Abstract
Flavescensin A (
Sophora flavescens Alton, known as “Kushen” in China, grows mainly in mainland China, Japan, Korea, Russia, and India. 1 The roots of S. flavescens is commonly used as the traditional Chinese medicine for the treatment of pruritus, dysentery, trichomonas vaginitis, eczema, and pyogenic infections of the skin. 1,2 Phytochemical studies have revealed that the main active constituents in S. flavescens are quinolizidine alkaloids, prenylated flavonoids, and pterocarpans. 3 -6 Pharmacological and chemical research on the quinolizidine alkaloids and prenylated flavonoids have been quite thorough, whereas study of the pterocarpans components remains relatively limited. However, pterocarpans have attracted much scientific attention because of their broad range of biological activities and unique structural characteristics. 7 Thus, the isolation of much more pterocarpans from medicinal plants is a worthwhile objective.
In the course of screening for anti-influenza virus and anti-inflammatory constituents from the root of S. flavescens, a novel rearrangement derivative of pterocarpan featuring a 6/5/5/6/5 pentracyclic ring system, named flavescensin A (

Chemical formulas of
Results and Discussion
Flavescensin A (
NMR data of 1 in Chloroform-d.
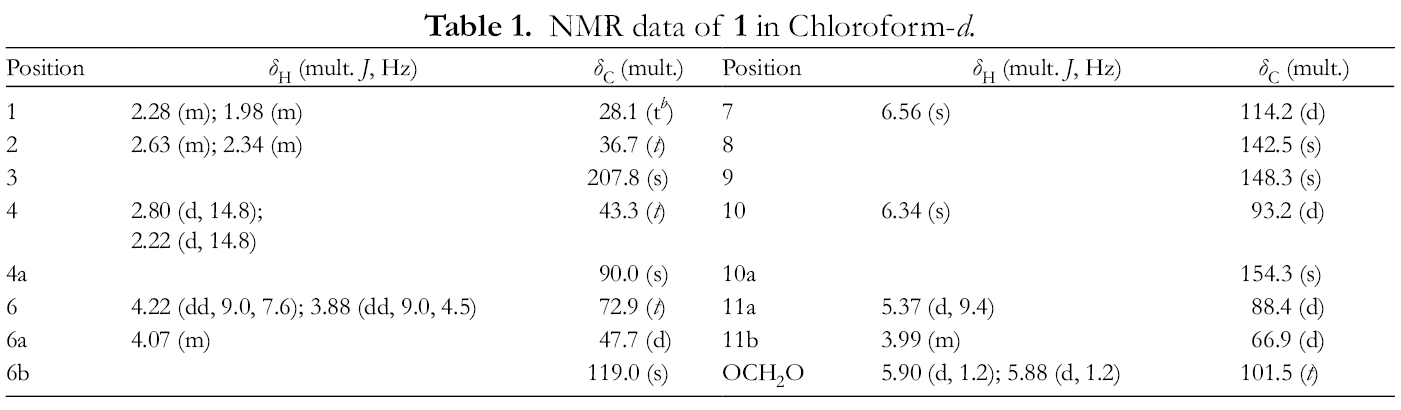
The above-mentioned NMR data indicated that

Key 2-dimensional-nuclear magnetic resonance correlations of compound 1. COSY, correlation spectroscopy; HMBC, heteronuclear multiple bond correlation; ROESY, rotating-frame Overhauser effect spectroscopy.
The relative configuration of compound
The absolute configuration of compound

Experimental and calculated electronic circular dichroism spectra of 1 in MeOH.
It is worth mentioning that flavescensin A (

Plausible biogenetic pathway of 1.
The 7 known compounds were identified as 11b-hydroxy-11b,1-dihydro-maackiain (
Compounds
Experimental
General Experimental Procedures
Optical rotations, Jasco DIP-370 digital polarimeter; ultraviolet (UV), Shimadzu UV-2401A spectrophotometer; IR, Tenor 27 spectrophotometer; CD, Applied Photophysics spectropolarimeter; 1D- and 2D-NMR, Bruker AVANCE III-600 spectrometer; HRESIMS, Agilent 6530Q of spectrometer; medium-pressure liquid chromatography (MPLC), Büchi Labortechnik AG CH-9230 Flawil; preparative high-performance liquid chromatography (HPLC), Agilent 1100; enzyme labeling instrument, Thermo Fisher Scientific; Silica gel, Qingdao Marine Chemical Factory; sephadex LH-20; mersham Biosciences AB; column, Soochow High Tech chromatography Co., Ltd.; ODS-C18, YMC Co., Ltd.; precoated silica gel plates, Qingdao Marine Chemical Factory.
Plant Material
The dried roots of Sophora flavescens were collected in June 2019 from Honghe County, Yunnan Province, China, and identified by one of the authors (Xuan-Qin Chen). A voucher specimen (number: KUMST20190608) has been deposited at the Key Laboratory of Phytochemistry (Kunming University of Science and Technology).
Extraction and Isolation
The dried roots of Sophora flavescens (5.0 kg) were extracted with 95% EtOH (3 × 40 L, 24 hours, each). The EtOH extracts were evaporated to dryness (450 g) under reduced pressure, suspended in distilled H2O, and partitioned with EtOAc and n-BuOH consecutively. The EtOAc fraction (124 g) was subjected to D-101 macroporous resineluting with EtOH-H2O (2:8 and 8:2) and obtained 2 fractions. The second part, the total flavonoid constituents (75 g) were subjected to silica-gel column chromatography (CC, 200‐300 mesh) eluting with petroleum ether-EtOAc (4:1-1:1) and got 9 fractions (F1-F9).
F1 (1.5 g) was separated by Sephadex LH-20 using MeOH as an eluent to afford 3 fractions (F1-1-F1-3). F1-2 (400 mg) was chromatographed over silica-gel column (200, 300 mesh) and eluted with petroleum ether–CH2Cl2 (1:1) to obtain 8 fractions (F1-2-18F1-2-8). F1-2-4 (22 mg) was subjected to silica gel CC using petroleum ether–EtOAc (20:1) as eluent to give
F2 (3.8 g) was fractionated using CH2Cl2–MeOH (20:1) to obtain 6 fractions (F2-1-F2-6). F2-4 (900 mg) was separated by Sephadex LH-20 using MeOH as an eluent to afford 3 fractions (F2-4-1-F2-4-3). F2-4-1 (80 mg) was chromatographed over silica-gel column (200, 300 mesh) and eluted with CH2Cl2–MeOH (60:1) to obtain 3 fractions (F2-4-1-1- F2-4-1-3). F2-4-1-1 (19 mg) was further separated by semipreparative HPLC (35% MeOH in H2O, t
R = 20.0 minutes, 3 mL/min, Agilent Zorbax SB-C18, 250 × 10 mm) to yield
F5 (2.6 g) was separated into 5 fractions (F5-1-F5-4). By silica-gel column (200, 300 mesh) eluting with CH2Cl2–MeOH (20:1) F5-3 (200 mg) was divided into 3 subfractions (F5-3-1-F5-3-3) by Sephadex LH-20 (MeOH). Compounds
F7 (3.5 g) was fractionated into 3 fractions (F7-1-F7-3). By Sephadex LH-20 using CH2Cl2–MeOH (1:1) F7-3 (180 mg) was divided into 4 subfractions (F7-3-1-F7-3-4). Compounds
Flavescensin A (
Anti-Influenza Virus Assay
In this study, influenza strain A/WSN/33/2009 (H1N1) was used. Oseltamivir as a positive control was used and purchased from LKT and Tszchem laboratories. The Madin–Darby canine kidney (MDCK) cells were cultured in Dulbecco’s modified Eagle’s medium (Gibco, Grand Island, New York, USA) supplemented with 1% streptomycin (10 000 µg/mL)-penicillin (10 000 U/mL), 10% (v/v) fetal bovine serum, and together with 10 mmol/L N-(2-hydroxyethyl)-piperazine-N′−2-ethanesulfonic acid (HEPES) in a humidified atmosphere with 5% CO2 at 37 °C. The anti-influenza virus activities of tested compounds were evaluated by previously reported methods. 15
Nitric Oxide Production Assay
The anti-inflammatory activities of all isolates were evaluated through measuring the level of nitrite accumulated in the culture medium. That was an important indicator of NO production according to the Griess reaction. RAW 264.7 cells (8 × 104 cells/well) were seeded onto 96-well plate and pretreated with 3.125, 6.25, 12.5, 25, and 50 µM of
Conclusions
From Sophora flavescens, 1 new compound (
Supplemental Material
Figure S1 - Supplemental material for A Novel Pterocarpan Derivative From the Roots of Sophora flavescens
Supplemental material, Figure S1, for A Novel Pterocarpan Derivative From the Roots of Sophora flavescens by Zhi-Jun Zhang, Guo-Xian Li, Dan Liu, Xuan-Qin Chen, Hong-Mei Li and Rong-Tao Li in Natural Product Communications
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported financially by the National Natural Science Foundation of China (Nos. U1602222 and 31960092), Yunnan innovation team (No. 2019HC018), and the Project funded by China Postdoctoral Science Foundation (No. 2019M653498).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
