Abstract
Indole-3-acetonitrile-6-
“Ban langen” (
The indole scaffold, as a privileged structure, is considered as one of the most promising heterocycles in drug discovery and embedded within the backbones of numerous pharmaceuticals and biologically important natural alkaloids. 17 Indole derivatives possess versatile biological activities, including antiviral, anti-inflammatory, antibacterial, antimalarial, anticonvulsant. and anticancer, etc. 18 -21 Due to their impressive natural structures and significant biological profiles, indole derivatives constantly call for the development of methods for their innovative synthetic strategies. Since the first synthesis of indole was completed by Adolf von Baeyer in 1866, 22 many efficient synthetic approaches have been successfully discovered and applied for the construction of indole and its complex derivatives, such as classical name reactions represented by Fischer indole synthesis, 23 organic electrosynthesis, 24 catalytic asymmetric dearomatization reactions, 25 organocatalytic asymmetric synthesis, 26 transition-metal-catalyzed C-H functionalization, 27 aerobic oxidative functionalization, 28 and photoredox catalysis. 29 Nevertheless, novel strategies for the synthesis of indole derivatives are still urgently needed.
In order to clarify the antiviral active constituents of
At present, it is time-consuming, laborious, and inefficient to extract enough amount of this indole alkaloid
Results and Discussion
Our retrosynthetic analysis is illustrated in Figure 1. We envisioned the key glycosylation formation via nucleophilic attack of 6-hydroxyindole-3-acetonitrile

Retrosynthetic strategy for synthesizing 3-indoleacetonitrile glucoside 1.
Preparation of the target compound

Synthetic route of 3-indoleacetonitrile glucoside 1.
Optimization of Transforming the Trimethylamine Moiety to the Cyano Group.

Abbreviations: CH2Cl2, dichloromethane; H2O, water; KCN, potassium cyanide; KF, potassium fluoride; MeOH, methanol; NaCN, sodium cyanide; NaF, sodium fluoride; TBAF, tetrabutylammonium fluoride; THF, tetrahydrofuran; TMSCN, trimethylsilyl cyanide.
Initial scouting of cyanomethylation was carried out by using sodium cyanide or potassium cyanide as a cyanide source by examining an array of conditions reported in the literature (Table 1, entry
It was another difficulty for us to perform the crucial glycosylation reaction, although various methods were developed for the construction of
Optimizing the Condition of Preparation of

Abbreviations: Ag2O, silver (I) oxide; CHCl3, chloroform; CH3CN, acetonitrile; DMF, dimethylformamide; H2O, water; K2CO3, potassium carbonate; LiOH, lithium hydroxide; TBAB, tetrabutylammonium bromide.
Having established an efficient method to synthesize the 3-indoleacetonitrile glucoside as described above, we also had access to the 3-indoleacetonitrile 4-position derivative

Synthetic route of cappariloside A.
The synthesized compounds
Anti-Influenza Virus Efficacy of the Compounds 1, 8, and 16 In Vitro. a

Abbreviations: CPE, cytopathic effect; EC50, half-maximal effective concentration; OSC, oseltamivir carboxylate; TC0, 50% cytotoxic concentration.
aThe effect of compounds was determined by measuring the survival of Madin-Darby canine kidney cells infected with the influenza A virus using the CPE assay.
bEC50 represents 50% effective concentration.
cTC0 represents the maximal nontoxic concentration, determined by CPE assay.
Compound
Conclusions
In conclusion, we accomplished a facile and efficient synthesis of indole-3-acetonitrile-6-
Experimental
General
The chemicals and all solvents used were of analytical grade without further purification unless otherwise noted. Reactions were monitored by thin-layer chromatography using Qing Dao Hai Yang GF254 silica gel plates (5 × 10 cm); zones were detected visually under ultraviolet (UV) irradiation (254 nm). 1H NMR spectra and 13C NMR were recorded on a Bruker NMR AVANCE 400 (400 MHz) or a Bruker NMR AVANCE 500 (500 MHz), using deuterated chloroform (CDCl3) or dimethylsulfoxide (DMSO)-
Synthesis of Indole-3-acetonitrile-6-O -β -d -glucopyranoside (Compound 1)
Synthesis of 1-(6-(benzyloxy)-1H-indol-3-yl)-N ,N -dimethylmethanamine (Compound
6
)
A 500 mL round-bottomed flask was charged with 37% formaldehyde solution (5.32 g, 4.92 mL, 65.59 mmol), dimethylamine 40% w/w in H2O (9.5 g, 84.42 mmol), acetic acid (100 mL), and 1,4-dioxane (100 mL). A solution of 6-benzyloxyindole (14.5 g, 64.94 mmol) in 1,4-dioxane (100 mL) was added dropwise while keeping the internal temperature between 0 °C and 5 °C. The reaction mixture was then stirred for 2 hours and kept at room temperature for 36 hours. The mixture was diluted with H2O (500 mL) and adjusted to pH 8-9 with aqueous sodium hydroxide (NaOH) solution (10%). The resulting precipitate was collected by filtration, and the filter cake was washed with ethyl acetate several times, dried in vacuum to afford the compound
Synthesis of 2-(6-(benzyloxy)-1H-indol-3-yl) acetonitrile (Compound
4
)
Methyl iodide (6.68 g, 2.93 ml, 47.08 mmol) was added to a suspension of compound
Synthesis of 2-(6-hydroxy-1H-indol-3-yl) acetonitrile (Compound
3
)
A 250 mL single-necked round-bottomed flask was charged with compound
Synthesis of (2R ,3R ,4S ,5R ,6S )-2-(acetoxymethyl)-6-((3-(cyanomethyl)-1H-indol-6-yl)oxy) tetrahydro-2H-pyran-3,4,5-triyl triacetate (Compound
8
)
A 500 mL round-bottomed flask was charged with compound
MP: 289.8-291.2 °C.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
HRMS (ESI): [M + Na]+ calcd for C24H27N2O 10 Na, 525.1480.1660; found, 525.1485 (supplemental Figure S16).
Deprotection of Compound
8
(Compound
1
)
LiOH·H2O (5.41 g, 129 mmol) was slowly added to a solution of ester (10.8 g, 215 mmol) in THF-MeOH-H2O (180 mL,
MP: 240.6 °C-242.1 °C.
13C NMR (125 MHz, DMSO-
HRMS (ESI):
Synthesis of 2-(4-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl) tetrahydro-2H-pyran-2-yl)oxy)-1H-indol-3-yl) acetonitrile 15 (cappariloside A)
A 500 mL round-bottomed flask was charged with 37% formaldehyde solution (3.83 mL, 50.5 mmol), dimethylamine 40% w/w in H2O (7.32 g, 65 mmol), acetic acid (100 mL), and 1,4-dioxane (100 mL) at 0 °C. A solution of 6-benzyloxyindole (11.16 g, 50 mmol) in 1,4-dioxane (100 mL) was added dropwise while keeping the internal temperature between 0 °C and 5 °C. The reaction mixture was then stirred for 2 hours and kept at room temperature for 36 hours. The mixture was diluted with H2O (500 mL) and adjusted to pH 8-9 with aqueous NaOH solution (10%). The resulting precipitate was collected by filtration, and the filter cake was washed with diethyl ether, dried in vacuum to afford the product
Methyl iodide (8.02 g, 3.52 mL, 56.5 mmol) was added to a suspension of compound
A 100 mL single-necked round-bottomed flask was charged with compound
A mixture of compound
LiOH·H2O (503.5 mg, 12 mmol) was slowly added to a solution of compound
MP: 234.8 °C-236.7 °C.
1H NMR (500 MHz, DMSO-
13C NMR (125 MHz, DMSO-
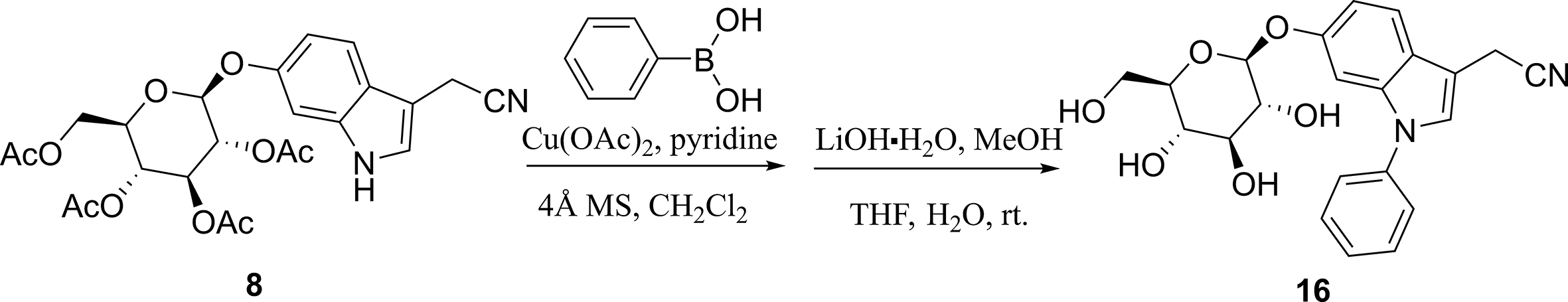
Synthesis of 2-(1-phenyl-6-(((2S,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl) tetrahydro-2H-pyran-2-yl)oxy)-1H-indol-3-yl)acetonitrile (Compound
16
)
A suspension of phenylboronic acid (73.1 mg, 0.6 mmol, 2 equiv.), copper (II) acetate (109 mg, 0.6 mmol, 2 equiv.), pyridine (95 mg, 1.2 mmol, 4 equiv.), and 4 Å molecular sieves (200 mg) in CH2Cl2 (5 mL) was stirred for 5 minutes at room temperature. To this stirring suspension was added compound
LiOH·H2O (50.4 mg, 1.2 mmol, 6 equiv.) was slowly added to a solution of the resulting ester (120.6 mg, 0.21 mmol, 1 equiv.) in THF/MeOH/H2O (3 mL,
MP: 256.4 °C-258.2 °C.
1H NMR (400 MHz, DMSO-
13C NMR (125 MHz, CDCl3):
HRMS (ESI):
CPE Assay Protocol
In CPE assay, MDCK cells were grown to a confluent monolayer in a 96-well culture plate at a concentration of 5 × 104 cells/well for 24 hours. The medium was removed, and the cells were rinsed twice. For the anti-influenza activity assay, an infectious virus at 100 TCID50 was inoculated into the MDCK cells, which were then incubated for 2 hours at 37 °C in 5% carbon dioxide (CO2). The virus supernatant was then removed, followed by the addition of serial 2-fold dilutions of antiviral compounds in Dulbecco’s modified Eagle medium containing 1.5 µg/mL trypsin. After being incubated at 34 °C in 5% CO2 for 48 hours, the infected cells displayed 100% CPE under the microscope, and the CPE percentages in the antiviral compound-treated groups were recorded. The EC50 of virus-induced CPE was detected by the Reed-Muench method. All data are obtained for at least 3 independent experiments.
Cytotoxicity Assay
Cytotoxicity of compounds was assessed by MTT assay. MDCK cells on 96-well plates were washed with sterile phosphate-buffered saline. Then 100 µL of compounds in serum-free MEM at 2-fold dilution was added to the cells, and the cells were incubated in CO2 at 37 °C. Cell controls were also performed. After 48 hours, 5 mg/mL of fresh MTT was added to each well, and the plates were incubated at 37 °C for 4 hours. Afterward, the medium was removed and formazan crystal was dissolved in DMSO (100 µL per well). Absorbance of each well at 490 nm was read by a CLARIOstar multi-mode microplate reader (BMG Labtech, Germany). The cell viability (%) = OD of compound well/average OD of control wells. The TC50 of each compound was obtained using a nonlinear regression model in GraphPad Prism 6.
Supplemental Material
Supplementary file 1 - Supplemental material for Multigram Scale Synthesis and Anti-Influenza Activity of 3-Indoleacetonitrile Glucosides
Supplemental material, Supplementary file 1, for Multigram Scale Synthesis and Anti-Influenza Activity of 3-Indoleacetonitrile Glucosides by Jianghong Dong, Fengmei Yan, Shuang Li, Zewen Li and Yuanhang Qin in Natural Product Communications
Footnotes
Acknowledgments
We thank Professor Yang Zifeng’s Group in State Key Laboratory of Respiratory Disease of China for the help in bioactivity assay.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: this work was supported by the Key Science and Technology Program of Henan Province, China (No.182102310258).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
