Abstract
The Mediterranean fruit fly or medfly, Ceratitis capitata (Wiedemann) (Diptera: Tephritidae), is one of the most economically important invasive pests worldwide, with over 300 known hosts. Essential oils have great promise for application in integrated pest management, where they function as natural repellents, attractants, and toxicants with a reduced impact on the environment. In this study, we evaluated essential oil from aerial parts of the African ginger bush, Tetradenia riparia (Hochst.) Codd (Lamiaceae), as a potential new attractant for male C. capitata. Tetradenia riparia essential oil (TREO) was analyzed by gas chromatography (GC)-flame ionization detection and GC-mass spectrometry (GC-MS). The primary compounds identified were fenchone (15%), δ-cadinene (11%), 14-hydroxy-β-caryophyllene (8%), and tau-cadinol (7%). In short-range laboratory bioassays with sterile male medflies, TREO exhibited attractancy comparable to that observed with a positive control, essential oil from tea tree (Melaleuca alternifolia (Maiden and Betche) Cheel.). This study provides the first report of C. capitata behavioral response to TREO. Further research is needed, particularly with the 2 enantiomers of fenchone, to determine the chemical constituents responsible for the attraction of C. capitata.
Keywords
The Mediterranean fruit fly (medfly), Ceratitis capitata Wiedemann (Diptera: Tephritidae), is one of the most important pests of fruits and vegetables worldwide. Because of the economic importance of C. capitata and increasing incursions due to anthropogenic activities (global travel and commerce), new attractants are needed for early detection and monitoring of this invasive pest. 1,2 The success of Sterile Insect Technique for suppression of C. capitata populations relies on the mating success of sterile males. 3,4 Studies have shown that a variety of plant essential oils have a behavioral effect on sterile male medflies. Essential oils, complex blends of volatile terpenoids, have received considerable attention for their potential as sources of attractive kairomones for C. capitata. 5 -21 For example, males of C. capitata are strongly attracted to angelica seed oil, Angelica archangelica L., which was used successfully for pest surveillance and detection in the Florida eradication program during 1956-1957. 22,23 There are a growing number of studies on essential oils as alternatives for pest management, and oils from Citrus aurantium L., Zingiber officinale Roscoe, Leptospermum scoparium Forst. & Forst., Melaleuca alternifolia (Maiden and Betche) Cheel., and Piper cubeba L. have been found to contain components attractive to C. capitata. 6,7,9,16 -18,20,21 As part of ongoing research to identify new semiochemicals for management of C. capitata, this study evaluated Tetradenia riparia essential oil (TREO) for potential attraction.
Tetradenia riparia (Hochst.) Codd [syn. Iboza riparia (Hochst) N. E. Br., Moschosma riparia Hochst.], a member of the Lamiaceae, is native to southern Africa. Commonly known as a ginger bush, plants grow as shrubs or occasionally as small trees up to 3 m in height. 24 The foliage of T. riparia is highly aromatic. Plants are monecious with small, pinkish to white flowers produced in dense, showy panicles. Readily cultivated as an ornamental, T. riparia is grown under glass or outdoors in relatively frost-free areas. 25 Tetradenia riparia has been traditionally used as a medicinal plant for its antibacterial, antiseptic, antispasmodic, fungicidal, soporific, stomachic, and tranquilizing properties. Ethnomedicinal use of leaf infusions has included treatment of cough, cramps, dengue, diarrhea, fever, flu, malaria, mumps, and sore throat. Cases of poisoning from leaf infusions due to severe toxic inflammatory reactions of mucous membranes have been reported, but are rare. 26 -28
Results and Discussion
The hydrodistilled essential oil from aerial parts of T. riparia, collected from vegetative growth of greenhouse-grown plants, was analyzed by gas chromatography-flame ionization detection (GC-FID) and GC-mass spectrometry (MS). A total of 48 compounds were identified from TREO, representing 86.6% of the total composition. The main components were found to be fenchone (14.8%), δ-cadinene (10.6%), 14-hydroxy-β-caryophyllene (7.9%), and tau-cadinol (6.9%) (Table 1). Sesquiterpenoids constituted the largest contribution to the overall TREO (56.9%), with an abundance of sesquiterpene hydrocarbons (S) (29.0%) and oxygenated sesquiterpenes (OS) (27.9%), along with monoterpenoids (29.7%) composed of oxygenated monoterpenes (OM) (26.4%) and monoterpene hydrocarbons (M) (3.3%). Distribution of the main compound groups detected in the TREO is presented in Figure 1.
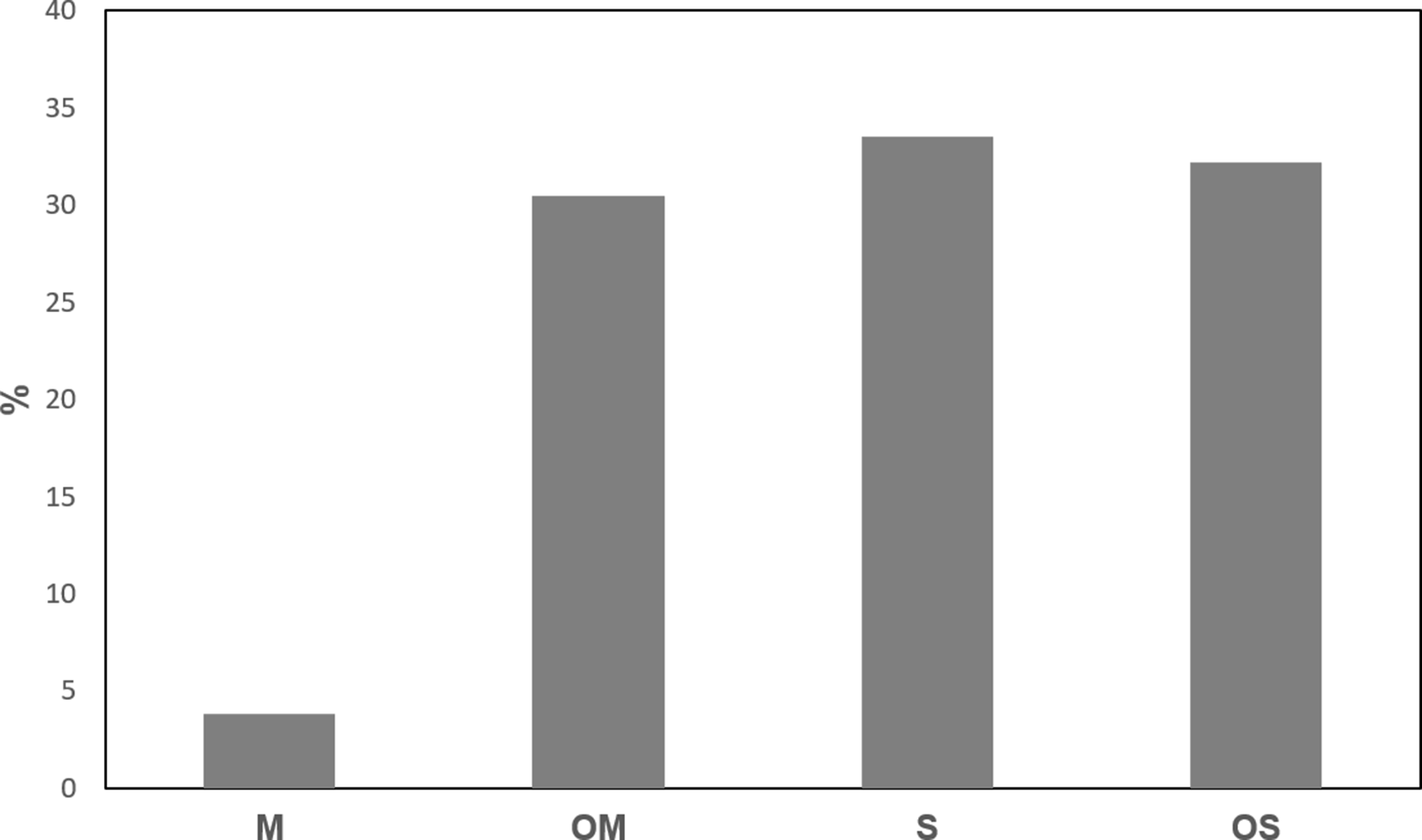
Distribution of the main compound groups detected in Tetradenia riparia essential oil. M, monoterpene hydrocarbons; OM, oxygenated sesquiterpenes; OS, oxygenated sesquiterpenes; S, sesquiterpene hydrocarbons.
Chemical Composition of the Essential Oil of Tetradenia riparia.

M, monoterpene hydrocarbons; MS, mass spectrometry; OM, oxygenated monoterpenes; OS, oxygenated sesquiterpenes; RRI, relative retention index; S, sesquiterpene hydrocarbons.
RRIs calculated against n-alkanes.
Percentage calculated from flame ionization detection data.
Mass spectrometry identified on the basis of computer matching of the mass spectra with those of the Wiley and MassFinder libraries and comparison with literature data.
In reviewing the literature, Fernandez et al examined the chemical composition of TREO in relation to seasonal variations. 29 Authors reported that the content of fenchone was higher in the summer (13.5%) and autumn (10.4%), whereas lower values were found in the spring (6%) and winter (2.9%). Our T. riparia sample was harvested from actively growing greenhouse plants in February, and the high amount of fenchone was similar to that found by Fernandez et al from field-grown plants in the summer. 29 This suggests that active plant growth under warmer temperatures is associated with the maximum fenchone composition of TREO. In another study, T. riparia leaf oil contained mainly fenchone (18.9%), followed by (E,E)-farnesol (17.7%) and aromadendrene oxide (17.3%). 30 These quantitative and qualitative differences between previous studies and our sample may be attributed to harvesting season, different geographic conditions, and environmental factors.
In short-range laboratory bioassays, the response of male C. capitata to TREO was assessed relative to response with a strong medfly attractant, tea tree oil (TTO), 21 and a mild medfly attractant, mastic gum essential oil (MGEO, 3 sources) 31 (Figure 2). There were significant differences in mean attraction of males among the 5 treatments (F = 10.543; df = 420; P < 0.001). The level of attraction observed with TREO was equivalent to that observed with TTO, and both essential oils were significantly more attractive than the 3 MGEO treatments.
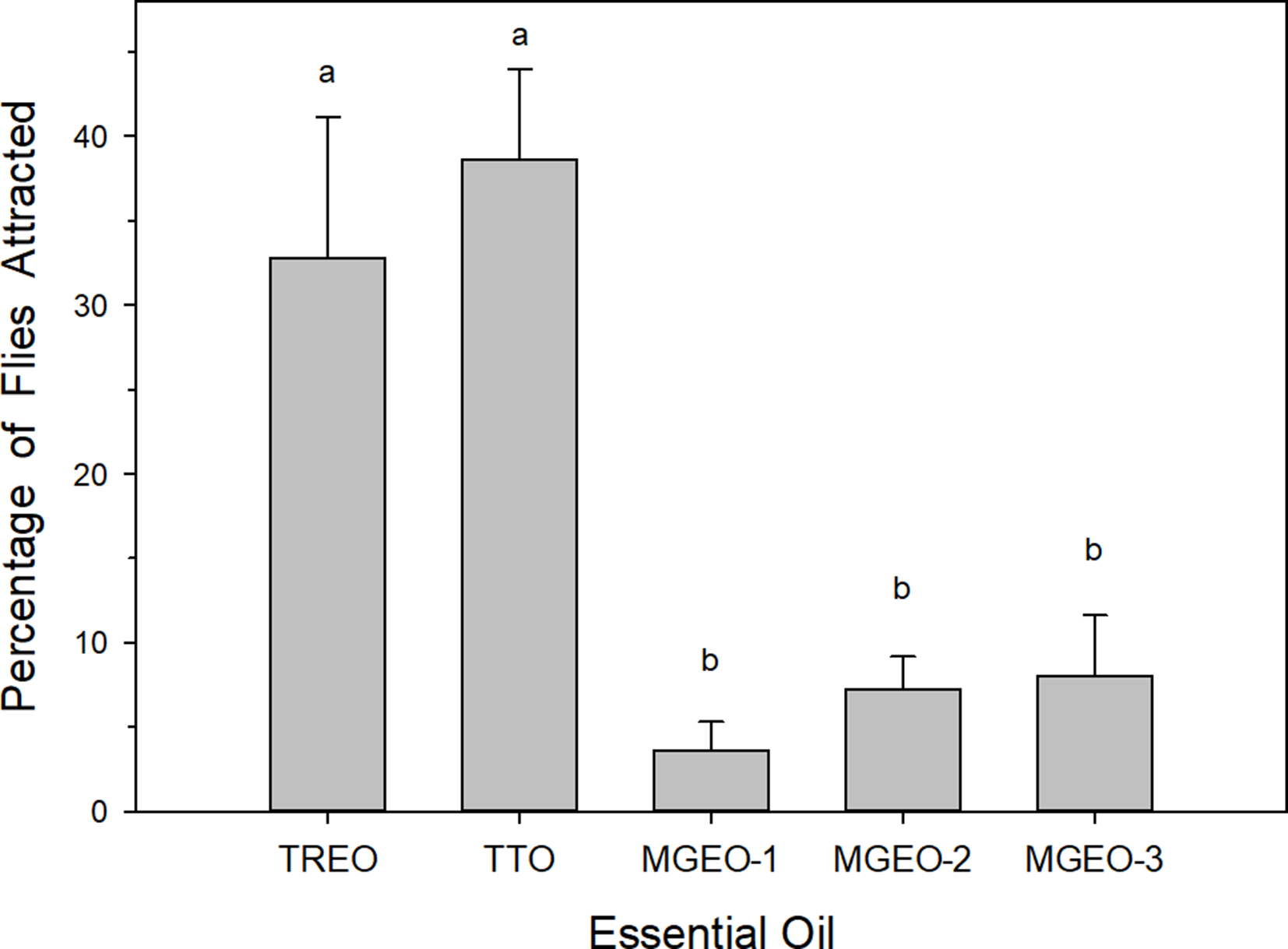
Attraction (mean ± SE) of the male Mediterranean fruit fly, Ceratitis capitata, to essential oils presented in short-range bioassays: TREO, TTO (a strong medfly attractant), 21 and 3 sources of MGEO 1-3 (a mild medfly attractant). 31 Bars topped with the same letter are not significantly different (Tukey’s Honest Significant Difference mean separation, P < 0.05). MGEO, mastic gum essential oil; TREO, Tetradenia riparia essential oil; TTO, tea tree oil.
With other essential oils studied, the attraction of C. capitata typically has been attributed to emissions of α-copaene or linalool 5,6,17,18,20 ; however, TREO is low in α-copaene, and linalool was not detected. Additional laboratory studies are needed to determine which components of TREO confer the attractant activity. Since fenchone and δ-cadinene are the most prominent, the examination of these 2 compounds is warranted. In addition, formulation of TREO lures is needed for field testing, and combinations of TREO lures with other medfly attractants should be evaluated for synergism, as has been achieved with kairomones for other insect pests. 32
Fenchone occurs in 2 enantiomers and the biological activity of the 2 forms may be very different. Fenchone appears to be an attractant of C. capitata in the present study. However, fenchone is known to exhibit contact and fumigant toxicity against other pest insects (Spodoptera littoralis, Sitophilus oryzae, S. zeamais, and Prostephanus truncates) and mites (Dermatophagoides farina and D. pteronyssinus). 33 Furthermore, fenchone caused 2-fold greater fumigant toxicity than methyl bromide against the land snail, Theba pisana. 34 Therefore, detailed studies are needed to determine the effects of each fenchone enantiomer on different taxa, including C. capitata.
Experimental
Plant Material
Plants were propagated by cuttings from a single clone of T. riparia and grown in 19 L containers in a controlled-environment greenhouse at the South Mississippi Branch Experiment Station in Poplarville, MI, USA (30.8364°N, 89.5455°W). Vegetative shoots were harvested from the plants on February 28, 2010. Plant material was air-dried for 3 weeks inside an air-conditioned building (25 °C maximum). The dried plant material was placed loosely into cardboard boxes and delivered to the USDA Natural Products Utilization Research facility in Oxford, MS, USA, for further processing. Voucher specimen #91 was deposited at the South Mississippi Branch Experiment Station for future reference.
Isolation of the Essential Oil
The aerial parts of T. riparia were subjected to hydrodistillation using a Clevenger type apparatus for 3 hours. The essential oil was dried over anhydrous sodium sulfate and stored at −4 °C until used. The yield of TREO was 0.8% (v/w).
Essential Oil Analysis
TREO was analyzed by an Agilent 5975 GC-Mass Selective Detector (MSD) system equipped with an Innowax FSC (60 m × 0.25 mm, 0.25 µm film thickness) column. The GC oven temperature was set at 60 °C for 10 minutes and programmed to 220 °C at a rate of 4 °C/min and kept constant at 220 °C for 10 minutes and then programmed to 240 °C at a rate of 1 °C/min. Helium was used as carrier gas (0.8 mL/min) and the split ratio was adjusted at 40:1. The injector temperature was set at 250 °C. The mass spectra were recorded at 70 eV, and the mass range was from m/z 35 to 450. The GC analysis was carried out using an Agilent 6890N GC system. FID detector temperature was 300 °C. A simultaneous autoinjection was done on a duplicate of the same column applying the same operational conditions in order to obtain the same elution order with GC-MS. Relative percentage amounts of the separated compounds were calculated from FID chromatograms.
Identification of the essential oil components was carried out by comparison of their relative retention times with those of authentic samples or by comparison of their relative retention index to a series of n-alkanes. Computer matching against commercial (Wiley GC-MS Library, MassFinder 3 Library) 35,36 and in-house “Başer Library of Essential Oil Constituents” built up by genuine compounds and components of known oils, as well as MS literature data, 37,38 was used for the identification.
Short-Range Attraction Bioassays
Sterile male C. capitata were used in this study. Source of insects, rearing procedures, and short-range laboratory bioassays were as described previously. 21,31 All observations were carried out at room temperature in small collapsible cages (20.3 × 20.3 × 20.3 cm) into which 50 flies were introduced 1 hour prior to the start of each experiment. Assays were initiated by introducing a Petri dish (53 mm diameter × 12 mm height) containing the test chemical (10 µL of a 10% dilution in acetone) applied to a filter paper disk (Whatman #1, 3.5 cm diameter). Each test used a separate cage to observe fly response to (1) TREO, (2) TTO, a known strong attractant, 21 obtained from Melaleuca alternifolia (Maiden and Betche) Cheel. (Essential Oil India-SAT Group, Kannauj, India), and (3) MGEO, a mild attractant 31 (obtained from 3 sources of Pistacia lentiscus L. var. chia), as described in Tabanca et al. 31 After 30 minutes, the response was recorded as the number of flies within a Petri dish, which was then converted to the percentage of flies attracted. Tests were replicated 5 times, and the position of the cages was randomized between replicate runs. Analysis of variance, followed by mean separation with Tukey test (P < 0.05), was used to analyze results with Systat Software (ver. 14; Systat Software, Inc., San Jose, CA, USA).
Footnotes
Acknowledgments
The authors are grateful to Micah Gill (USDA-ARS, Miami, FL, USA) for bioassays and to Monica Blanco, Amanda Perez-Castro and Sean Brown (USDA-ARS, Miami, FL, USA) for technical assistance, and to Dr Charles Cantrell (USDA-ARS, NPURU, Oxford, MS, USA) and Dr Jeremy Pickens (Auburn University, Ornamental Horticulture Research Center, Mobile, AL, USA) for helpful suggestions, and journal reviewers/editors for critical reviewers of an earlier version of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this study was supported by the Mississippi Agricultural and Forestry Experiment Station and bythe United States Department of Agriculture (Project Number: 6038-22000-006-00-D).
