC-700603), Salmonella typhinerium (ATCC-14082), Pseudomonas aeruginosa (ATCC-27853), and Candida albicans (ATCC-10231). Tested mice were BALB/c strain.
Extraction of Quinonoid Pigments
The internal organs of the urchins were eliminated. The shells and spines were cleaned with tap water, dried in shade, and extracted exhaustively with ethanolic acid sulfuric 10% (v/v) at ambient temperature. The ethanol extract was concentrated in vacuo. The residue in water was partitioned respectively by chloroform (CHCl3) and ethyl acetate (EtOAc). After that, the CHCl3 and EtOAc fractions containing PHNQ were then combined, and concentrated under reduced pressure to collect crude PHNQ extract.
Determination of PHNQ Content
The total PHNQ content was quantified by UV absorbance method at 470 nm wavelength (UV-1800, Shimadzu, Japan) using a quartz cuvette (1 cm path length). Ech A was used as standard with a concentration range of 10‐50 µg/mL. All the experiments in this study were performed with 3 replicates.
PHNQ Pigment Identification
The crude PHNQ extract was dissolved in ethanol. The ethanol solution was filtrated by 0.2 µm cellulose acetate membrane (Advantec MFS, Inc., California, USA) prior to HPLC-DAD-MS analyses on an LC-MS-2020 instrument (Shimadzu, Japan) with a Discovery-HS-C18 column (3 µm size, 150 × 2.1 mm). A 0.5% solution A (acetic acid) and a solution B (acetonitrile containing 0.5% acetic acid) were applied following gradient elution as follows: 10%-40% B (0, 6 minutes), 40%-100% B (6, 11 minutes), 100% B (11, 12 minutes), 100%-10% B (12, 13 minutes) và 10% B (13, 17 minutes). The flow rate and the injection volume were fixed at 0.2 mL/min and 2 µL, respectively. The spectra were recorded on a diode array detector at λ = 254 nm.
DPPH Free Radical Scavenging Activity
The crude PHNQ extract was dissolved in methanol to obtain a stock solution (800 mg/L) for testing antioxidative activity. The DPPH radical scavenging capacity was performed pursuant to the method of Yen and Chen. 20 The UV absorption coefficient at 516 nm was determined using an ultraviolet-visible (UV-Vis) spectrophotometer (UV-1800, Shimadzu, Japan). Scavenging activity was calculated following the formula as below:
where Ac s the absorption of comparative sample (DPPH in ethanol solution), Am the absorption of sample (DPPH and sample in ethanol solution), and Amt absorption of blank (sample in ethanol solution).
The half maximal effective concentration (EC50) value was calculated based on the table curve program with the formula:
where
Antimicrobial Activity
The crude PHNQ extract was dissolved in ethyl acetate at a concentration of 25 mg/mL for testing antimicrobial activity. The PHNQ solution was screened based on a paper disc-diffusion assay following to El Masry HA et al. 21 In short, the PHNQ solution was infused at a concentration of 500 µg/disc onto sterile Whatman discs (6 mm diameter). After 1 hour under air hood, EtOAc solvent was evaporated. The antimicrobial activity was assessed against 8 human pathogens namely Gram-positive bacteria (Bacillus cereus ATCC-11778, Staphylococcus aureus ATCC-25923, and Listeria monocytogenes ATCC-19111), Gram-negative bacteria (Klebsiella pneumonia ATCC-700603, Salmonella typhimurium ATCC-14082, Escherichia coli ATCC-25922, and Pseudomonas aeruginosa ATCC- 27853), and yeast (Candida albicans ATCC-10231). These tested microorganisms were grown up on bacto nutrient agar, and the turbidity of suspensions was adjusted to 108 cells/mL utilizing a UV-Vis spectrophotometer (UV-1800, Shimadzu) at 625 nm wavelength. Similar procedure with pure EtOAc solvent and ampicillin was performed for negative and positive control, respectively. The plates were incubated for 24 hours at 37 °C and then the diameter of the inhibition zones was measured for activity comparison.
Inhibition of Tyrosinase Activity
The crude PHNQ extract was dissolved in DMSO for testing tyrosinase inhibition activity following the procedure stated by Park et al. 22 The test mixture was prepared by supplementing 70 units of mushroom tyrosinase (Sigma-Aldrich, USA), 0.5 mL of L-tyrosine (Merck, Germany) (0.1 mg/mL), and 0.5 mL of sodium phosphate buffer (pH 6.8) to 0.5 mL of PHNQ solution (250 µg/mL). The mixture was incubated for 15 minutes at 37 °C and then the absorbance at 490 nm was recorded using an UV-Vis spectrophotometer (UV-1800, Shimadzu). A similar mixture, in which the crude PHNQ extract was replaced by Kojic acid, was measured as the positive control. The half-maximal inhibitory concentration (IC50) (the PHNQ concentration that half the initial tyrosinase was inhibited) was recorded. The tyrosinase inhibition rate was computed as follows:
where T is the absorbance at 490 nm without test sample, and I is the absorbance at 490 nm with test sample.
Skin Irritation Test
Moisturizing cream was prepared following the formula published by Karemore et al 23 with some modification. In brief, cream samples with 0.5% of PHNQ pigment extracted from D. setosum and from Tripneustes gratilla were symbolled KM1 and KM2, respectively. The phases A, B, and C were prepared by weighting and mixing different ingredients with detailed contents described in Supplemental Table S1.
Phases A and B were heated to 75 °C ± 1 °C and then phase B was added slowly to phase A to form a homogeneous emulsion. The emulsion was stirred at 9000 rpm. Phase C was added when the temperature of the emulsion dropped to 45 °C ± 2 °C. The product was maintained at room temperature for stability in 1 hour and was stored in fridge at 5 °C for further skin irritation tests. The experiments on skin irritation were performed following the testing guideline 404 of the Organization for Economic Cooperation and Development with some modifications. In brief, 20 hours before the experiment, the back mice’ hair was cleansed to both sides of the spine with a sufficiently wide skin area of 2.5 × 2.5 cm (Supplemental Figure S3). All mice were housed in plastic cages at a temperature-controlled room on a 12-hour light/12-hour dark cycle. Mice were provided food and water ad libitum. Then, 0.2 mL of cream containing 0.5% of PHNQ pigment was applied gently to the tested skin area. The test skin area was observed and recorded the reaction phenomenon compared to the adjacent skin at 1 hour, 12 hours, 1 day, 4 days, 7 days, and 14 days after cleaning the cream. The observation period may be extended if deep lesions occurred to allow a more complete assessment of the healing or nonhealing of wounds but should not exceed 14 days. Skin irritation was evaluated based on the Draize scale. 24
Result and Discussion
Chemical Composition of PHNQ Pigment From Vietnam Sea Urchin Species
The composition and content of PHNQ pigment extracted from 4 species of Vietnam sea urchins are exhibited in Table 1. As shown in Table 1, the total PHNQ content from the sea urchins distributed in the range of 1910 ± 340‐6150 ± 510 µg/g. The PHNQ content from the urchin species in this study was much higher than those of the species in Okhotsk sea, Pacific Ocean 14 and E. mathaei species in Madagascar sea. 25 Besides differences in extraction conditions, it was supposed that the climatic conditions can affect the biosynthetic ability of the urchins in producing PHNQ pigments. 25
The Composition and Content of PHNQ Pigment Extract From the Vietnam Sea Urchins.

Abbreviations: Ech A, echinochrome A; PHNQ, polyhydroxynaphthoquinone; Sp A, spinochrome A; Sp D, spinochrome D; Sp E, spinochrome E.
Structural identification of the compounds contributing to PHNQ pigment was determined based on comparison of the retention time of the HPLC peaks, UV-Vis λmax of PHNQ sample in this paper with those of published PHNQ pigment (see Supplemental Table S2 for more details of referenced peaks). As the result, the peaks 1, 2, 3, and 4 were assigned to the compounds spinochrome E (Sp E), spinochrome D (Sp D), Ech A, and spinochrome A (Sp A), respectively (Supplemental Figures S2 and S3).
It can be seen from HPLC-DAD-MS spectra that the 2 sea urchins Stomopneustes variolaris and D. setosum possessed similar PHNQ composition with the occurrence of Sp E (26% and 6%, respectively) and Ech A (73% and 87%, respectively). Noticeably, there were 3 compounds in PHNQ pigment from D. savignyi (Sp E, Sp D, and Ech A). Compared to the 3 sea urchins above where Ech A was the main component of PHNQ pigment, Sp E was the major component of PHNQ pigment extracted from Tripneustes gratilla (72%), beside 26% of Sp A.
It is noticeable that the PHNQ pigment from D. setosum reported by Anderson et al 26 contained not only Ech A and Sp E but also Sp D and Sp A. Moreover, according to Brasseur et al, 25 there was not Sp E in composition of PHNQ pigment from D. savignyi while PHNQ pigment from Tripneustes gratilla contained Sp D and Ech A rather than Sp A. Despite divergency in composition due to growing in different geographical regions, the major component of PHNQ pigment in each urchin species remains unchanged, ie, Ech A in Diadema species and Sp E in Tripneustes gratilla.
Antioxidant Activity of PHNQ Pigment From the Sea Urchin Species
PHNQ pigment extracted from shells and spines of the sea urchins contained the phenolic compounds with several hydroxyl groups, which was considered to exhibit highly antioxidant activity. 26 According to Da-yong Zhou et al, 27 the activity intensity was contingent on the ratio of the components in PHNQ. In this research, the antioxidant activity of PHNQ pigments was investigated based on DPPH free radical scavenging assay and the results were displayed in Supplemental Table S3 and Figure 1.
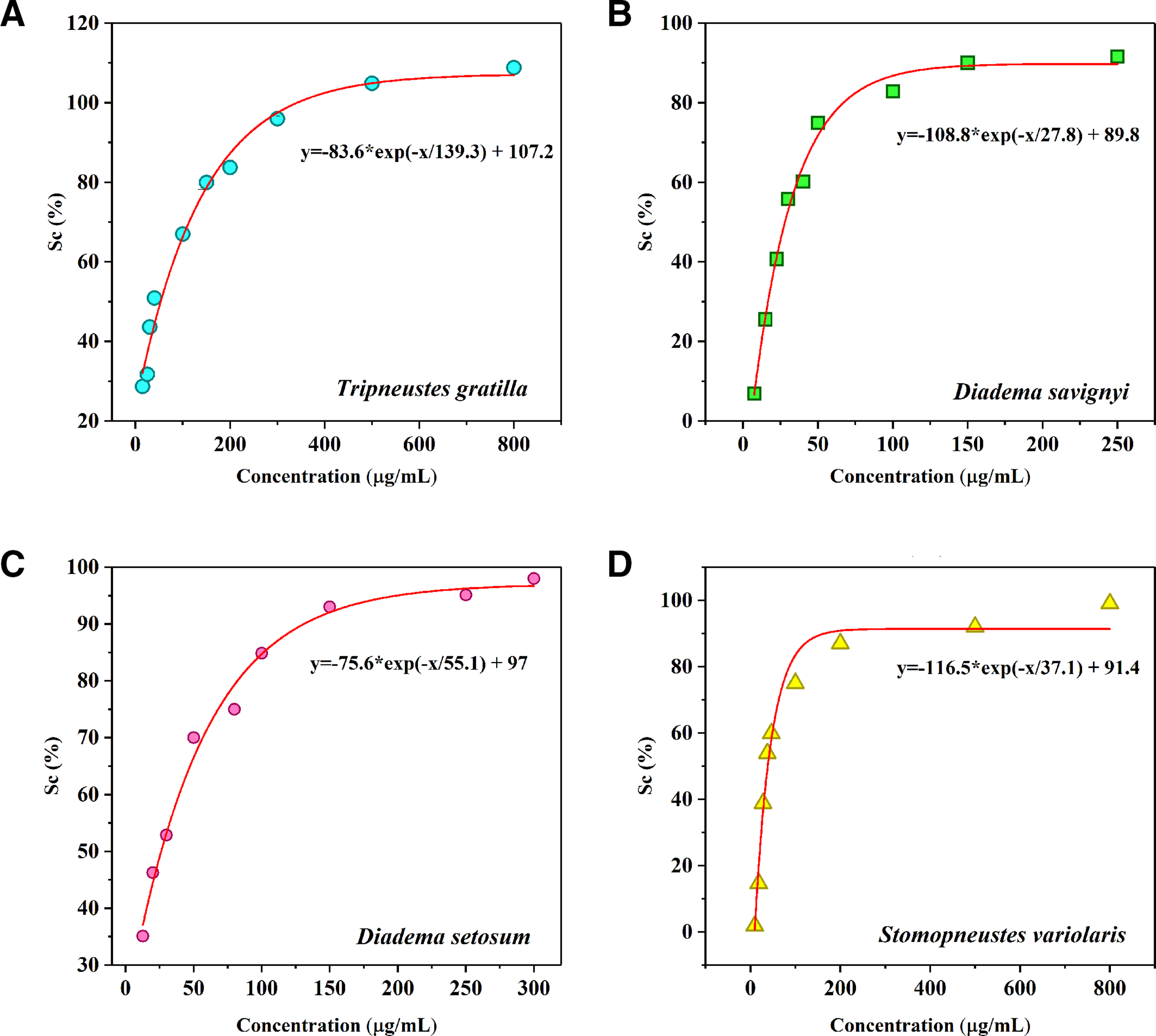
DPPH free radical scavenging activity of PHNQ pigment extracts from the four Vietnam sea urchins. DPPH, 2,2-diphenyl-1-picryl-hydrazyl-hydrate; PHNQ, polyhydroxynaphthoquinone.
It can be observed from Supplemental Table S3 that the PHNQ pigment from Tripneustes gratilla displayed the lowest antioxidant activity among the 4 sea urchins with an EC50 value of 58.47 µg/mL. Highest DPPH scavenging capacity belonged to the PHNQ pigments from the species of Diadema genus (EC50 = 28.07 µg/mL for D. savignyi and EC50 = 26.82 µg/mL for D. setosum).
There existed some published works on DPPH scavenging capacity of PHNQ extracted from different sea urchin species. To be specific, the PHNQ pigment extracted from Echinometra mathaei species with diethyl ether solvent included Sp A, Sp B, Sp C, and Ech A components and exhibited higher DPPH scavenging capacity than the commercial phenolic antioxidants butylated hydroxytoluene. 28 Li et al in their work 27 found that the PHNQ pigment extracted from Glyptocidaris crenularis contained Sp B, Sp D, and Sp E in composition, and was more active in DPPH scavenging than Strongylocentrotus intermedius PHNQ, which contained only Sp B (IC50 = 60 µg/mL compared to IC50 = 80 µg/mL, respectively). Vasileva et al 29 were successful in isolating the components in PHNQ extracted from the urchin species namely Scaphechinus mirabilis, Strongylocentrotus pallidus, and Mesocentrotus nudus. The components were examined for DPPH scavenging activity, which followed the order of echinamine B (EC50 = 6.5 µM) >Ech A >spinamine E >echinamine A >Sp E >α-tocopherol. Thus, it was supposed from our work that PHNQ pigments from D. setosum were significantly more antioxidative to those from other sea species via the highest contribution of Ech A in composition. From the experiment on DPPH scavenging capacity, the crude PHNQ extracts from the sea urchins expressed comparable antioxidant activity to PHNQ pigment from purple sea urchin Anthocidaris crassispina 30 and the polyphenolic components from edible plants, 31 which were applied previously in cosmetics as natural antioxidants.
Antimicrobial Activity of PHNQ Pigment From the Sea Urchin Species
The antimicrobial activity of PHNQ pigment from the sea urchin species against tested bacteria is exhibited in Table 2 and Supplemental Figure S4. Larger inhibition zone diameter demonstrated a more effective antimicrobial activity. The crude extract from the sea urchins demonstrated activity against all 7 bacteria but was ineffective against yeast Candida albicans. PHNQ extracted from D. setosum showed the highest performance inhibiting 6 per 7 bacteria except Bacillus cereus, whereas PHNQ extracted from D. savignyi expressed the lowest antibacterial activity against all the bacteria except Staphylococcus aureus. The outcome would originate from high contribution of Ech A in composition of the extracted PHNQ pigment (87% in PHNQ from D. setosum and 59% in PHNQ from D. savignyi). Component Ech A was reported previously by Service et al 32 that displayed strong activity inhibiting the growth of Pseudomonas strain 111, Vibrio fischeri, and Micrococcus sp. As stated by Stekhova et al in their study, 33 Ech A also exhibited outstanding performance against the bacteria species namely Trichophyton mentagrophytes and Staphylococcus aureus compared to Sp B, Sp C, and Sp D. The result suggested further orientation of producing natural antimicrobial products containing PHNQ pigment with Ech A as the major ingredient.
Antimicrobial Activity of PHNQ Pigment Extract From the Vietnam Sea Urchins Against Tested Bacteria.

Abbreviation: PHNQ, polyhydroxynaphthoquinone.
In addition, it was noticeable that the PHNQ pigment extracted from Stomopneustes variolaris inhibited significantly Bacillus cereus and PHNQ from Tripneustes gratilla acted markedly against Listeria monocytogenes and Escherichia coli. One common point from PHNQ extracted from the 2 urchins above was the high proportion of Sp E in their composition; thus, the component Sp E was assumed here to possess variable effect depending on bacteria strains. The result was consistent with the report of Brasseur et al, 25 in which the isolated Sp E were strongly active against E. coli (EC50 = 28.53 µM) with a minimum inhibitory concentration of 7.25 µg/mL but not relatively effective in case of Caloplaca marina and Shewanella oneidensis. In our study, the crude PHNQ extract from the sea urchins, in which 2 main components were Ech A and Sp E, exhibited antimicrobial activity against all the tested bacteria and were very promising in application as an antibacterial ingredient in cosmeceutical products.
Tyrosinase Inhibition Activity of PHNQ Pigment From the Vietnam Sea Urchins
When human skin was exposed to UV radiation, the melanocytes produced a protective pigment called melanin to block the UV light. However, redundancy of melanin in skin was considered to cause negative symptoms such as hyperpigmentation, tanning, age spots, and freckles. Tyrosinase is a rate-limiting enzyme involving in controlling the melanin generation. 34 Thence, in manufacturing process of skin care products, inhibitors of tyrosinase enzyme were commonly implemented to manipulate the formation of melanin. 35 In this study, the PHNQ crude extracts were tested for tyrosinase inhibition with a fixed concentration of 250 µg/mL and the results are shown in Table 3.
Tyrosinase Inhibition Activity of PHNQ Pigment Extracts From the Vietnam Sea Urchins.

Abbreviation: PHNQ, polyhydroxynaphthoquinone.
The most effective inhibitors seemed to be the PHNQ pigment extracted from D. setosum. The pigment can restrain 87.9% ± 4.1% of tyrosinase enzyme, which nearly reached the activity of whitening substance Kojic acid (88.5% ± 3.3%). In contrast, PHNQ pigment from Tripneustes gratilla showed the lowest performance when only 68.5% ± 2.9% of tyrosinase was inhibited. The result was correlated well with the antioxidant effect when the contribution of Ech A in the composition of PHNQ pigment was likely to determine the antioxidant and tyrosinase inhibition activity of the PHNQ pigments.
Skin Irritation Assessment
In order to evaluate the safety level of using PHNQ crude extracts in cosmeceuticals, 2 cream samples with PHNQ pigment in ingredient were assessed of skin irritation. Sample KM1 with PHNQ pigment extracted from D. setosum and sample KM2 with PHNQ pigment extracted from Tripneustes gratilla represented 2 components Ech A and Sp E, respectively. Erythema and edema symptoms were tracked at time intervals and the results are displayed in Supplemental Table S4, S5 and Figure S5.
From Supplemental Tables S4 and S5, the mouse expressed no irritation indication (skin erythema or edema) after treating with 0.2 mL of cream containing 0.5% of PHNQ pigment. After 14 days of assessment, the primary irritation index grade was still 0.
Conclusion
In this research, valuable PHNQ pigments were recovered successfully from food waste of the 4 most popular sea urchin species in Vietnam namely D. setosum, D. savignyi, Tripneustes gratilla, and Stomopneustes variolaris. HPLC-DAD-MS analysis was effective to determine the active composition of PHNQ pigments. The 4 crude PHNQ extracts all exhibited promising bioactivities without causing skin irritation, which were promising to be applied for replacing the synthetic chemicals in cosmeceuticals.
For the active components in PHNQ pigments, the PHNQ pigments with Ech A in composition were determined to exhibit highly antioxidant activity and those containing echinochrome A and Sp E were significantly active against various bacteria. The PHNQ comprising Ech A component can inhibit the growth of tyrosinase enzyme to a great extent. The PHNQ pigment from D. setosum species with Ech A as the major component was exceptional among those from other species when it exhibited the highest performance in all bioactivity tests. The interesting results were a promising suggestion for employing specific components of PHNQ into specific types of cosmeceuticals.
Supplemental Material
Supplementary Material 1. - Supplemental material for Polyhydroxynaphthoquinone Pigment From Vietnam Sea Urchins as a Potential Bioactive Ingredient in Cosmeceuticals
Supplemental material, Supplementary Material 1., for Polyhydroxynaphthoquinone Pigment From Vietnam Sea Urchins as a Potential Bioactive Ingredient in Cosmeceuticals by Vo Mai Nhu Hieu, Tran Thi Thanh Van, Cao Thi Thuy Hang, Natalia P. Mischenko, Fedoreyev Sergey A. and Hai Bang Truong in Natural Product Communications
Footnotes
Statement of Human and Animal Rights
Experiments were performed in accordance with Vietnamese ethical laws and European Communities Council Directives of November 24, 1986 (86/609/EEC) guidelines for the care and use of laboratory animals.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research is funded by Vietnam Academy of Science and Technology (Project number VAST06.01/19-20).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.



